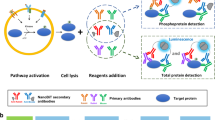
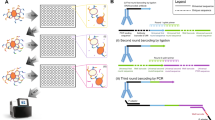
Abstract
We have developed a multiplexed reverse phase protein (RPP) microarray platform for simultaneous monitoring of site-specific phosphorylation of numerous signaling proteins using nanogram amounts of lysates derived from stimulated living cells. We first show the application of RPP microarrays to the study of signaling kinetics and pathway delineation in Jurkat T lymphocytes. RPP microarrays were used to profile the phosphorylation state of 62 signaling components in Jurkat T cells stimulated through their membrane CD3 and CD28 receptors, identifying a previously unrecognized link between CD3 crosslinking and dephosphorylation of Raf-1 at Ser259. Finally, the potential of this technology to analyze rare primary cell populations is shown in a study of differential STAT protein phosphorylation in interleukin (IL)-2-stimulated CD4+CD25+ regulatory T cells. RPP microarrays, prepared using simple procedures and standard microarray equipment, represent a powerful new tool for the study of signal transduction in both health and disease.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Ficarro, S.B. et al. Phosphoproteome analysis by mass spectrometry and its application to Saccharomyces cerevisiae. Nat. Biotechnol. 20, 301–305 (2002).
Robinson, W.H. et al. Autoantigen microarrays for multiplex characterization of autoantibody responses. Nat. Med. 8, 295–301 (2002).
Robinson, W.H. et al. Protein microarrays guide tolerizing DNA vaccine treatment of autoimmune encephalomyelitis. Nat. Biotechnol. 21, 1033–1039 (2003).
Nielsen, U.B., Cardone, M.H., Sinskey, A.J., MacBeath, G. & Sorger, P.K. Profiling receptor tyrosine kinase activation by using Ab microarrays. Proc. Natl. Acad. Sci. USA 100, 9330–9335 (2003).
Haab, B.B., Dunham, M.J. & Brown, P.O. Protein microarrays for highly parallel detection and quantitation of specific proteins and antibodies in complex solutions. Genome Biol. 2, RESEARCH0004 (2001).
MacBeath, G. Protein microarrays and proteomics. Nat. Genet. 32 Suppl., 526–532 (2002).
Paweletz, C.P. et al. Reverse phase protein microarrays which capture disease progression show activation of pro-survival pathways at the cancer invasion front. Oncogene 20, 1981–1989 (2001).
Espina, V. et al. Protein microarrays: molecular profiling technologies for clinical specimens. Proteomics 3, 2091–2100 (2003).
Liotta, L.A. et al. Protein microarrays: meeting analytical challenges for clinical applications. Cancer Cell 3, 317–325 (2003).
Irish, J.M. et al. Single cell profiling of potentiated phospho-protein networks in cancer cells. Cell 118, 217–228 (2004).
Bobrow, M.N., Litt, G.J., Shaughnessy, K.J., Mayer, P.C. & Conlon, J. The use of catalyzed reporter deposition as a means of signal amplification in a variety of formats. J. Immunol. Methods 150, 145–149 (1992).
Wang, Z., Gluck, S., Zhang, L. & Moran, M.F. Requirement for phospholipase C-gamma1 enzymatic activity in growth factor-induced mitogenesis. Mol. Cell. Biol. 18, 590–597 (1998).
Kane, L.P., Andres, P.G., Howland, K.C., Abbas, A.K. & Weiss, A. Akt provides the CD28 costimulatory signal for up-regulation of IL-2 and IFN-gamma but not TH2 cytokines. Nat. Immunol. 2, 37–44 (2001).
Zhang, J. et al. p38 mitogen-activated protein kinase mediates signal integration of TCR/CD28 costimulation in primary murine T cells. J. Immunol. 162, 3819–3829 (1999).
Cantrell, D. T cell antigen receptor signal transduction pathways. Annu. Rev. Immunol. 14, 259–274 (1996).
Matthews, S.A., Rozengurt, E. & Cantrell, D. Protein kinase D. A selective target for antigen receptors and a downstream target for protein kinase C in lymphocytes. J. Exp. Med. 191, 2075–2082 (2000).
Yuan, J., Bae, D., Cantrell, D., Nel, A.E. & Rozengurt, E. Protein kinase D is a downstream target of protein kinase C theta. Biochem. Biophys. Res. Commun. 291, 444–452 (2002).
Kleijn, M. & Proud, C.G. The regulation of protein synthesis and translation factors by CD3 and CD28 in human primary T lymphocytes. BMC Biochem. 3, 11 (2002).
Michaud, N.R., Fabian, J.R., Mathes, K.D. & Morrison, D.K. 14–3-3 is not essential for Raf-1 function: identification of Raf-1 proteins that are biologically activated in a 14–3-3- and Ras-independent manner. Mol. Cell. Biol. 15, 3390–3397 (1995).
Zimmermann, S. & Moelling, K. Phosphorylation and regulation of Raf by Akt (protein kinase B). Science 286, 1741–1744 (1999).
Jaumot, M. & Hancock, J.F. Protein phosphatases 1 and 2A promote Raf-1 activation by regulating 14–3-3 interactions. Oncogene 20, 3949–3958 (2001).
Sakaguchi, S. Naturally arising CD4+ regulatory T cells for immunologic self-tolerance and negative control of immune responses. Annu. Rev. Immunol. 22, 531–562 (2004).
Almeida, A.R., Legrand, N., Papiernik, M. & Freitas, A.A. Homeostasis of peripheral CD4+ T cells: IL-2R alpha and IL-2 shape a population of regulatory cells that controls CD4+ T cell numbers. J. Immunol. 169, 4850–4860 (2002).
Furtado, G.C., Curotto de Lafaille, M.A., Kutchukhidze, N. & Lafaille, J.J. Interleukin 2 signaling is required for CD4+ regulatory T cell function. J. Exp. Med. 196, 851–857 (2002).
Antov, A., Yang, L., Vig, M., Baltimore, D. & Van Parijs, L. Essential role for STAT5 signaling in CD25+CD4+ regulatory T cell homeostasis and the maintenance of self-tolerance. J. Immunol. 171, 3435–3441 (2003).
Snow, J.W. et al. Loss of tolerance and autoimmunity affecting multiple organs in STAT5A/5B-deficient mice. J. Immunol. 171, 5042–5050 (2003).
Acknowledgements
We thank W.H. Robinson and M. Kattah for insightful discussions; and A. Zhang for technical assistance. S.M.C. is supported by the Stanford Medical Scientist Training Program. P.J.U. is supported by grants from the Dana Foundation, Northern California Chapter of the Arthritis Foundation, the Stanford Program in Molecular and Genetic Medicine (PMGM), NIH Grants DK61934, AI50854, AI50865, and AR49328, and NHLBI Proteomics Contract N01-HV-28183.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
Representative image of a RPP microarray in an 8-pad subarray format. (PDF 132 kb)
Supplementary Fig. 2
Kinetics of Akt (Ser473), p38 MAPK and SAPK/JNK phosphorylation. (PDF 153 kb)
Supplementary Fig. 3
Differential STAT protein phosphorylation in T regulatory cells in response to IL-2. (PDF 66 kb)
Supplementary Fig. 4
Proof-of-concept experiment demonstrating the use of RPP microarrays for screening compounds with kinase inhibition activity. (PDF 54 kb)
Supplementary Table 1
List of 62 phospho-specific antibodies used in Figure 4 (PDF 8 kb)
Rights and permissions
About this article
Cite this article
Chan, S., Ermann, J., Su, L. et al. Protein microarrays for multiplex analysis of signal transduction pathways. Nat Med 10, 1390–1396 (2004). https://doi.org/10.1038/nm1139
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm1139
This article is cited by
-
Chi3l3 induces oligodendrogenesis in an experimental model of autoimmune neuroinflammation
Nature Communications (2019)
-
Microfluidic Fabrication of Encoded Hydrogel Microparticles for Application in Multiplex Immunoassay
BioChip Journal (2019)
-
Prediction of individual response to anticancer therapy: historical and future perspectives
Cellular and Molecular Life Sciences (2015)
-
Semi-quantitative measurement of specific proteins in human cumulus cells using reverse phase protein array
Reproductive Biology and Endocrinology (2013)
-
On silico peptide microarrays for high-resolution mapping of antibody epitopes and diverse protein-protein interactions
Nature Medicine (2012)