Abstract
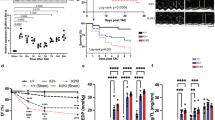
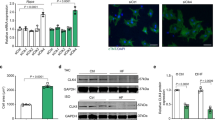
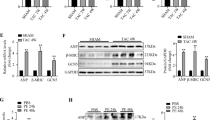
The serine-threonine kinases Pim-1 and Akt regulate cellular proliferation and survival. Although Akt is known to be a crucial signaling protein in the myocardium, the role of Pim-1 has been overlooked. Pim-1 expression in the myocardium of mice decreased during postnatal development, re-emerged after acute pathological injury in mice and was increased in failing hearts of both mice and humans. Cardioprotective stimuli associated with Akt activation induced Pim-1 expression, but compensatory increases in Akt abundance and phosphorylation after pathological injury by infarction or pressure overload did not protect the myocardium in Pim-1–deficient mice. Transgenic expression of Pim-1 in the myocardium protected mice from infarction injury, and Pim-1 expression inhibited cardiomyocyte apoptosis with concomitant increases in Bcl-2 and Bcl-XL protein levels, as well as in Bad phosphorylation levels. Relative to nontransgenic controls, calcium dynamics were significantly enhanced in Pim-1–overexpressing transgenic hearts, associated with increased expression of SERCA2a, and were depressed in Pim-1–deficient hearts. Collectively, these data suggest that Pim-1 is a crucial facet of cardioprotection downstream of Akt.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Wang, Z. et al. Pim-1: a serine/threonine kinase with a role in cell survival, proliferation, differentiation and tumorigenesis. J. Vet. Sci. 2, 167–179 (2001).
Bachmann, M. & Moroy, T. The serine/threonine kinase Pim-1. Int. J. Biochem. Cell Biol. 37, 726–730 (2005).
Aho, T.L. et al. Pim-1 kinase promotes inactivation of the pro-apoptotic Bad protein by phosphorylating it on the Ser112 gatekeeper site. FEBS Lett. 571, 43–49 (2004).
Hammerman, P.S., Fox, C.J., Birnbaum, M.J. & Thompson, C.B. Pim and Akt oncogenes are independent regulators of hematopoietic cell growth and survival. Blood 105, 4477–4483 (2005).
Pekarsky, Y. et al. Tcl1 enhances Akt kinase activity and mediates its nuclear translocation. Proc. Natl. Acad. Sci. USA 97, 3028–3033 (2000).
Shiraishi, I. et al. Nuclear targeting of Akt enhances kinase activity and survival of cardiomyocytes. Circ. Res. 94, 884–891 (2004).
Krumenacker, J.S., Narang, V.S., Buckley, D.J. & Buckley, A.R. Prolactin signaling to pim-1 expression: a role for phosphatidylinositol 3-kinase. J. Neuroimmunol. 113, 249–259 (2001).
Krishnan, N., Pan, H., Buckley, D.J. & Buckley, A. Prolactin-regulated pim-1 transcription: identification of critical promoter elements and Akt signaling. Endocrine 20, 123–130 (2003).
Koike, N., Maita, H., Taira, T., Ariga, H. & Iguchi-Ariga, S.M. Identification of heterochromatin protein 1 (HP1) as a phosphorylation target by Pim-1 kinase and the effect of phosphorylation on the transcriptional repression function of HP1(1). FEBS Lett. 467, 17–21 (2000).
Sussman, M.A. et al. Myofibril degeneration caused by tropomodulin overexpression leads to dilated cardiomyopathy in juvenile mice. J. Clin. Invest. 101, 51–61 (1998).
Bhattacharya, N. et al. Pim-1 associates with protein complexes necessary for mitosis. Chromosoma 111, 80–95 (2002).
Camper-Kirby, D. et al. Myocardial Akt activation and gender: increased nuclear activity in females versus males. Circ. Res. 88, 1020–1027 (2001).
Domen, J., van der Lugt, N.M., Laird, P.W., Saris, C.J. & Berns, A. Analysis of Pim-1 function in mutant mice. Leukemia 7 (Suppl. 2), S108–S112 (1993).
Konietzko, U. et al. Pim kinase expression is induced by LTP stimulation and required for the consolidation of enduring LTP. EMBO J. 18, 3359–3369 (1999).
Domen, J. et al. Pim-1 levels determine the size of early B lymphoid compartments in bone marrow. J. Exp. Med. 178, 1665–1673 (1993).
Bullock, A.N., Debreczeni, J., Amos, A., Knapp, S. & Turk, B.E. Structure and substrate specificity of the Pim-1 kinase. J. Biol. Chem. 280, 41675–41682 (2005).
Palaty, C.K. et al. Identification of the autophosphorylation sites of the Xenopus laevis Pim-1 proto-oncogene-encoded protein kinase. J. Biol. Chem. 272, 10514–10521 (1997).
Subramaniam, A. et al. Tissue-specific regulation of the alpha-myosin heavy chain gene promoter in transgenic mice. J. Biol. Chem. 266, 24613–24620 (1991).
Sugden, P.H. & Clerk, A. Cellular mechanisms of cardiac hypertrophy. J. Mol. Med. 76, 725–746 (1998).
Izumo, S., Nadal-Ginard, B. & Mahdavi, V. Protooncogene induction and reprogramming of cardiac gene expression produced by pressure overload. Proc. Natl. Acad. Sci. USA 85, 339–343 (1988).
Katakami, N. et al. Role of pim-1 in smooth muscle cell proliferation. J. Biol. Chem. 279, 54742–54749 (2004).
Hoefnagel, J.J. et al. Distinct types of primary cutaneous large B-cell lymphoma identified by gene expression profiling. Blood 105, 3671–3678 (2004).
Ionov, Y. et al. Pim-1 protein kinase is nuclear in Burkitt's lymphoma: nuclear localization is necessary for its biologic effects. Anticancer Res. 23, 167–178 (2003).
Evans, K.E. & Fox, S.W. Interleukin-10 inhibits osteoclastogenesis by reducing NFATc1 expression and preventing its translocation to the nucleus. BMC Cell Biol. 8, 4 (2007).
Barre, B., Avril, S. & Coqueret, O. Opposite regulation of myc and p21waf1 transcription by STAT3 proteins. J. Biol. Chem. 278, 2990–2996 (2003).
Poolman, R.A., Gilchrist, R. & Brooks, G. Cell cycle profiles and expressions of p21CIP1 and P27KIP1 during myocyte development. Int. J. Cardiol. 67, 133–142 (1998).
Phoon, C.K. et al. Embryonic heart failure in NFATc1−/− mice: novel mechanistic insights from in utero ultrasound biomicroscopy. Circ. Res. 95, 92–99 (2004).
Torella, D. et al. Cardiac stem cell and myocyte aging, heart failure, and insulin-like growth factor-1 overexpression. Circ. Res. 94, 514–524 (2004).
Dupays, L. et al. Dysregulation of connexins and inactivation of NFATc1 in the cardiovascular system of Nkx2–5 null mutants. J. Mol. Cell. Cardiol. 38, 787–798 (2005).
Lilly, M., Sandholm, J., Cooper, J.J., Koskinen, P.J. & Kraft, A. The PIM-1 serine kinase prolongs survival and inhibits apoptosis-related mitochondrial dysfunction in part through a bcl-2-dependent pathway. Oncogene 18, 4022–4031 (1999).
Fujiwara, Y. et al. Inhibition of the PI3 kinase/Akt pathway enhances doxorubicin-induced apoptotic cell death in tumor cells in a p53-dependent manner. Biochem. Biophys. Res. Commun. 340, 560–566 (2006).
Wingett, D., Long, A., Kelleher, D. & Magnuson, N.S. pim-1 proto-oncogene expression in anti-CD3-mediated T cell activation is associated with protein kinase C activation and is independent of Raf-1. J. Immunol. 156, 549–557 (1996).
Rahman, Z., Yoshikawa, H., Nakajima, Y. & Tasaka, K. Down-regulation of Pim-1 and Bcl-2 is accompanied with apoptosis of interleukin-6-depleted mouse B-cell hybridoma 7TD1 cells. Immunol. Lett. 75, 199–208 (2001).
Eiken, H.G. et al. Myocardial gene expression of leukaemia inhibitory factor, interleukin-6 and glycoprotein 130 in end-stage human heart failure. Eur. J. Clin. Invest. 31, 389–397 (2001).
Khaleghpour, K., Pyronnet, S., Gingras, A.C. & Sonenberg, N. Translational homeostasis: eukaryotic translation initiation factor 4E control of 4E-binding protein 1 and p70 S6 kinase activities. Mol. Cell. Biol. 19, 4302–4310 (1999).
Hacker, H. & Karin, M. Regulation and function of IKK and IKK-related kinases. Sci. STKE 2006, re13 (2006).
Zhu, N. et al. CD40 signaling in B cells regulates the expression of the Pim-1 kinase via the NF-kappa B pathway. J. Immunol. 168, 744–754 (2002).
Krumenacker, J.S. et al. Prolactin-regulated apoptosis of Nb2 lymphoma cells: pim-1, bcl-2, and bax expression. Endocrine 9, 163–170 (1998).
Gude, N. et al. Akt promotes increased cardiomyocyte cycling and expansion of the cardiac progenitor cell population. Circ. Res. 99, 381–388 (2006).
Tsujita, Y. et al. Nuclear targeting of Akt antagonizes aspects of cardiomyocyte hypertrophy. Proc. Natl. Acad. Sci. USA 103, 11946–11951 (2006).
Takizawa, T. et al. Transcription of the SERCA2 gene is decreased in pressure-overloaded hearts: A study using in vivo direct gene transfer into living myocardium. J. Mol. Cell. Cardiol. 31, 2167–2174 (1999).
Prasad, A.M. et al. Phenylephrine hypertrophy, Ca2+-ATPase (SERCA2), and Ca2+ signaling in neonatal rat cardiac myocytes. Am. J. Physiol. Cell Physiol. 292, C2269–C2275 (2007).
Asahi, M. et al. Cardiac-specific overexpression of sarcolipin inhibits sarco(endo)plasmic reticulum Ca2+ ATPase (SERCA2a) activity and impairs cardiac function in mice. Proc. Natl. Acad. Sci. USA 101, 9199–9204 (2004).
Suarez, J. et al. Doxycycline inducible expression of SERCA2a improves calcium handling and reverts cardiac dysfunction in pressure overload-induced cardiac hypertrophy. Am. J. Physiol. Heart Circ. Physiol. 287, H2164–H2172 (2004).
Sakata, S. et al. Restoration of mechanical and energetic function in failing aortic-banded rat hearts by gene transfer of calcium cycling proteins. J. Mol. Cell. Cardiol. 42, 852–861 (2007).
del Monte, F. et al. Abrogation of ventricular arrhythmias in a model of ischemia and reperfusion by targeting myocardial calcium cycling. Proc. Natl. Acad. Sci. USA 101, 5622–5627 (2004).
Kim, Y.K. et al. Mechanism of enhanced cardiac function in mice with hypertrophy induced by overexpressed Akt. J. Biol. Chem. 278, 47622–47628 (2003).
Rota, M. et al. Nuclear targeting of Akt enhances ventricular function and myocyte contractility. Circ. Res. 97, 1332–1341 (2005).
Jacobs, M.D. et al. Pim-1 ligand-bound structures reveal the mechanism of serine/threonine kinase inhibition by LY294002. J. Biol. Chem. 280, 13728–13734 (2005).
Kato, T. et al. Atrial natriuretic peptide promotes cardiomyocyte survival by cGMP-dependent nuclear accumulation of zyxin and Akt. J. Clin. Invest. 115, 2716–2730 (2005).
Acknowledgements
This work was supported by US National Institutes of Health grants (5R01HL067245, 1R01HL091102 and 1P01HL085577) to M.A.S. and a US National Heart, Lung, and Blood Institute grant 1P01AG023071 to P.A. J.A.M. and N.G. are Fellows of the Rees-Stealy Research Foundation and the San Diego State University Heart Institute. We appreciate the contribution of P. Bonine for outstanding administrative assistance.
Author information
Authors and Affiliations
Contributions
J.A.M. planned and performed experiments and wrote the manuscript; M.R. and Y.M. planned and performed experiments; J.F., C.C., G.E., F.D., M.A., R.A. and S.S. performed experiments; G.N.E. and W.W. performed surgeries; K.F., J.J.M., C.C.G., A.L. and J.K. performed experiments and advised on the experimental approach; N.M. provided Pim1 cDNAs and advised on the technical proposal; A.B. provided Pim-KO animals; R.M.B. and S.R.H. provided human samples and advice; E.M.S. provided technical advice and advice on the experimental approach; P.A. provided advice on the experimental approach, rewriting and editing, and laboratory resources for experiments; M.A.S. supervised all experimental procedures and edited and composed the manuscript.
Corresponding author
Supplementary information
Supplementary Text and Figures
Supplementary Figs. 1–5, Supplementary Tables 1 and 2, and Supplementary Methods (PDF 15857 kb)
Rights and permissions
About this article
Cite this article
Muraski, J., Rota, M., Misao, Y. et al. Pim-1 regulates cardiomyocyte survival downstream of Akt. Nat Med 13, 1467–1475 (2007). https://doi.org/10.1038/nm1671
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm1671