Abstract
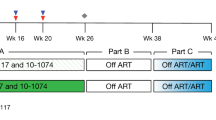
To determine the protective potential of the humoral immune response against HIV-1 in vivo we evaluated the potency of three neutralizing antibodies (2G12, 2F5 and 4E10) in suppressing viral rebound in six acutely and eight chronically HIV-1–infected individuals undergoing interruption of antiretroviral treatment (ART). Only two of eight chronically infected individuals showed evidence of a delay in viral rebound during the passive immunization. Rebound in antibody-treated acutely infected individuals upon cessation of ART was substantially later than in a control group of 12 individuals with acute infection. Escape mutant analysis showed that the activity of 2G12 was crucial for the in vivo effect of the neutralizing antibody cocktail. By providing further direct evidence of the potency, breadth and titers of neutralizing antibodies that are required for in vivo activity, these data underline both the potential and the limits of humoral immunity in controlling HIV-1 infection.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Letvin, N.L. & Walker, B.D. Immunopathogenesis and immunotherapy in AIDS virus infections. Nat. Med. 9, 861–866 (2003).
Pantaleo, G. & Koup, R.A. Correlates of immune protection in HIV-1 infection: what we know, what we don't know, what we should know. Nat. Med. 10, 806–810 (2004).
Haigwood, N.L. & Stamatatos, L. Role of neutralizing antibodies in HIV infection. AIDS 17 Suppl 4, S67–S71 (2003).
Parren, P.W., Moore, J.P., Burton, D.R. & Sattentau, Q.J. The neutralizing antibody response to HIV-1: viral evasion and escape from humoral immunity. AIDS 13, S137–S162 (1999).
Baba, T.W. et al. Human neutralizing monoclonal antibodies of the IgG1 subtype protect against mucosal simian-human immunodeficiency virus infection. Nat. Med. 6, 200–206 (2000).
Mascola, J.R. et al. Protection of macaques against vaginal transmission of a pathogenic HIV- 1/SIV chimeric virus by passive infusion of neutralizing antibodies. Nat. Med. 6, 207–210 (2000).
Mascola, J.R. et al. Protection of Macaques against pathogenic simian/human immunodeficiency virus 89.6PD by passive transfer of neutralizing antibodies. J. Virol. 73, 4009–4018 (1999).
Veazey, R.S. et al. Prevention of virus transmission to macaque monkeys by a vaginally applied monoclonal antibody to HIV-1 gp120. Nat. Med. 9, 343–346 (2003).
Haigwood, N.L. et al. Passive immunotherapy in simian immunodeficiency virus-infected macaques accelerates the development of neutralizing antibodies. J. Virol. 78, 5983–5995 (2004).
Parren, P.W. et al. Antibody protects macaques against vaginal challenge with a pathogenic R5 simian/human immunodeficiency virus at serum levels giving complete neutralization in vitro . J. Virol. 75, 8340–8347 (2001).
Poignard, P. et al. Neutralizing antibodies have limited effects on the control of established HIV-1 infection in vivo . Immunity 10, 431–438 (1999).
Gauduin, M.C. et al. Passive immunization with a human monoclonal antibody protects hu-PBL- SCID mice against challenge by primary isolates of HIV-1. Nat. Med. 3, 1389–1393 (1997).
Shibata, R. et al. Neutralizing antibody directed against the HIV-1 envelope glycoprotein can completely block HIV-1/SIV chimeric virus infections of macaque monkeys. Nat. Med. 5, 204–210 (1999).
Andrus, L. et al. Passive immunization with a human immunodeficiency virus type 1- neutralizing monoclonal antibody in Hu-PBL-SCID mice: isolation of a neutralization escape variant. J. Infect. Dis. 177, 889–897 (1998).
Montefiori, D.C., Hill, T.S., Vo, H.T., Walker, B.D. & Rosenberg, E.S. Neutralizing antibodies associated with viremia control in a subset of individuals after treatment of acute human immunodeficiency virus type 1 infection. J. Virol. 75, 10200–10207 (2001).
Cao, Y., Qin, L., Zhang, L., Safrit, J. & Ho, D.D. Virologic and immunologic characterization of long-term survivors of human immunodeficiency virus type 1 infection. N. Engl. J. Med. 332, 201–208 (1995).
Carotenuto, P., Looij, D., Keldermans, L., de Wolf, F. & Goudsmit, J. Neutralizing antibodies are positively associated with CD4+ T-cell counts and T-cell function in long-term AIDS-free infection. AIDS 12, 1591–1600 (1998).
Montefiori, D.C. et al. Neutralizing and infection-enhancing antibody responses to human immunodeficiency virus type 1 in long-term nonprogressors. J. Infect. Dis. 173, 60–67 (1996).
Pilgrim, A.K. et al. Neutralizing antibody responses to human immunodeficiency virus type 1 in primary infection and long-term-nonprogressive infection. J. Infect. Dis. 176, 924–932 (1997).
Armbruster, C. et al. A phase I trial with two human monoclonal antibodies (hMAb 2F5, 2G12) against HIV-1. AIDS 16, 227–233 (2002).
Stiegler, G. et al. Antiviral activity of the neutralizing antibodies 2F5 and 2G12 in asymptomatic HIV-1-infected humans: a phase I evaluation. AIDS 16, 2019–2025 (2002).
Armbruster, C. et al. Passive immunization with the anti-HIV-1 human monoclonal antibody (hMAb) 4E10 and the hMAb combination 4E10/2F5/2G12. J. Antimicrob. Chemother. 54, 915–920 (2004).
Vittecoq, D. et al. Passive immunotherapy in AIDS: a double-blind randomized study based on transfusions of plasma rich in anti-human immunodeficiency virus 1 antibodies vs. transfusions of seronegative plasma. Proc. Natl. Acad. Sci. USA 92, 1195–1199 (1995).
Jacobson, J.M. et al. Passive immunotherapy in the treatment of advanced human immunodeficiency virus infection. J. Infect. Dis. 168, 298–305 (1993).
Gunthard, H.F. et al. A phase I/IIA clinical study with a chimeric mouse-human monoclonal antibody to the V3 loop of human immunodeficiency virus type 1 gp120. J. Infect. Dis. 170, 1384–1393 (1994).
Cavacini, L.A. et al. Phase I study of a human monoclonal antibody directed against the CD4-binding site of HIV type 1 glycoprotein 120. AIDS Res. Hum. Retroviruses 14, 545–550 (1998).
Arendrup, M. et al. Autologous HIV-1 neutralizing antibodies: emergence of neutralization- resistant escape virus and subsequent development of escape virus neutralizing antibodies. J. Acquir. Immune Defic. Syndr. 5, 303–307 (1992).
McKeating, J.A. et al. Characterization of HIV-1 neutralization escape mutants. AIDS 3, 777–784 (1989).
Richman, D.D., Wrin, T., Little, S.J. & Petropoulos, C.J. Rapid evolution of the neutralizing antibody response to HIV type 1 infection. Proc. Natl. Acad. Sci. USA 100, 4144–4149 (2003).
Watkins, B.A. et al. Immune escape by human immunodeficiency virus type 1 from neutralizing antibodies: evidence for multiple pathways. J. Virol. 67, 7493–7500 (1993).
Wei, X. et al. Antibody neutralization and escape by HIV-1. Nature 422, 307–312 (2003).
Calarota, S.A. & Weiner, D.B. Present status of human HIV vaccine development. Aids 17 Suppl 4, S73–S84 (2003).
Trkola, A. et al. Human monoclonal antibody 2G12 defines a distinctive neutralization epitope on the gp120 glycoprotein of human immunodeficiency virus type 1. J. Virol. 70, 1100–1108 (1996).
Zwick, M.B. et al. Broadly neutralizing antibodies targeted to the membrane-proximal external region of human immunodeficiency virus type 1 glycoprotein gp41. J. Virol. 75, 10892–10905 (2001).
Sanders, R.W. et al. The mannose-dependent epitope for neutralizing antibody 2G12 on human immunodeficiency virus type 1 glycoprotein gp120. J. Virol. 76, 7293–7305 (2002).
Trkola, A. et al. Cross-clade neutralization of primary isolates of human immunodeficiency virus type 1 by human monoclonal antibodies and tetrameric CD4-IgG. J. Virol. 69, 6609–6617 (1995).
Muster, T. et al. A conserved neutralizing epitope on gp41 of human immunodeficiency virus type 1. J. Virol. 67, 6642–6647 (1993).
Purtscher, M. et al. A broadly neutralizing human monoclonal antibody against gp41 of human immunodeficiency virus type 1. AIDS Res. Hum. Retroviruses 10, 1651–1658 (1994).
Stiegler, G. et al. A potent cross-clade neutralizing human monoclonal antibody against a novel epitope on gp41 of human immunodeficiency virus type 1. AIDS Res. Hum. Retroviruses 17, 1757–1765 (2001).
Binley, J.M. et al. Comprehensive cross-clade neutralization analysis of a panel of anti-human immunodeficiency virus type 1 monoclonal antibodies. J. Virol. 78, 13232–13252 (2004).
Rusert, P. et al. Virus isolates during acute and chronic HIV infection show distinct pattern of sensitivity to entry inhibitors. J. Virol. (in the press).
Fagard, C. et al. A prospective trial of structured treatment interruptions in human immunodeficiency virus infection. Arch. Intern. Med. 163, 1220–1226 (2003).
Fagard, C. et al. A controlled trial of granulocyte macrophage-colony stimulating factor during interruption of HAART. AIDS 17, 1487–1492 (2003).
Smith, D.E., Walker, B.D., Cooper, D.A., Rosenberg, E.S. & Kaldor, J.M. Is antiretroviral treatment of primary HIV infection clinically justified on the basis of current evidence? AIDS 18, 709–718 (2004).
Jolly, C., Kashefi, K., Hollinshead, M. & Sattentau, Q.J. HIV-1 cell to cell transfer across an Env-induced, actin-dependent synapse. J. Exp. Med. 199, 283–293 (2004).
Pantaleo, G. et al. Effect of anti-V3 antibodies on cell-free and cell-to-cell human immunodeficiency virus transmission. Eur. J. Immunol. 25, 226–231 (1995).
Gibaldi, M. & Perrier, D. Pharmacokinetics p. 329 (Marcel Dekker, New York, 1975).
Schockmel, G.A., Yerly, S. & Perrin, L. Detection of low HIV-1 RNA levels in plasma. J. Acquir. Immune Defic. Syndr. Hum. Retrovirol. 14, 179–183 (1997).
Rusert, P. et al. Quantification of infectious HIV-1 plasma viral load using a boosted in vitro infection protocol. Virology 326, 113–129 (2004).
Acknowledgements
Support was provided by the Swiss National Science Foundation (grant PP00B-102647 to A.T. and grant 3100A0-103748 to H.G. and A.T.), research grants from the Union Bank of Switzerland, the Gebert-Rüf foundation (P-041/02) and the FAIR Foundation to A.T. and H.G., and by a research grant of the Kanton Zürich. We thank our patients for their commitment, M. Winniger, U. Berberat, R. Hafner, B. Hasse, U. Karrer, R. Oberholzer, C. Schneider and C. Grube for patient care, J. Böni and F. Burgener for technical help, M. Schlumpf for infrastructural help, E. Gremlich for study monitoring, I. Nievergelt for administrative assistance and H. Hengartner and R. Zinkernagel for their support in initiating this study.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
Gabriela Stiegler, Brigitta Vcelar and Hermann Katinger are from Polymun Scientific, a company whose aim is to advance the development of the human monoclonal antibodies from clinical trials to market approval for prevention and treatment of HIV infection.
Supplementary information
Supplementary Fig. 1
Viremia rebounds during previous STIs in chronic patients participating in passive immunization trial. (PDF 81 kb)
Supplementary Fig. 2
Characteristics of patients with acute HIV-1 infection. (PDF 97 kb)
Supplementary Table 1
Patient characteristics (PDF 24 kb)
Supplementary Table 2
Pharmacokinetic parameters (PDF 18 kb)
Supplementary Table 3
History of treatment interruption of chronically infected individuals participating in passive immunization trial (PDF 25 kb)
Supplementary Table 4
Sequence analysis of 2F5 and 4E10 epitope (PDF 14 kb)
Supplementary Table 5
Antibody sensitivity of sequential isolates derived during STIs (PDF 16 kb)
Rights and permissions
About this article
Cite this article
Trkola, A., Kuster, H., Rusert, P. et al. Delay of HIV-1 rebound after cessation of antiretroviral therapy through passive transfer of human neutralizing antibodies. Nat Med 11, 615–622 (2005). https://doi.org/10.1038/nm1244
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm1244
This article is cited by
-
Mutational scanning of spike RBD protein for enhanced ACE2 affinity emerging Southeast Asia in the late transmission phase
Scientific Reports (2022)
-
Broadly neutralizing anti-HIV-1 monoclonal antibodies in the clinic
Nature Medicine (2019)
-
Induction of neutralizing antibodies against tier 2 human immunodeficiency virus 1 in rhesus macaques infected with tier 1B simian/human immunodeficiency virus
Archives of Virology (2019)
-
Adeno-associated virus gene delivery of broadly neutralizing antibodies as prevention and therapy against HIV-1
Retrovirology (2018)
-
Antibody-mediated prevention and treatment of HIV-1 infection
Retrovirology (2018)