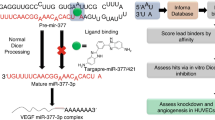
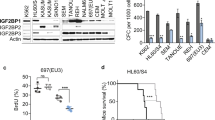
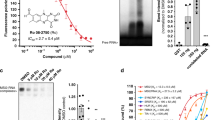
Abstract
The control of cell proliferation is of central importance to the proper development of a multicellular organism, the homeostatic maintenance of tissues, and the ability of certain cell types to respond appropriately to environmental cues. Disruption of normal cell growth control underlies many pathological conditions, including endothelial proliferative disorders in cardiovascular disease as well as the development of malignant tumors. Particularly critical for the control of cell growth is the pathway involving the G1 cyclin–dependent kinases that regulate the Rb family of proteins, which in turn control E2F transcription factor activity1–7. Because E2F is critical for regulation of cell proliferation, we sought to identify and to develop specific inhibitors of E2F function that might also be useful in the control of cellular proliferation. Moreover, because the control of E2F activity appears to be the end result of G1 regulatory cascades, the ability to inhibit E2F may be particularly effective in impeding a wide variety of proliferative events. We have used in vitro selection to isolate several unique RNA species from high complexity RNA libraries that avidly bind to the E2F family of proteins. These RNAs also inhibit the DNA binding capacity of the E2F proteins. We also show that an E2F RNA ligand can block the induction of S phase in quiescent cells stimulated by serum addition. As such, these data demonstrate the critical role for E2F activity in cell proliferation and suggest that such RNA molecules may be effective as therapeutic entities to control cellular proliferation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Sherr, C.J. Mammalian G1 cyclins. Cell 73, 1059–1065 (1993).
Hunter, T. Braking the cycle. Cell 75, 839–841 (1993).
Nevins, J.R. E2F: A link between the Rb tumor suppressor protein and viral onco-proteins. Science 258, 424–429 (1992).
Helin, K. & Harlow, E. The retinoblastoma protein as a transcriptional repressor. Trends Cell Biol. 3, 43–46 (1993).
La Thangue, N.B. DRTF1/E2F: An expanding family of heterodimeric transcription factors implicated in cell-cycle control. Trends Biochem. Sci. 19, 108–114 (1994).
Sherr, C.J. & Roberts, J.M. Inhibitors of mammalian G1 cyclin-dependent kinases. Genes Dev. 9, 1149–1163 (1995).
Weinberg, R.A. The retinoblastoma protein and cell cycle control. Cell 81, 323–330 (1995).
Gold, L., Polisky, B., Uhlenbeck, O. & Yarus, M. Diversity of oligonucleotide functions. Annu. Rev. Biochem. 64, 763–797 (1995).
Szostak, J.W. In vitro genetics. Trends Biochem. Sci. 17, 89–93 (1992).
Tuerk, C. & Gold, L. Systematic evolution of ligands by exponential enrichment: RNA ligands to bacteriophage T4 DNA polymerase. Science 249, 505–510 (1990).
Tuerk, C. & MacDougal-Waugh, S. In vitro evolution of functional nucleic acids: High-affinity RNA ligands of HIV-1 proteins. Gene 137, 33–39 (1993).
Cress, W.D., Johnson, D.G. & Nevins, J.R. A genetic analysis of the E2F1 gene distinguishes regulation by Rb, p107, and adenovirus E4. Mol. Cell. Biol. 13, 6314–6325 (1993).
Zuker, M. On finding all suboptimal foldings of an RNA molecule. Science 244, 48–52 (1989).
Jaeger, J.A., Turner, D.H. & Zuker, M. Improved predictions of secondary structures for RNA. Proc. Natl. Acad. Sci. USA 86, 7706–7710 (1989).
Jaeger, J.A., Turner, D.H. & Zuker, M. Predicting optimal and suboptimal secondary structure for RNA. Methods Enzymol. 183, 281–306 (1989).
Duronio, R.J., O´Farrell, P.H., Xie, J.-E., Brook, A. & Dyson, N. The transcription factor E2F is required for S phase during Drosophila embryogenesis. Genes Dev. 9, 1445–1455 (1995).
Dobrowolski, S.F., Stacey, D.W., Harter, M.L., Stine, J.T. & Hiebert, S.W. An E2F dominant negative mutant blocks E1A induced cell cycle progression. Oncogene 9, 2605–2612 (1994).
Wu, C.-L., Classon, M., Dyson, N. & Harlow, E. Expression of dominant-negative mutant DP-1 blocks cell cycle progression in G1 . Mol. Cell. Biol. 16, 3698–3706 (1996).
Asano, M., Nevins, J.R. & Wharton, R.P. Ectopic E2F expression induces S phase and apoptosis in Drosophila imaginal discs. Genes Dev. 10, 1422–1432 (1996).
DeGregori, J., Leone, G., Ohtani, K., Miron, A. & Nevins, J.R. E2F1 accumulation bypasses a G1 arrest resulting from the inhibition of G1 cyclin-dependent kinase activity. Genes Dev. 9, 2873–2887 (1995).
Johnson, D.G., Schwarz, J.K., Cress, W.D. & Nevins, J.R. Expression of transcription factor E2F1 induces quiescent cells to enter S phase. Nature 365, 349–352 (1993).
Qin, X.-Q., Livingston, D.M., Kaelin, W.G. & Adams, P.D. Deregulated transcription factor E2F-1 expression leads to S-phase entry and p53-mediated apoptosis. Proc. Natl. Acad. Sci. USA 91, 10918–10922 (1994).
Yamasaki, L. et al Tumor induction and tissue atrophy in mice lacking E2F-1. Cell 85, 537–548 (1996).
Field, S.J. et al. E2F-1 functions in mice to promote apoptosis and suppress proliferation. Cell 85, 549–561 (1996).
Doudna, J.A., Cech, T.R. & Sullenger, B. Selection of an RNA molecule that mimics a major autoantigenic epitope of human insulin receptor. Proc. Natl. Acad. Sci. USA 92, 2355–2359 (1995).
Ikeda, M.-A., Jakoi, L. & Nevins, J.R. A unique role for the Rb protein in controlling E2F accumulation during cell growth and differentiation. Proc. Natl. Acad. Sci. USA 93, 3215–3220 (1996).
Li, J.-M., Nichols, M.A., Chandrasekharan, S., Xiong, Y. & Wang, X.-F. Transforming growth factor activates the promoter of cyclin-dependent kinase inhibitor p15INK4B through an Sp1 consensus site. J. Biol. Chem. 270, 26750–26753 (1995).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ishizaki, J., Nevins, J. & Sullenger, B. Inhibition of cell proliferation by an RNA ligand that selectively blocks E2F function. Nat Med 2, 1386–1389 (1996). https://doi.org/10.1038/nm1296-1386
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nm1296-1386
This article is cited by
-
Differential regulation of mammalian and avian ATOH1 by E2F1 and its implication for hair cell regeneration in the inner ear
Scientific Reports (2021)
-
Synthetic feedback control using an RNAi-based gene-regulatory device
Journal of Biological Engineering (2015)
-
E2F-1 as an anticancer drug target
Oncology Reviews (2009)
-
Inhibition of cell proliferation and induction of apoptosis by novel tetravalent peptides inhibiting DNA binding of E2F
Oncogene (2003)
-
The proliferative and apoptotic activities of E2F1 in the mouse retina
Oncogene (2001)