Abstract
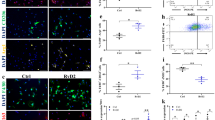
Skeletal muscle has the ability to achieve rapid repair in response to injury or disease1. Many individuals with Marfan syndrome (MFS), caused by a deficiency of extracellular fibrillin-1, exhibit myopathy and often are unable to increase muscle mass despite physical exercise. Evidence suggests that selected manifestations of MFS reflect excessive signaling by transforming growth factor (TGF)-β (refs. 2,3). TGF-β is a known inhibitor of terminal differentiation of cultured myoblasts; however, the functional contribution of TGF-β signaling to disease pathogenesis in various inherited myopathic states in vivo remains unknown4,5. Here we show that increased TGF-β activity leads to failed muscle regeneration in fibrillin-1–deficient mice. Systemic antagonism of TGF-β through administration of TGF-β–neutralizing antibody or the angiotensin II type 1 receptor blocker losartan normalizes muscle architecture, repair and function in vivo. Moreover, we show TGF-β–induced failure of muscle regeneration and a similar therapeutic response in a dystrophin-deficient mouse model of Duchenne muscular dystrophy. NOTE: In the version of this article initially published, the same panels were inadvertently used to show negative pSmad2/3 and periostin staining in muscle of Fbn1C1039G/+ mice treated with TGF-β‐neutralizing antibody in both the steady-state (Fig. 1a, right column, second and third rows, respectively) and muscle-regeneration (Fig. 1b, right column, third and fourth rows, respectively) experiments. In reality, these images only relate to the steady-state experiment (Fig. 1a). The intended images for Figure 1b are provided (red, pSmad2/3 staining; green, periostin staining). As both sets of images show negative staining in neutralizing antibody–treated Fbn1C1039G/+ mice, this does not alter any observations or conclusions discussed in the manuscript. The error has been corrected in the HTML and PDF versions of the article.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Change history
27 February 2007
In the version of this article initially published, the same panels were inadvertently used to show negative pSmad2/3 and periostin staining in muscle of Fbn1C1039G/+ mice treated with TGF-β‐neutralizing antibody in both the steady-state (Fig. 1a, right column, second and third rows, respectively) and muscle-regeneration (Fig. 1b, right column, third and fourth rows, respectively) experiments. In reality, these images only relate to the steady-state experiment (Fig. 1a). The intended images for Figure 1b are provided (red, pSmad2/3 staining; green, periostin staining). As both sets of images show negative staining in neutralizing antibody–treated Fbn1C1039G/+ mice, this does not alter any observations or conclusions discussed in the manuscript. The error has been corrected in the HTML and PDF versions of the article.
References
Charge, S.B. & Rudnicki, M.A. Cellular and molecular regulation of muscle regeneration. Physiol. Rev. 84, 209–238 (2004).
Ng, C.M. et al. TGF-beta-dependent pathogenesis of mitral valve prolapse in a mouse model of Marfan syndrome. J. Clin. Invest. 114, 1586–1592 (2004).
Neptune, E.R. et al. Dysregulation of TGF-beta activation contributes to pathogenesis in Marfan syndrome. Nat. Genet. 33, 407–411 (2003).
Allen, R.E. & Boxhorn, L.K. Inhibition of skeletal muscle satellite cell differentiation by transforming growth factor-beta. J. Cell. Physiol. 133, 567–572 (1987).
Olson, E.N., Sternberg, E., Hu, J.S., Spizz, G. & Wilcox, C. Regulation of myogenic differentiation by type beta transforming growth factor. J. Cell Biol. 103, 1799–1805 (1986).
Dietz, H.C. et al. Marfan syndrome caused by a recurrent de novo missense mutation in the fibrillin gene. Nature 352, 337–339 (1991).
Habashi, J.P. et al. Losartan, an AT1 antagonist, prevents aortic aneurysm in a mouse model of Marfan syndrome. Science 312, 117–121 (2006).
Heldin, C.H., Miyazono, K. & ten Dijke, P. TGF-beta signalling from cell membrane to nucleus through SMAD proteins. Nature 390, 465–471 (1997).
Li, Y. et al. Transforming growth factor-beta1 induces the differentiation of myogenic cells into fibrotic cells in injured skeletal muscle: a key event in muscle fibrogenesis. Am. J. Pathol. 164, 1007–1019 (2004).
Sato, K. et al. Improvement of muscle healing through enhancement of muscle regeneration and prevention of fibrosis. Muscle Nerve 28, 365–372 (2003).
Gosselin, L.E. et al. Localization and early time course of TGF-beta 1 mRNA expression in dystrophic muscle. Muscle Nerve 30, 645–653 (2004).
Judge, D.P. et al. Evidence for a critical contribution of haploinsufficiency in the complex pathogenesis of Marfan syndrome. J. Clin. Invest. 114, 172–181 (2004).
Goetsch, S.C., Hawke, T.J., Gallardo, T.D., Richardson, J.A. & Garry, D.J. Transcriptional profiling and regulation of the extracellular matrix during muscle regeneration. Physiol. Genomics 14, 261–271 (2003).
Reimann, J., Irintchev, A. & Wernig, A. Regenerative capacity and the number of satellite cells in soleus muscles of normal and mdx mice. Neuromuscul. Disord. 10, 276–282 (2000).
Jin, Y. et al. Expression of MyoD and myogenin in dystrophic mice, mdx and dy, during regeneration. Acta Neuropathol. (Berl.) 99, 619–627 (2000).
Lavoie, P. et al. Neutralization of transforming growth factor-beta attenuates hypertension and prevents renal injury in uremic rats. J. Hypertens. 23, 1895–1903 (2005).
Lim, D.S. et al. Angiotensin II blockade reverses myocardial fibrosis in a transgenic mouse model of human hypertrophic cardiomyopathy. Circulation 103, 789–791 (2001).
Yamazaki, M. et al. Expression of transforming growth factor-beta 1 and its relation to endomysial fibrosis in progressive muscular dystrophy. Am. J. Pathol. 144, 221–226 (1994).
Cohn, R.D. et al. Disruption of DAG1 in differentiated skeletal muscle reveals a role for dystroglycan in muscle regeneration. Cell 110, 639–648 (2002).
Lee, S.J. Regulation of muscle mass by myostatin. Annu. Rev. Cell Dev. Biol. 20, 61–86 (2004).
Zhu, X., Topouzis, S., Liang, L.F. & Stotish, R.L. Myostatin signaling through Smad2, Smad3 and Smad4 is regulated by the inhibitory Smad7 by a negative feedback mechanism. Cytokine 26, 262–272 (2004).
Wagner, K.R., McPherron, A.C., Winik, N. & Lee, S.J. Loss of myostatin attenuates severity of muscular dystrophy in mdx mice. Ann. Neurol. 52, 832–836 (2002).
Bogdanovich, S. et al. Functional improvement of dystrophic muscle by myostatin blockade. Nature 420, 418–421 (2002).
Tkatchenko, A.V. et al. Identification of altered gene expression in skeletal muscles from Duchenne muscular dystrophy patients. Neuromuscul. Disord. 11, 269–277 (2001).
Zhou, Y., Poczatek, M.H., Berecek, K.H. & Murphy-Ullrich, J.E. Thrombospondin 1 mediates angiotensin II induction of TGF-beta activation by cardiac and renal cells under both high and low glucose conditions. Biochem. Biophys. Res. Commun. 339, 633–641 (2006).
Delafontaine, P. & Akao, M. Angiotensin II as candidate of cardiac cachexia. Curr. Opin. Clin. Nutr. Metab. Care 9, 220–224 (2006).
Assereto, S. et al. Pharmacological rescue of the dystrophin-glycoprotein complex in Duchenne and Becker skeletal muscle explants by proteasome inhibitor treatment. Am. J. Physiol. Cell Physiol. 290, C577–C582 (2006).
Briguet, A., Courdier-Fruh, I., Foster, M., Meier, T. & Magyar, J.P. Histological parameters for the quantitative assessment of muscular dystrophy in the mdx-mouse. Neuromuscul. Disord. 14, 675–682 (2004).
Pereira, L. et al. Targetting of the gene encoding fibrillin-1 recapitulates the vascular aspect of Marfan syndrome. Nat. Genet. 17, 218–222 (1997).
Horsley, V., Jansen, K.M., Mills, S.T. & Pavlath, G.K. IL-4 acts as a myoblast recruitment factor during mammalian muscle growth. Cell 113, 483–494 (2003).
Acknowledgements
We would like to thank K. Wagner and S.-J. Lee for providing muscle samples of mdx Mstn−/− mice. We would like to thank P. Sponseller for providing human muscle samples. H.C. Dietz is an Investigator of the Howard Hughes Medical Institute and is also supported by the US National Institutes of Health, the Smilow Center for Marfan Syndrome Research, the Dana and Albert “Cubby” Broccoli Center for Aortic Diseases and the National Marfan Foundation.
Author information
Authors and Affiliations
Contributions
R.D.C. conducted most of the experiments and wrote the manuscript; C.E. participated in the analysis of the ex vivo muscle data; J.P.H. participated in the development and execution of treatment protocols for both fibrillin-1–deficient and mdx mice; A.A.S. participated in immunofluorescent analyses; E.C.K., M.G. and T.M.H. maintained the mouse colonies and contributed to tissue harvesting and preparation; M.T.L. and C.M.R. conducted protein expression analyses; B.L.L. contributed to study design and interpretation, F.R. supplied Fbn1mgR/mgR mice and contributed to study design; D.P.J. was extensively involved in analyzing and interpreting all the data; C.W.W. conducted the ex vivo analyses of skeletal muscles; H.C.D. supervised all aspects of this study including study design, execution and interpretation, and manuscript preparation.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
Evidence for myopathy in fibrillin-1 deficient mouse and human skeletal muscles. (PDF 577 kb)
Supplementary Fig. 2
Morphometric analyses of satellite cells in Fbn1C1039G/+ mice. (PDF 276 kb)
Supplementary Fig. 3
Losartan treatment decreases thrombospondin-1 and periostin expression and improves in vivo muscle function of fibrillin-1 deficient mice. (PDF 97 kb)
Supplementary Fig. 4
Losartan treatment does not alter expression of the dystrophin-glycoprotein complex but improves histologic and in vivo functional properties of skeletal muscles of mdx mice. (PDF 666 kb)
Supplementary Table 1
Physiologic and morphometric analyses of explanted EDL muscles (PDF 26 kb)
Rights and permissions
About this article
Cite this article
Cohn, R., van Erp, C., Habashi, J. et al. Angiotensin II type 1 receptor blockade attenuates TGF-β–induced failure of muscle regeneration in multiple myopathic states. Nat Med 13, 204–210 (2007). https://doi.org/10.1038/nm1536
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm1536
This article is cited by
-
Altered muscle niche contributes to myogenic deficit in the D2-mdx model of severe DMD
Cell Death Discovery (2023)
-
Is the anti-aging effect of ACE2 due to its role in the renin-angiotensin system?—Findings from a comparison of the aging phenotypes of ACE2-deficient, Tsukuba hypertensive, and Mas-deficient mice—
Hypertension Research (2023)
-
Losartan treatment attenuates hindlimb unloading-induced atrophy in the soleus muscle of female rats via canonical TGF-β signaling
The Journal of Physiological Sciences (2022)
-
The jam session between muscle stem cells and the extracellular matrix in the tissue microenvironment
npj Regenerative Medicine (2022)
-
Solid stress impairs lymphocyte infiltration into lymph-node metastases
Nature Biomedical Engineering (2021)