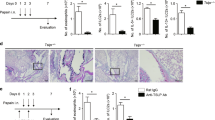
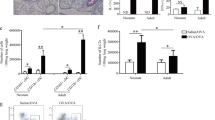
Abstract
Osteopontin (Opn) is important for T helper type 1 (TH1) immunity and autoimmunity. However, the role of this cytokine in TH2-mediated allergic disease as well as its effects on primary versus secondary antigenic encounters remain unclear. Here we demonstrate that OPN is expressed in the lungs of asthmatic individuals and that Opn-s, the secreted form of Opn, exerts opposing effects on mouse TH2 effector responses and subsequent allergic airway disease: pro-inflammatory at primary systemic sensitization, and anti-inflammatory during secondary pulmonary antigenic challenge. These effects of Opn-s are mainly mediated by the regulation of TH2-suppressing plasmacytoid dendritic cells (DCs) during primary sensitization and TH2-promoting conventional DCs during secondary antigenic challenge. Therapeutic administration of recombinant Opn during pulmonary secondary antigenic challenge decreased established TH2 responses and protected mice from allergic disease. These effects on TH2 allergic responses suggest that Opn-s is an important therapeutic target and provide new insight into its role in immunity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Mosmann, T.R., Cherwinski, H., Bond, M.W., Giedlin, M.A. & Coffman, R.L. Two types of murine helper T cell clone. I. Definition according to profiles of lymphokine activities and secreted proteins. J. Immunol. 136, 2348–2357 (1986).
Lucey, D.R., Clerici, M. & Shearer, G.M. Type 1 and type 2 cytokine dysregulation in human infectious, neoplastic, and inflammatory diseases. Clin. Microbiol. Rev. 9, 532–562 (1996).
Liu, A.H. Early intervention for asthma prevention in children. Allergy Asthma Proc. 23, 289–293 (2002).
Weber, G.F. & Cantor, H. The immunology of Eta-1/osteopontin. Cytokine Growth Factor Rev. 7, 241–248 (1996).
Patarca, R. et al. Structural and functional studies of the early T lymphocyte activation 1 (Eta-1) gene. Definition of a novel T cell-dependent response associated with genetic resistance to bacterial infection. J. Exp. Med. 170, 145–161 (1989).
Ashkar, S. et al. Eta-1 (osteopontin): an early component of type-1 (cell-mediated) immunity. Science 287, 860–864 (2000).
Patarca, R., Saavedra, R.A. & Cantor, H. Molecular and cellular basis of genetic resistance to bacterial infection: the role of the early T-lymphocyte activation-1/osteopontin gene. Crit. Rev. Immunol. 13, 225–246 (1993).
Jansson, M., Panoutsakopoulou, V., Baker, J., Klein, L. & Cantor, H. Cutting edge: attenuated experimental autoimmune encephalomyelitis in eta-1/osteopontin-deficient mice. J. Immunol. 168, 2096–2099 (2002).
Chabas, D. et al. The influence of the proinflammatory cytokine, osteopontin, on autoimmune demyelinating disease. Science 294, 1731–1735 (2001).
Hur, E.M. et al. Osteopontin-induced relapse and progression of autoimmune brain disease through enhanced survival of activated T cells. Nat. Immunol. 8, 74–83 (2007).
Gassler, N. et al. Expression of osteopontin (Eta-1) in Crohn disease of the terminal ileum. Scand. J. Gastroenterol. 37, 1286–1295 (2002).
Xu, G. et al. Role of osteopontin in amplification and perpetuation of rheumatoid synovitis. J. Clin. Invest. 115, 1060–1067 (2005).
D'Alfonso, S. et al. Two single-nucleotide polymorphisms in the 5′ and 3′ ends of the osteopontin gene contribute to susceptibility to systemic lupus erythematosus. Arthritis Rheum. 52, 539–547 (2005).
Niino, M., Kikuchi, S., Fukazawa, T., Yabe, I. & Tashiro, K. Genetic polymorphisms of osteopontin in association with multiple sclerosis in Japanese patients. J. Neuroimmunol. 136, 125–129 (2003).
Shinohara, M.L. et al. Osteopontin expression is essential for interferon-α production by plasmacytoid dendritic cells. Nat. Immunol. 7, 498–506 (2006).
Renkl, A.C. et al. Osteopontin functionally activates dendritic cells and induces their differentiation toward a Th1-polarizing phenotype. Blood 106, 946–955 (2005).
Kawamura, K. et al. Differentiation, maturation, and survival of dendritic cells by osteopontin regulation. Clin. Diagn. Lab. Immunol. 12, 206–212 (2005).
Weiss, J.M. et al. Osteopontin is involved in the initiation of cutaneous contact hypersensitivity by inducing Langerhans and dendritic cell migration to lymph nodes. J. Exp. Med. 194, 1219–1229 (2001).
Begum, M.D. et al. Suppression of the bacterial antigen-specific T cell response and the dendritic cell migration to the lymph nodes by osteopontin. Microbiol. Immunol. 51, 135–147 (2007).
Lambrecht, B.N. & Hammad, H. Taking our breath away: dendritic cells in the pathogenesis of asthma. Nat. Rev. Immunol. 3, 994–1003 (2003).
Humbles, A.A. et al. A critical role for eosinophils in allergic airways remodeling. Science 305, 1776–1779 (2004).
Walter, M.J., Kajiwara, N., Karanja, P., Castro, M. & Holtzman, M.J. Interleukin 12 p40 production by barrier epithelial cells during airway inflammation. J. Exp. Med. 193, 339–351 (2001).
Rimoldi, M. et al. Monocyte-derived dendritic cells activated by bacteria or by bacteria-stimulated epithelial cells are functionally different. Blood 106, 2818–2826 (2005).
Dahl, M.E., Dabbagh, K., Liggitt, D., Kim, S. & Lewis, D.B. Viral-induced T helper type 1 responses enhance allergic disease by effects on lung dendritic cells. Nat. Immunol. 5, 337–343 (2004).
Hansen, G., Berry, G., DeKruyff, R.H. & Umetsu, D.T. Allergen-specific TH1 cells fail to counterbalance TH2 cell-induced airway hyperreactivity but cause severe airway inflammation. J. Clin. Invest. 103, 175–183 (1999).
Krug, N. et al. T-cell cytokine profile evaluated at the single cell level in BAL and blood in allergic asthma. Am. J. Respir. Cell Mol. Biol. 14, 319–326 (1996).
Magnan, A.O. et al. Assessment of the Th1/Th2 paradigm in whole blood in atopy and asthma. Increased IFN-γ-producing CD8+ T cells in asthma. Am. J. Respir. Crit. Care Med. 161, 1790–1796 (2000).
de Heer, H.J. et al. Essential role of lung plasmacytoid dendritic cells in preventing asthmatic reactions to harmless inhaled antigen. J. Exp. Med. 200, 89–98 (2004).
Wakkach, A. et al. Characterization of dendritic cells that induce tolerance and T regulatory 1 cell differentiation in vivo. Immunity 18, 605–617 (2003).
Oriss, T.B. et al. Dynamics of dendritic cell phenotype and interactions with CD4+ T cells in airway inflammation and tolerance. J. Immunol. 174, 854–863 (2005).
Nakano, H., Yanagita, M. & Gunn, M.D. CD11c+B220+Gr-1+ cells in mouse lymph nodes and spleen display characteristics of plasmacytoid dendritic cells. J. Exp. Med. 194, 1171–1178 (2001).
Kohl, J. et al. A regulatory role for the C5a anaphylatoxin in type 2 immunity in asthma. J. Clin. Invest. 116, 783–796 (2006).
Idzko, M. et al. Local application of FTY720 to the lung abrogates experimental asthma by altering dendritic cell function. J. Clin. Invest. 116, 2935–2944 (2006).
Haeryfar, S.M. The importance of being a pDC in antiviral immunity: the IFN mission versus Ag presentation? Trends Immunol 26, 311–317 (2005).
Asselin-Paturel, C., Brizard, G., Pin, J.J., Briere, F. & Trinchieri, G. Mouse strain differences in plasmacytoid dendritic cell frequency and function revealed by a novel monoclonal antibody. J. Immunol. 171, 6466–6477 (2003).
Vermaelen, K.Y., Carro-Muino, I., Lambrecht, B.N. & Pauwels, R.A. Specific migratory dendritic cells rapidly transport antigen from the airways to the thoracic lymph nodes. J. Exp. Med. 193, 51–60 (2001).
Stock, P. et al. Induction of T helper type 1-like regulatory cells that express Foxp3 and protect against airway hyper-reactivity. Nat. Immunol. 5, 1149–1156 (2004).
Shinohara, M.L. et al. T-bet-dependent expression of osteopontin contributes to T cell polarization. Proc. Natl. Acad. Sci. USA 102, 17101–17106 (2005).
van Rijt, L.S. et al. In vivo depletion of lung CD11c+ dendritic cells during allergen challenge abrogates the characteristic features of asthma. J. Exp. Med. 201, 981–991 (2005).
Smit, J.J., Rudd, B.D. & Lukacs, N.W. Plasmacytoid dendritic cells inhibit pulmonary immunopathology and promote clearance of respiratory syncytial virus. J. Exp. Med. 203, 1153–1159 (2006).
Ochando, J.C. et al. Alloantigen-presenting plasmacytoid dendritic cells mediate tolerance to vascularized grafts. Nat. Immunol. 7, 652–662 (2006).
Blasius, A.L. et al. Bone marrow stromal cell antigen 2 is a specific marker of type I IFN-producing cells in the naive mouse, but a promiscuous cell surface antigen following IFN stimulation. J. Immunol. 177, 3260–3265 (2006).
Farkas, L., Kvale, E.O., Johansen, F.E., Jahnsen, F.L. & Lund-Johansen, F. Plasmacytoid dendritic cells activate allergen-specific Th2 memory cells: modulation by CpG oligodeoxynucleotides. J. Allergy Clin. Immunol. 114, 436–443 (2004).
Hessel, E.M. et al. Immunostimulatory oligonucleotides block allergic airway inflammation by inhibiting Th2 cell activation and IgE-mediated cytokine induction. J. Exp. Med. 202, 1563–1573 (2005).
Konno, S. et al. Level of osteopontin is increased after bee venom immunotherapy. J. Allergy Clin. Immunol. 115, 1317–1318 (2005).
Gavett, S.H. et al. Interleukin 12 inhibits antigen-induced airway hyperresponsiveness, inflammation, and Th2 cytokine expression in mice. J. Exp. Med. 182, 1527–1536 (1995).
McMillan, S.J., Xanthou, G. & Lloyd, C.M. Manipulation of allergen-induced airway remodelling by treatment with anti-TGF-β antibody: effect on the Smad signaling pathway. J. Immunol. 174, 5774–5780 (2005).
Gaga, M. et al. Eosinophils are a feature of upper and lower airway pathology in non-atopic asthma, irrespective of the presence of rhinitis. Clin. Exp. Allergy 30, 663–669 (2000).
Panoutsakopoulou, V. et al. Analysis of the relationship between viral infection and autoimmune disease. Immunity 15, 137–147 (2001).
McMillan, S.J., Bishop, B., Townsend, M.J., McKenzie, A.N. & Lloyd, C.M. The absence of interleukin 9 does not affect the development of allergen-induced pulmonary inflammation nor airway hyperreactivity. J. Exp. Med. 195, 51–57 (2002).
Acknowledgements
We thank S. Spyridakis for assistance in flow-cytometry, A. Agapaki for histology preparations and S. Pagakis for assistance with final figure preparation. We thank L. Liaw (Maine Medical Center Research Institute) for permission to use the Spp1-deficient mice. We are grateful to M. Doufexis, I. Scotiniotis, C. Tsatsanis and M. Aggelakopoulou for critical reading of the manuscript, and to C. Davos, K. Karalis and A. Tsouroplis for discussions. This work was supported by the Hellenic Ministries of Health and Education (V.P. and G.X.) and by a grant award from the Hellenic Ministry of Development, General Secretariat of Research and Technology (03ED750; V.P.). C.M.L. is supported by a Senior Fellowship from the Wellcome Trust (#057704). B.N.L. is supported by a Vidi grant from the Dutch Organization for Scientific Research. D.C.M.S. is supported by the Thorax Foundation.
Author information
Authors and Affiliations
Contributions
G.X. designed experiments, performed animal studies and immunohistochemistry, generated figures, analyzed data and wrote the manuscript. T.A. performed animal studies, tissue-culture experiments, generated figures and performed flow cytometry. M.S. assisted with the animal studies and tissue-culture experiments. D.C.M.S. assisted with the animal studies and image analysis. E.E. and M.G. performed bronchoscopies, and provided human lung biopsies and clinical characteristics of the individuals. B.N.L. provided antibodies, assisted with the design of experiments and participated in discussions. C.M.L. assisted with experimental design, writing and critical editing of the manuscript. V.P. provided crucial ideas, designed experiments, analyzed data, supervised the study and wrote the manuscript with G.X.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Xanthou, G., Alissafi, T., Semitekolou, M. et al. Osteopontin has a crucial role in allergic airway disease through regulation of dendritic cell subsets. Nat Med 13, 570–578 (2007). https://doi.org/10.1038/nm1580
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm1580
This article is cited by
-
Osteopontin: A Novel Therapeutic Target for Respiratory Diseases
Lung (2024)
-
MMP-3-mediated cleavage of OPN is involved in copper oxide nanoparticle-induced activation of fibroblasts
Particle and Fibre Toxicology (2023)
-
Osteopontin contributes to late-onset asthma phenotypes in adult asthma patients
Experimental & Molecular Medicine (2020)
-
Neutralizing antibody against osteopontin attenuates non-alcoholic steatohepatitis in mice
Journal of Cell Communication and Signaling (2020)
-
Heparin-binding epidermal growth factor contributes to COPD disease severity by modulating airway fibrosis and pulmonary epithelial–mesenchymal transition
Laboratory Investigation (2018)