Abstract
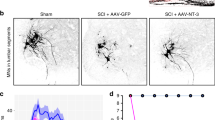
In rodents, after spinal lesion, neutralizing the neurite growth inhibitor Nogo-A promotes axonal sprouting and functional recovery. To evaluate this treatment in primates, 12 monkeys were subjected to cervical lesion. Recovery of manual dexterity and sprouting of corticospinal axons were enhanced in monkeys treated with Nogo-A–specific antibody as compared to monkeys treated with control antibody. NOTE: In the version of this article initially published, the cut corticospinal tract (CST) stumps rostral to the lesion site in Figure 2d and Supplementary Fig. 3a online were meant to be represented schematically, a fact not explained in the figure legend. These representations should therefore have been replaced by full camera lucida reconstructions of these rostral cut CST stumps for the corresponding animals, requiring the consideration of additional sections of the spinal cord located more laterally than those drawn here for the reconstruction of the CST axonal arbors caudal to the lesion (sections for which the contours are represented here). The figure has been corrected in the HTML and the PDF versions of the article.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
Change history
13 September 2006
In the version of this article initially published, the cut corticospinal tract (CST) stumps rostral to the lesion site in Figure 2d and Supplementary Fig. 3a online were meant to be represented schematically, a fact not explained in the figure legend. These representations should therefore have been replaced by full camera lucida reconstructions of these rostral cut CST stumps for the corresponding animals, requiring the consideration of additional sections of the spinal cord located more laterally than those drawn here for the reconstruction of the CST axonal arbors caudal to the lesion (sections for which the contours are represented here). The figure has been corrected in the HTML and the PDF versions of the article.
References
Filbin, M.T. Nat. Rev. Neurosci. 4, 703–713 (2003).
Schwab, M.E. Curr. Opin. Neurobiol. 14, 118–124 (2004).
Bregman, B.S. et al. Nature 378, 498–501 (1995).
Liebscher, T. et al. Ann. Neurol. 58, 706–719 (2005).
Nudo, R.J. & Frost, S.B. in Evolution of Nervous Systems Vol 4 (eds. Krubitzer, L. & Kaas, J.) (Elsevier Science, San Diego, 2006).
Lawrence, D.G. & Hopkins, D.A. Brain 99, 235–254 (1976).
Porter, R. Electroencephalogr. Clin. Neurophysiol. 76, 282–293 (1990).
Lemon, R.N. Exp. Physiol. 78, 263–301 (1993).
Fouad, K., Klusman, I. & Schwab, M.E. Eur. J. Neurosci. 20, 2479–2482 (2004).
Jenny, A.B. & Inukai, J. J. Neurosci. 3, 567–575 (1983).
Galea, M.P. & Darian-Smith, I. J. Comp. Neurol. 381, 282–306 (1997).
Galea, M.P. & Darian-Smith, I. J. Comp. Neurol. 381, 307–319 (1997).
Sasaki, S. et al. J. Neurophysiol. 92, 3142–3147 (2004).
Schmidlin, E., Wannier, T., Bloch, J. & Rouiller, E.M. Brain Res. 1017, 172–183 (2004).
Schmidlin, E. et al. BMC Neurosci. 6, 56 (2005).
Acknowledgements
The authors wish to thank the following for their technical assistance: G. Fischer, V. Moret, C. Roulin and F. Tinguely (histology and behavioral evaluations), J. Corpataux, B. Bapst and B. Morandi (animal house keeping), A. Gaillard (mechanics), B. Aebischer (electronics), L. Monney (informatics). This study was supported by the Swiss National Science Foundation grants 31-61857.00 (to E.M.R.), 31-63633.00 (to M.E.S.) and 4038043918/2 (PNR-38); the Novartis Foundation, the National Centre of Competence in Research on “Neural plasticity and repair,” and the Christopher Reeves Foundation (Springfield, NJ). Antibodies were provided by Novartis Pharma.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Nogo-A–specific antibody treatment enhances sprouting and functional recovery after cervical lesion in adult primates. P Freund, E Schmidlin, T Wannier, J Bloch, A Mir, M E Schwab & E M Rouiller. Novartis Pharma AG provided the antibodies used in this study.
Supplementary information
Supplementary Fig. 1
Organization of the corticospinal projection in the macaque monkey and reconstructions of the cervical lesions performed in the 12 monkeys. (PDF 650 kb)
Supplementary Fig. 2
Assessment of motor capacity of forelimbs and hindlimbs (PDF 145 kb)
Supplementary Fig. 3
Morphological analysis of labeled corticospinal axons; health condition of monkeys. (PDF 1194 kb)
Supplementary Table 1
List of cervical cord lesioned monkeys included in the present study with identification code. (PDF 28 kb)
Rights and permissions
About this article
Cite this article
Freund, P., Schmidlin, E., Wannier, T. et al. Nogo-A–specific antibody treatment enhances sprouting and functional recovery after cervical lesion in adult primates. Nat Med 12, 790–792 (2006). https://doi.org/10.1038/nm1436
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm1436
This article is cited by
-
Epidural electrical stimulation of the cervical dorsal roots restores voluntary upper limb control in paralyzed monkeys
Nature Neuroscience (2022)
-
NogoA-expressing astrocytes limit peripheral macrophage infiltration after ischemic brain injury in primates
Nature Communications (2021)
-
Baseline-adjusted proportional odds models for the quantification of treatment effects in trials with ordinal sum score outcomes
BMC Medical Research Methodology (2020)
-
The adeno-associated virus rh10 vector is an effective gene transfer system for chronic spinal cord injury
Scientific Reports (2019)
-
The translational landscape in spinal cord injury: focus on neuroplasticity and regeneration
Nature Reviews Neurology (2019)