Abstract
Induction of immunity after DNA vaccination is generally considered a slow process. Here we show that DNA delivery to the skin results in a highly transient pulse of antigen expression. Based on this information, we developed a new rapid and potent intradermal DNA vaccination method. By short-interval intradermal DNA delivery, robust T-cell responses, of a magnitude sufficient to reject established subcutaneous tumors, are generated within 12 d. Moreover, this vaccination strategy confers protecting humoral immunity against influenza A infection within 2 weeks after the start of vaccination. The strength and speed of this newly developed strategy will be beneficial in situations in which immunity is required in the shortest possible time.
Similar content being viewed by others
Main
Over the past decade, DNA vaccines have emerged as a promising approach for the induction of immune responses. Generally, current DNA vaccination strategies use a regimen of multiple intramuscular or intradermal administrations, at intervals of 2 weeks or more, and require at least 1 month to achieve immunity1,2,3,4,5,6. The slow development of T-cell responses after DNA vaccination contrasts sharply with immune responses induced by a physiological antigen encounter, such as viral infection, that build up rapidly and often peak within 10 d7. The reason for this slow development of immune responses has remained unclear. It has been postulated that DNA vaccination leads to transfection of few cells and to the expression of relatively small amounts of antigen8,9, therefore requiring a time-consuming prime-boost strategy. But direct evidence for the proposed low antigen expression is scarce and its role in immune induction has not been addressed. To examine whether the slow induction of immune responses could be causally related to the kinetics of antigen expression induced by DNA vaccines, we analyzed in vivo levels of antigen expression after intramuscular and intradermal DNA delivery, and correlated these with the induction of T-cell immunity. Based on these data, we developed a short-interval DNA vaccination method that generates functional T- and B-cell responses within a minimal time frame.
Results
Intradermal DNA vaccination by skin tattooing
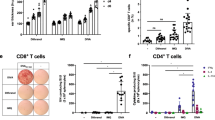
To be able to administer DNA to the skin in a controlled manner, over a large surface and only in the upper nonvascularized layers, we made use of a simple tattoo device10 for intradermal DNA delivery. Histochemical analysis of 'DNA-tattooed' skin showed that transfected cells were distributed over the upper layers of the dermis and the epidermis. (Fig. 1a,b). To test the immunogenicity of this method, we vaccinated two groups of mice with a DNA vaccine encoding the influenza A nucleoprotein epitope (amino acids 366–374; NP366) fused to the carboxy terminus of a tetanus toxin fragment (d1TTFC-NP)11, either by intramuscular injection or by skin tattoo, following the conventional DNA vaccination regimen of three administrations at 2-week intervals. This comparison shows that the intradermal delivery of a DNA vaccine using a tattoo device is an efficient strategy for the induction of T-cell immunity (Fig. 1c).
(a) The abdominal skin of a mouse was tattooed with a β-galactosidase (lacZ)-encoding construct. Six hours after treatment, transfected cells were shown in a skin biopsy, using the X-gal substrate to generate a blue precipitate in cells expressing the transgene (arrows). (b) The abdominal skin of a mouse tattooed with empty vector and processed as in a. (c) NP366-specific CD8+ T-cell responses induced by tattoo (tat., circles, n = 5) or intramuscular (i.m., triangles, n = 5) DNA vaccination. The d1TTFC-NP DNA vaccine was administered three times at 2-week intervals. T-cell responses were measured 7 d after the last DNA administration, by staining peripheral blood lymphocytes with H-2Db/NP366–374 tetramers. Horizontal bars depict averages. (d) NP366-specific T-cell responses induced by tattoo DNA vaccination (circles, n = 5) and i.m. DNA vaccination (triangles, n = 5) with the Luc-NP DNA vaccine. (e) Kinetics of Luc-NP antigen expression after a single intradermal DNA tattoo (circle) and after a single intramuscular DNA injection (triangle).
Imaging of in vivo antigen expression
To monitor antigen expression upon DNA vaccination, we constructed a plasmid encoding the NP366 epitope fused to the carboxy terminus of firefly luciferase (Luc-NP). The Luc-NP vaccine elicits potent NP366-specific T-cell responses (Fig. 1d). After tattoo or intramuscular administration of the Luc-NP vaccine, we used a light-sensitive camera to determine longitudinal in vivo antigen expression. Notably, a single intramuscular injection of DNA resulted in high levels of luciferase activity, peaking after 1 week and remaining detectable up to 1 month after injection. The antigen expression kinetics induced by DNA tattooing were markedly different. First, peak values of antigen expression were at least ten times lower. Second, luciferase activity in the skin peaked after 6 h (data not shown) and disappeared over the next 4 d (Fig. 1e).
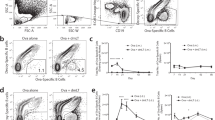
We subsequently determined the capacity of both methods to present the vaccine-encoded NP366 epitope to naive, lymph node–resident T cells. For this purpose, 5 million carboxy-fluorescein diacetate succinimidyl ester (CSFE)-labeled splenocytes from F5 TCR transgenic mice12 were injected into mice at different time points after a single intramuscular DNA injection or DNA intradermal tattoo. Luciferase activity was measured on the day of cell transfer, and 3 d later the animals were killed, lymphoid organs excised and the CFSE signal of the F5 cells assessed by flow cytometry. These experiments showed that although antigen production is markedly greater upon intramuscular delivery, presentation of this antigen to naive T cells is markedly more efficient upon intradermal DNA delivery. Specifically, in spite of the high antigen expression in the muscle after intramuscular injection, the fraction of F5 cells in the draining lymph node that had undergone proliferation was marginal in all of the three tested time windows (days 1–4, days 8–11 and days 22–25; Fig. 2a–c). In marked contrast, the priming of F5 T cells after tattooing was very efficient (Fig. 2a). Independent of the number of F5 T cells that were infused (5 million or 1 million; latter not shown), over 80% of the draining lymph node–resident F5 cells had lost CFSE signal on day 4, despite the low antigen level expressed in skin (Fig. 2d). At later time points, NP366-specific T-cell activation became substantially less (Fig. 2b,c), consistent with the loss of luciferase signal observed by in vivo imaging. Similar results were obtained with spleen-resident F5 cells. No F5 T-cell division could be detected in nondraining lymph nodes (data not shown).
(a–c) Cohorts of mice (n = 7) were vaccinated once with the Luc-NP vaccine, either by intradermal DNA tattooing (filled circles) or by intramuscular DNA injection (filled triangles). Control groups (n = 3) received a plasmid encoding luciferase only (intradermal, open circles; intramuscular, open triangles). CFSE-labeled naive F5 T cells were transferred on day 1 (a), day 8 (b) or day 22 (c) after DNA administration, and were recovered from the draining lymph nodes 3 d after transfer. Proliferation was assessed by analysis of CFSE loss of H-2Db/NP366–374tetramer–positive CD8+ cells. (d) Representative draining lymph node sample from the tattooed cohort on day 4.
Induction of T-cell immunity by short-interval DNA tattooing
Based on the transient antigen expression and presentation induced by DNA tattooing as compared to intramuscular injection, we speculated that in the case of dermal DNA vaccination, shortening of the conventional 2-week interval could result in faster T-cell induction. To test this hypothesis, we reduced the interval between consecutive vaccinations to 3 d, resulting in a day 0, 3 and 6 regimen. Notably, this compact vaccination protocol induced profound T-cell responses of 4–8% of total CD8+ T cells within 12 d after the start of vaccination (Fig. 3a). This regimen of short-interval vaccination does not lead to detectable T-cell responses when the vaccine is administered intramuscularly, consistent with the observation that the duration of antigen expression is not a limiting factor in intramuscular DNA vaccination.
(a) NP366-specific T-cell responses in cohorts of mice (n = 5) upon vaccination with d1TTFC-NP on day 0, 3 and 6 either by tattoo (filled circles) or intramuscular injection (open triangles). Control mice (open circles) were tattooed with a TTFC mock vaccine. NP366-specific T-cell responses were determined at indicated time points by tetramer staining of peripheral blood lymphocytes (n = 5). (b) The E749-specific T-cell response after intradermal application of the sigE7hsp DNA vaccine at day 0, 3 and 6 in cohorts of mice (n = 6), either by DNA tattooing on the left leg (filled circles) or by gene gun on both flanks of the abdomen (filled triangles). Control groups of mice (n = 3) were vaccinated with empty vector (open triangles and open circles). (c) NP366-specific T-cell responses in cohorts of mice (n = 5) upon subcutaneous vaccination with the NP366 peptide in incomplete Freund adjuvant at day 0 (open triangles), subcutaneous injection of NP366 peptide with CpG in PBS at day 0 (open circles) or at day 0, 3 and 6 (open squares), NP366 peptide with CpG in PBS tattoo vaccination at day 0, 3 and 6 (open diamonds) and DNA tattoo vaccination with d1TTFC-NP on day 0, 3 and 6 (filled circles). (d) Antigen expression level after single tattoos of 16, 8, 4 and 1 s, as determined by a light-sensitive camera 1 d after tattooing with the Luc-NP vaccine. (e) NP366-specific T-cell responses (n = 5) induced by 16 s (open circles), 8 s (open triangles) and 4 s (open diamonds) tattoos on day 0, 3 and 6, or a single 16 s tattoo on day 0 (open squares). (f) In mice vaccinated with the Luc-NP DNA vaccine at day 0, 3 and 6, the replacement of Luc-NP with a mock DNA vaccine on day 0 (open squares; C-2x) or day 6 (open diamonds; 2x-C) reduces T-cell responses from levels induced by three consecutive tattoos (open circles; 3x), to levels induced by two consecutive tattoos (open triangles; 2x).
To test the value of this new vaccination regimen for other methods of intradermal DNA delivery, we compared T-cell induction by short-interval gene-gun and short-interval tattoo vaccination, using the previously described Hsp70-HPV-E7 DNA vaccine13. Using the short-interval regimen, both methods generated strong T-cell responses within 12 d against the immunodominant human papillomavirus (HPV) E749–57 cytotoxic T lymphocyte (CTL) epitope (E749) (Fig. 3b). Comparison of T-cell responses induced by short-interval DNA tattooing with other previously established vaccination strategies showed that short-interval DNA tattooing yields markedly higher T-cell responses (tenfold or more) than peptide–incomplete Freund adjuvant–CD40-specific monoclonal antibody14 or peptide-synthetic CpG oligodeoxynucleotide15 vaccines (Fig. 3c). Furthermore, T-cell induction is efficient for both internal epitopes and those fused at the carboxy terminus (Supplementary Fig. 1 online). The resulting T cells are capable of direct effector function, as indicated by antigen-induced interferon (IFN)-γ production (Supplementary Fig. 2 online).
Repetitive application of DNA may conceivably boost T-cell responses by enhancing the absolute amount of antigen expression, or by prolonging the duration of antigen expression. To address this issue, we compared antigen expression levels and T-cell responses induced by three consecutive 4 s tattoos with those induced by one single 16 s tattoo. The cumulative antigen produced by one 16 s application exceeds that of three 4 s applications (Fig. 3d). Whereas three consecutive 4 s applications induce a T-cell response that is readily detectable, a single 16 s application is essentially without effect (Fig. 3e). To further assess whether the observed requirement for repetition reflects a need for continued antigen presence, or is related to the prolongation of nonspecific inflammatory signals, groups of mice were vaccinated at day 0, 3 and 6 with different combinations of a mock vaccine and the Luc-NP vaccine. Replacing either the first or the last Luc-NP vaccine with a mock vaccine lead to a significant drop in the size of the ensuing T-cell response (Student t-test, P = 0.002 and P = 0.003, respectively; Fig. 3f). Together these results show that for the induction of primary T-cell responses by intradermal DNA vaccination, prolonged antigen expression is crucial.
Functional immunity induced by short-interval DNA tattooing
To assess the ability of short-interval tattoo DNA vaccination to induce therapeutic amounts of tumor-specific T cells, we used the transplantable HPV E6/E7-transformed TC-1 tumor cell model16. Three days after subcutaneous injection of 105 TC-1 cells, we vaccinated B6 mice using the short-interval tattoo method with a plasmid encoding E749 attached to the carboxy terminus of green fluorescent protein (GFP-E7; Fig. 4a). Control mice were either given an intramuscular injection in the same regimen with the same plasmid (Fig. 4b), or were tattooed with a control plasmid encoding green fluorescent protein (GFP) only (Fig. 4c). In both control groups, no T-cell responses could be detected after vaccination. In contrast, mice that had been tattooed with GFP-E7 mounted a sizeable T-cell response, up to 15% of the circulating CD8+ T-cell pool. Notably, the onset of E749-specific T-cell responses coincided with rejection of established subcutaneous tumors, whereas in both control groups tumors grew out rapidly (Fig. 4d). Furthermore, median survival of GFP-E7–tattooed animals was 50 d compared to 17 d in control animals (Fig. 4e).
(a–c) Mice were injected subcutaneously with 105 TC-1 cells. Three days later, mice were vaccinated three times at 3-d intervals either by GFP-E7 tattooing (open circles; a), by intramuscular injection (open triangles; b), or by tattooing with a mock vaccine (open diamonds; c). E749-specific CD8+ T-cell responses were determined by major histocompatibility complex tetramer staining of peripheral blood cells at indicated time points. Horizontal bars depict averages. (d) Tumor outgrowth in the cohorts of mice depicted in a–c; symbols are as in a–c. (e) Long-term survival of the mice depicted in a–c; symbols are as in a–c. Values represent mean ± s.d.
To assess the value of short-interval DNA vaccination in providing antibody-mediated protection against acute infections, we evaluated its ability to confer protection against influenza A virus infection. Given that protection against reinfection with homotypic influenza A strains is primarily mediated by antibodies17,18,19,20, we generated a plasmid that encodes the gene for hemagglutinin of influenza A/HK/2/68, a protein that forms a major target for neutralizing antibodies. Using this DNA vaccine, mice were vaccinated by short-interval DNA tattooing, whereas control groups were either tattooed with a GFP-encoding plasmid, or given intramuscular injection with the hemagglutinin-encoding DNA vaccine. Two weeks after vaccination, we intranasally infected mice with a sublethal dose of influenza A, and determined virus-induced morbidity by measuring body weight loss1. In the week after infection, mice that had previously been exposed to influenza A virus showed a minimal weight loss (1% at day 4 after infection). In contrast, mice given tattoo vaccination with the control plasmid or given an intramuscular injection of the hemagglutinin construct showed a sizable (10% and 14%, respectively) drop in body weight. Notably, mice that had received a short-interval hemagglutinin tattoo were largely protected from influenza A–induced morbidity (maximal weight loss of 4%; Student t-test, P = 0.039 versus control vaccine; Fig. 5a). The protection correlated with the induction of neutralizing antibodies (Fig. 5b). Furthermore, intradermal DNA tattooing also conferred long-term (4 months) protection (Supplementary Fig. 3 online). Collectively, these experiments indicate that short-interval intradermal DNA vaccination leads to the rapid and sustained development of both T- and B-cell responses and that such responses can mediate the regression of established tumors and can prevent virus-induced morbidity.
(a) Cohorts of Balb/c mice (n = 6) were vaccinated at day 0, 3 and 6 either by intramuscular injection (filled triangles) or by tattooing (filled circles) with a construct encoding hemagglutinin. A positive control group was infected intranasally with influenza (open diamonds) and a negative control group tattooed using a mock vaccine (open circles). All mice were challenged with a sublethal dose of the influenza A virus at day 14 (indicated by the arrow), and weighed daily. The hemagglutinin-tattooed mice showed a significantly smaller drop in weight than the mock-tattooed mice (Student t-test, P = 0.039). Both in the mock-vaccinated and the intramuscularly vaccinated group, one mouse died from influenza-induced pneumonia. These animals were not included in the analysis. Values represent the weight relative to the weight at time of challenge. Error bars, s.e.m. (b) Influenza A/HK/2/68-specific antibodies in the sera of the mice depicted in a, at various time points after the start of vaccination and the influenza A challenge (symbols as in a). The presence of antibodies was assessed in an HAI assay. Values represent mean ± s.d.
Discussion
In this study we established a short-interval regimen that induces T- and B-cell immunity within 12 d. Analysis of antigen expression levels showed that after intradermal tattoo, 1/10 to 1/100 of the amount of antigen is produced than after intramuscular injection, and that compared to intramuscular administration, tattoo-induced antigen production occurs over a limited timespan. In spite of this, the presentation of the vaccine-encoded epitope is markedly better upon intradermal tattoo vaccination. The efficiency with which dermally expressed antigens are presented to T cells probably results from the high numbers of antigen-presenting cells (APCs) present in this tissue. Also, the infliction of thousands of perforations could conceivably serve as a potent adjuvant.
Contrary to murine skin dendritic cells (DCs), human skin DCs do not express Toll-like receptor 9 (TLR9), and this may partly explain why DNA vaccines have performed poorly in human trials as compared to mouse model systems21,22. Encouragingly, a comparison of the immunogenicity of tattoo DNA vaccination in wild-type and Tlr9−/− mice showed that the absence of TLR9 does not measurably affect the immunogenicity of this method (Supplementary Fig. 4 online).
The short-interval repetitive schedule developed here leads to rapid induction of T-cell responses upon either intradermal tattoo or gene-gun vaccination, and may well work for all intradermal vaccination techniques. Because of its simplicity and because the required equipment is orders of magnitude less expensive, DNA tattooing may become the preferred method, in particular in countries with a less developed healthcare system.
In the past years, outbreaks of Ebola virus, severe acute respiratory syndrome and influenza A have been causes for concern. To contain such outbreaks conventional DNA vaccination strategies appear too slow at inducing immunity23,24. The vaccination regimen described here retains the rapid production and the safety of DNA vaccines, but gains the speed of immune induction that is the characteristic of physiological antigen encounters.
Methods
Animals.
We obtained C57BL/6 mice and Balb/c mice from the experimental animal department of The Netherlands Cancer Institute. The Tlr9−/− mice were a gift of H. Wagner ((with permission of S. Akira) Institute for Microbiology, Immunology and Hygiene, University of Munich, Germany). We purchased from the US National Cancer Institute the C57BL/6 mice used for the experiments involving a gene gun and kept them in the oncology animal facility of the Johns Hopkins Hospital. We performed all animal procedures according to approved protocols and in accordance with recommendations for the proper use and care of laboratory animals. All animal experiments were approved by the Dutch Animal Research Committee.
DNA vaccines.
We generated DNA vaccines by the introduction of fusion genes in pcDNA3.1. The GFP-E7 fusion gene has been described previously25, all other constructs were generated following a similar design. We included the four naturally flanking amino acid residues of NP366–374 (amino acids GVQI) as a linker at the amino terminus of each epitope in all constructs. We constructed the d1TTFC-NP DNA vaccine with optimized codon usage, in a template-free PCR using overlapping oligonucleotides of 100 bp, spanning the entire first domain of the TTFC gene and the preceding p2 epitope (amino acids 831–1,315; ref. 26). We generated the Luc-NP vaccine, encoding influenza A NP366–374 epitope fused to the carboxy terminus of firefly luciferase, by genetic linkage of the NP366–374 fragment to the carboxy terminus of the gene encoding full-length firefly luciferase. The gene encoding influenza A/HK/2/68 hemagglutinin was obtained by RT-PCR of RNA isolated from virus particles, and inserted in pcDNA3.1. to generate the hemagglutinin DNA vaccine. We purchased the construct used for histochemical detection of transfected cells (pVAX:LacZ) from Invitrogen. Replacement of the lacZ gene with chicken ovalbumin generated the construct encoding whole ovalbumin. The generation of the sigE7hsp DNA vaccine was described previously13. Sequences were confirmed by sequence analysis. All DNA batches were purified using EndoFree Plasmid kit (Qiagen).
DNA vaccination.
For intramuscular DNA vaccination, we shaved the hind leg of the mouse and injected it with 100 μg of DNA in 50 μl HBSS (Life Technologies). For intradermal DNA vaccination, we shaved the left hind leg of the mouse, applied a droplet of 20 μg DNA in 10 μl HBSS to the skin and used a sterile disposable 11-needle bar (Radical Clean Magnum11, Eurl Toupera) mounted on a rotary tattoo device (Cold skin, B&A Trading) to apply the vaccine. Needle depth was adjusted to 0.5 mm, and the needle bar oscillated at 100 Hz. DNA vaccines were applied to the skin by a 16 s tattoo. Gene-gun particle-mediated DNA vaccination was performed using a helium-driven gene gun (Bio-Rad) according to the manufacturer's protocol. At various time points after immunization, we drew approximately 20 μl of peripheral blood for analysis of T-cell responses.
Detection of NP- and HPV-specific T cells in peripheral blood.
Peripheral blood lymphocytes were stained with phycoerythrin-conjugated antibody to CD8β (BD Pharmingen) plus allophycocyanin-conjugated H-2Db/NP366–374-tetramers or allophycocyanin-conjugated H-2Db/E749–47 tetramers, at 20 °C for 15 min in FACS buffer (1× PBS, 0.5% BSA and 0.02% sodium azide). We washed cells three times in PBA and analyzed them using flow cytometry. Live cells were selected based on 7-AAD exclusion. We performed IFN-γ assays using the BD Cytofix/Cytoperm kit (BD Biosciences) according to the manufacturer's protocol. Peripheral blood lymphocytes were stimulated for 4 h at a 100 nM peptide concentration.
Intravital imaging.
We anesthetized mice with isofluorane (Abbott Laboratories). We intraperitoneally injected an aqueous solution of the substrate luciferin (150 mg/kg, Xenogen) and 18 min later the luminescence produced by active luciferase was acquired during 30 s in an IVIS system100 CCD camera (Xenogen). Signal intensity was quantified as the sum of all detected light within the region of interest, after subtraction of background luminescence.
Influenza A infection and detection of influenza A–specific antibodies.
Purified influenza A/HK/2/68 virus was provided by G. Rimmelzwaan (Department of Virology, Erasmus University). We infected mice intranasally with 20 hemagglutinating units (HAU) of virus in PBS, under general anesthesia. Influenza A/HK/2/68-specific antibodies were detected in a hemagglutination inhibition (HAI) assay. Serum samples (50 μl) were serially 1:2 diluted with PBS (1% BSA) in round-bottomed polystyrene microtiter plates, in duplicate, and 50 μl of A/HK/2/68 influenza virus corresponding to 4 HAU was added to each well. After 45 min incubation at room temperature, we added 50 μl of 0.5% chicken red blood cells to each well. After 1 h, we determined the maximal serum dilution that fully inhibited the hemagglutination caused by the virus in the duplicate wells. The HAI titer was expressed as the reciprocal of this serum dilution.
Note: Supplementary information is available on the Nature Medicine website.
References
Ulmer, J.B. et al. Heterologous protection against influenza by injection of DNA encoding a viral protein. Science 259, 1745–1749 (1993).
Kirman, J.R. et al. Enhanced immunogenicity to Mycobacterium tuberculosis by vaccination with an alphavirus plasmid replicon expressing antigen 85A. Infect. Immun. 71, 575–579 (2003).
Yang, Z.Y. et al. A DNA vaccine induces SARS coronavirus neutralization and protective immunity in mice. Nature 428, 561–564 (2004).
Gurunathan, S., Klinman, D.M. & Seder, R.A. DNA vaccines: immunology, application, and optimization. Annu. Rev. Immunol. 18, 927–974 (2000).
Cappello, P. et al. LAG-3 enables DNA vaccination to persistently prevent mammary carcinogenesis in HER-2/neu transgenic BALB/c mice. Cancer Res. 63, 2518–2525 (2003).
De Marco, F. et al. DNA vaccines against HPV-16 E7-expressing tumor cells. Anticancer Res. 23, 1449–1454 (2003).
Murali-Krishna, K. et al. Counting antigen-specific CD8 T cells: a reevaluation of bystander activation during viral infection. Immunity 8, 177–187 (1998).
Kalat, M. et al. In vivo plasmid electroporation induces tumor antigen-specific CD8+ T-cell responses and delays tumor growth in a syngeneic mouse melanoma model. Cancer Res. 62, 5489–5494 (2002).
Smerdou, C. & Liljestrom, P. Non-viral amplification systems for gene transfer: vectors based on alphaviruses. Curr. Opin. Mol. Ther. 1, 244–251 (1999).
Ciernik, I.F., Krayenbuhl, B.H. & Carbone, D.P. Puncture-mediated gene transfer to the skin. Hum. Gene Ther. 7, 893–899 (1996).
Rice, J., Buchan, S. & Stevenson, F.K. Critical components of a DNA fusion vaccine able to induce protective cytotoxic T cells against a single epitope of a tumor antigen. J. Immunol. 169, 3908–3913 (2002).
Mamalaki, C. et al. Positive and negative selection in transgenic mice expressing a T-cell receptor specific for influenza nucleoprotein and endogenous superantigen. Dev. Immunol. 3, 159–174 (1993).
Chen, C. et al. Enhancement of DNA vaccine potency by linkage of antigen gene to an HSP70 gene. Cancer Res. 60, 1035–1042 (2000).
Diehl, L. et al. CD40 activation in vivo overcomes peptide-induced peripheral cytotoxic T-lymphocyte tolerance and augments anti-tumor vaccine efficacy. Nat. Med. 5, 774–779 (1999).
Lipford, G.B. et al. CpG-containing synthetic oligonucleotides promote B and cytotoxic T cell responses to protein antigen: a new class of vaccine adjuvants. Eur. J. Immunol. 27, 2340–2344 (1997).
Lin, K.Y. et al. Treatment of established tumors with a novel vaccine that enhances major histocompatibility class II presentation of tumor antigen. Cancer Res. 56, 21–26 (1996).
Virelizier, J.L. Host defenses against influenza virus: the role of anti-hemagglutinin antibody. J. Immunol. 115, 434–439 (1975).
Eichelberger, M., Allan, W., Zijlstra, M., Jaenisch, R. & Doherty, P.C. Clearance of influenza virus respiratory infection in mice lacking class I major histocompatibility complex-restricted CD8+ T cells. J. Exp. Med. 174, 875–880 (1991).
Epstein, S.L. et al. Beta 2-microglobulin-deficient mice can be protected against influenza A infection by vaccination with vaccinia-influenza recombinants expressing hemagglutinin and neuraminidase. J. Immunol. 150, 5484–5493 (1993).
Scherle, P.A., Palladino, G. & Gerhard, W. Mice can recover from pulmonary influenza virus infection in the absence of class I-restricted cytotoxic T cells. J. Immunol. 148, 212–217 (1992).
Wang, R. et al. Induction of antigen-specific cytotoxic T lymphocytes in humans by a malaria DNA vaccine. Science 282, 476–480 (1998).
Wang, R. et al. Induction of CD4(+) T cell-dependent CD8(+) type 1 responses in humans by a malaria DNA vaccine. Proc. Natl Acad. Sci. USA 98, 10817–10822 (2001).
Sullivan, N.J., Sanchez, A., Rollin, P.E., Yang, Z.Y. & Nabel, G.J. Development of a preventive vaccine for Ebola virus infection in primates. Nature 408, 605–609 (2000).
Sullivan, N.J. et al. Accelerated vaccination for Ebola virus haemorrhagic fever in non-human primates. Nature 424, 681–684 (2003).
Wolkers, M.C., Toebes, M., Okabe, M., Haanen, J.B.A.G. & Schumacher, T.N.M. Optimizing the efficacy of epitope-directed DNA vaccination. J. Immunol. 168, 4998–5004 (2002).
Panina-Bordignon, P. et al. Universally immunogenic T cell epitopes: promiscuous binding to human MHC class II and promiscuous recognition by T cells. Eur. J. Immunol. 19, 2237–2242 (1989).
Acknowledgements
The authors would like to acknowledge G. Schory for supplying tattoo accessories. This research was funded by the Dutch Cancer Society, project grant 2001-2417.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
DNA tattoo vaccination efficiently induces T cells against internal epitopes. (PDF 19 kb)
Supplementary Fig. 2
T cells induced by DNA tattoo vaccination are capable of direct effector function. (PDF 61 kb)
Supplementary Fig. 3
Tattoo DNA vaccination induces long-term immunity against influenza A. (PDF 24 kb)
Supplementary Fig. 4
Absence of TLR9 does not measurably affect the immunogenicity of DNA tattoo vaccination. (PDF 19 kb)
Rights and permissions
About this article
Cite this article
Bins, A., Jorritsma, A., Wolkers, M. et al. A rapid and potent DNA vaccination strategy defined by in vivo monitoring of antigen expression. Nat Med 11, 899–904 (2005). https://doi.org/10.1038/nm1264
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm1264
This article is cited by
-
A novel intradermal tattoo-based injection device enhances the immunogenicity of plasmid DNA vaccines
npj Vaccines (2022)
-
Optimized polyepitope neoantigen DNA vaccines elicit neoantigen-specific immune responses in preclinical models and in clinical translation
Genome Medicine (2021)
-
Labeling and tracking of immune cells in ex vivo human skin
Nature Protocols (2021)
-
Investigating BB0405 as a novel Borrelia afzelii vaccination candidate in Lyme borreliosis
Scientific Reports (2021)
-
A mouse model that is immunologically tolerant to reporter and modifier proteins
Communications Biology (2020)