Abstract
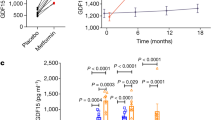
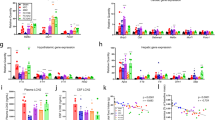
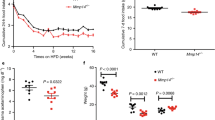
Anorexia and weight loss are part of the wasting syndrome of late-stage cancer, are a major cause of morbidity and mortality in cancer, and are thought to be cytokine mediated. Macrophage inhibitory cytokine-1 (MIC-1) is produced by many cancers. Examination of sera from individuals with advanced prostate cancer showed a direct relationship between MIC-1 abundance and cancer-associated weight loss. In mice with xenografted prostate tumors, elevated MIC-1 levels were also associated with marked weight, fat and lean tissue loss that was mediated by decreased food intake and was reversed by administration of antibody to MIC-1. Additionally, normal mice given systemic MIC-1 and transgenic mice overexpressing MIC-1 showed hypophagia and reduced body weight. MIC-1 mediates its effects by central mechanisms that implicate the hypothalamic transforming growth factor-β receptor II, extracellular signal–regulated kinases 1 and 2, signal transducer and activator of transcription-3, neuropeptide Y and pro-opiomelanocortin. Thus, MIC-1 is a newly defined central regulator of appetite and a potential target for the treatment of both cancer anorexia and weight loss, as well as of obesity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Tisdale, M.J. Cachexia in cancer patients. Nat. Rev. Cancer 2, 862–871 (2002).
Huhmann, M.B. & Cunningham, RS. Importance of nutritional screening in treatment of cancer-related weight loss. Lancet Oncol. 6, 334–343 (2005).
Marks, D.L., Ling, N. & Cone, RD. Role of the central melanocortin system in cachexia. Cancer Res. 61, 1432–1438 (2001).
Rubin, H. Cancer cachexia: its correlations and causes. Proc. Natl. Acad. Sci. USA 100, 5384–5389 (2003).
Rall, L.C. & Roubenoff, R. Rheumatoid cachexia: metabolic abnormalities, mechanisms and interventions. Rheumatology (Oxford) 43, 1219–1223 (2004).
Anker, S.D. & Sharma, R. The syndrome of cardiac cachexia. Int. J. Cardiol. 85, 51–66 (2002).
Bootcov, M.R. et al. MIC-1, a novel macrophage inhibitory cytokine, is a divergent member of the TGF-β superfamily cluster. Proc. Natl. Acad. Sci. USA 94, 11514–11519 (1997).
Welsh, J.B. et al. Large-scale delineation of secreted protein biomarkers overexpressed in cancer tissue and serum. Proc. Natl. Acad. Sci. USA 100, 3410–3415 (2003).
Buckhaults, P. et al. Secreted and cell surface genes expressed in benign and malignant colorectal tumours. Cancer Res. 61, 6996–7001 (2001).
Koopmann, J. et al. Serum macrophage inhibitory cytokine 1 as a marker of pancreatic and other periampullary cancers. Clin. Cancer Res. 10, 2386–2392 (2004).
Bauskin, A.R. et al. The propeptide mediates formation of stromal stores of proMIC-1: Role in determining prostate cancer outcome. Cancer Res. 65, 2330–2336 (2005).
Hsiao, E.C. et al. Characterization of growth-differentiation factor 15, a transforming growth factor β superfamily member induced following liver injury. Mol. Cell. Biol. 20, 3742–3751 (2000).
Schober, A. et al. Expression of growth differentiation factor-15/ macrophage inhibitory cytokine-1 (GDF-15/MIC-1) in the perinatal, adult, and injured rat brain. J. Comp. Neurol. 439, 32–45 (2001).
Bauskin, A.R. et al. Role of MIC-1 in tumorigenesis and diagnosis of cancer. Cancer Res. 66, 4983–4986 (2006).
Kamel, H.K., Maas, D. & Duthie, E.H. Role of hormones in the pathogenesis and management of sarcopenia. Drugs Aging 19, 865–877 (2002).
Rosen, C.J. et al. Congenic mice with low serum IGF-I have increased body fat, reduced bone mineral density, and an altered osteoblast differentiation program. Bone 35, 1046–1058 (2004).
Pfitzenmaier, J. et al. Elevation of cytokine levels in cachectic patients with prostate carcinoma. Cancer 97, 1211–1216 (2003).
Thompson, I.M. et al. Operating characteristics of prostate-specific antigen in men with an initial PSA level of 3.0 ng/ml or lower. J. Am. Med. Assoc. 294, 66–70 (2005).
Kovesdy, C.P., Anderson, J.E. & Kalantar-Zadeh, K. Inverse association between lipid levels and mortality in men with chronic kidney disease who are not yet on dialysis: effects of case mix and the malnutrition-inflammation-cachexia syndrome. J. Am. Soc. Nephrol. 18, 304–311 (2007).
Apple, F.S., Murakami, M.M., Pearce, L.A. & Herzog, C.A. Predictive value of cardiac troponin I and T for subsequent death in end-stage renal disease. Circulation 106, 2941–2945 (2002).
Naylor, J.L. et al. Further evidence that the blood/brain barrier impedes paraquat entry into the brain. Hum. Exp. Toxicol. 14, 587–594 (1995).
Karl, T. et al. Y1 receptors regulate aggressive behavior by modulating serotonin pathways. Proc. Natl. Acad. Sci. USA 101, 12742–12747 (2004).
Bouret, S.G. et al. Formation of projection pathways from the arcuate nucleus of the hypothalamus to hypothalamic regions implicated in the neural control of feeding behavior in mice. J. Neurosci. 24, 2797–2805 (2004).
Lin, E.J. et al. Combined deletion of Y1, Y2, and Y4 receptors prevents hypothalamic neuropeptide Y overexpression–induced hyperinsulinemia despite persistence of hyperphagia and obesity. Endocrinology 147, 5094–5101 (2006).
Paxinos, G. & Franklin, K.B.J. The mouse brain in stereotaxic coordinates (Academic Press, San Diego, USA, 2001).
Acknowledgements
This work was supported by grants from the National Health and Medical Research Council of Australia (NHMRC), Cancer Council New South Wales, the Richard M. Lucas Foundation and a New South Wales Health Research and Development Infrastructure grant. A.S. and H.H. are both recipients of NHMRC Fellowships and D.A.B. is a Neil Hamilton Fairley Postdoctoral Fellow of the NHMRC. We would like to thank D. Hume (Institute for Molecular Biosciences, The University of Queensland) for the gift of the promoter construct used to create the transgene for the fmsMIC transgenic mice and P. Sawchenko for critical evaluation of this manuscript. This work is dedicated to the memory of Mary Christine Kennedy.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
S.N.B., D.A.B., and A.R.B. declare that St. Vincent's Hospital Sydney, Ltd. has taken out patents and provisional patents covering some aspects of the area of MIC-1 research. Under this institution's intellectual property policy, should aspects of MIC-1 be commercialized and should financial returns flow, the inventors would receive a share of those returns, in line with the institutional policy.
Supplementary information
Supplementary Text and Figures
Supplementary Figs. 1–8, Supplementary Table 1 and Supplementary Methods (PDF 442 kb)
Rights and permissions
About this article
Cite this article
Johnen, H., Lin, S., Kuffner, T. et al. Tumor-induced anorexia and weight loss are mediated by the TGF-β superfamily cytokine MIC-1. Nat Med 13, 1333–1340 (2007). https://doi.org/10.1038/nm1677
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm1677
This article is cited by
-
Cancer cachexia: molecular mechanisms and treatment strategies
Journal of Hematology & Oncology (2023)
-
Context-dependent TGFβ family signalling in cell fate regulation
Nature Reviews Molecular Cell Biology (2023)
-
A class of secreted mammalian peptides with potential to expand cell-cell communication
Nature Communications (2023)
-
Fully implantable and battery-free wireless optoelectronic system for modulable cancer therapy and real-time monitoring
npj Flexible Electronics (2023)
-
Cancer-associated cachexia — understanding the tumour macroenvironment and microenvironment to improve management
Nature Reviews Clinical Oncology (2023)