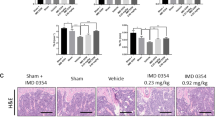
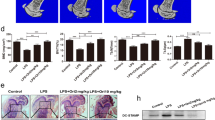
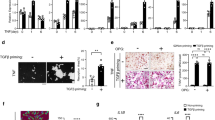
Abstract
Bone destruction is a pathological hallmark of several chronic inflammatory diseases, including rheumatoid arthritis and periodontitis. Inflammation-induced bone loss of this sort results from elevated numbers of bone-resorbing osteoclasts. Gene targeting studies have shown that the transcription factor nuclear factor-κB (NF-κB) has a crucial role in osteoclast differentiation, and blocking NF-κB is a potential strategy for preventing inflammatory bone resorption. We tested this approach using a cell-permeable peptide inhibitor of the IκB-kinase complex, a crucial component of signal transduction pathways to NF-κB. The peptide inhibited RANKL-stimulated NF-κB activation and osteoclastogenesis both in vitro and in vivo. In addition, this peptide significantly reduced the severity of collagen-induced arthritis in mice by reducing levels of tumor necrosis factor-α and interleukin-1β, abrogating joint swelling and reducing destruction of bone and cartilage. Therefore, selective inhibition of NF-κB activation offers an effective therapeutic approach for inhibiting chronic inflammatory diseases involving bone resorption.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Manolagas, S.C. Cellular and molecular mechanisms of osteoporosis. Aging (Milano) 10, 182–190 (1998).
Lam, J. et al. Tumour necrosis factor superfamily cytokines and the pathogenesis of inflammatory osteolysis. Ann. Rheum. Dis. 61 (Suppl. 2), ii82–ii83 (2002).
Kanis, J.A., Melton, L.J., III, Christiansen, C., Johnston, C.C. & Khaltaev, N. The diagnosis of osteoporosis. J. Bone Miner. Res. 9, 1137–1141 (1994).
Feldmann, M., Brennan, F.M., Foxwell, B.M. & Maini, R.N. The role of TNFα and IL-1 in rheumatoid arthritis. Curr. Dir. Autoimmun. 3, 188–199 (2001).
Baroukh, B. & Saffar, J.L. Identification of osteoclasts and their mononuclear precursors. A comparative histological and histochemical study in hamster periodontitis. J. Periodontal Res. 26, 161–166 (1991).
Horowitz, S.M. & Purdon, M.A. Mechanisms of cellular recruitment in aseptic loosening of prosthetic joint implants. Calcif. Tissue Int. 57, 301–305 (1995).
Makarov, S.S. NF-κB in rheumatoid arthritis: a pivotal regulator of inflammation, hyperplasia, and tissue destruction. Arthritis Res. 3, 200–206 (2001).
Nakashima, T., Wada, T. & Penninger, J.M. RANKL and RANK as novel therapeutic targets for arthritis. Curr. Opin. Rheumatol. 15, 280–287 (2003).
Romas, E., Gillespie, M.T. & Martin, T.J. Involvement of receptor activator of NF-κB ligand and tumor necrosis factor-α in bone destruction in rheumatoid arthritis. Bone 30, 340–346 (2002).
Boyle, W.J., Simonet, W.S. & Lacey, D.L. Osteoclast differentiation and activation. Nature 423, 337–342 (2003).
Redlich, K. et al. Osteoclasts are essential for TNF-α-mediated joint destruction. J. Clin. Invest. 110, 1419–1427 (2002).
Missbach, M. et al. A novel inhibitor of the tyrosine kinase Src suppresses phosphorylation of its major cellular substrates and reduces bone resorption in vitro and in rodent models in vivo. Bone 24, 437–449 (1999).
Yoneda, T. et al. Herbimycin A, a pp60c-src tyrosine kinase inhibitor, inhibits osteoclastic bone resorption in vitro and hypercalcemia in vivo. J. Clin. Invest. 91, 2791–2795 (1993).
Takayanagi, H. et al. Suppression of arthritic bone destruction by adenovirus-mediated csk gene transfer to synoviocytes and osteoclasts. J. Clin. Invest. 104, 137–146 (1999).
Feldmann, M. et al. Is NF-κB a useful therapeutic target in rheumatoid arthritis? Ann. Rheum. Dis. 61 (suppl.) 2, ii13–ii18 (2002).
Lacey, D.L. et al. Osteoprotegerin ligand is a cytokine that regulates osteoclast differentiation and activation. Cell 93, 165–176 (1998).
Yasuda, H. et al. Osteoclast differentiation factor is a ligand for osteoprotegerin/osteoclastogenesis-inhibitory factor and is identical to TRANCE/RANKL. Proc. Natl Acad. Sci. USA 95, 3597–3602 (1998).
Kong, Y.Y. et al. OPGL is a key regulator of osteoclastogenesis, lymphocyte development and lymph-node organogenesis. Nature 397, 315–323 (1999).
Dougall, W.C. et al. RANK is essential for osteoclast and lymph node development. Genes Dev. 13, 2412–2424 (1999).
Li, J. et al. RANK is the intrinsic hematopoietic cell surface receptor that controls osteoclastogenesis and regulation of bone mass and calcium metabolism. Proc. Natl Acad. Sci. USA 97, 1566–1571 (2000).
Iotsova, V. et al. Osteopetrosis in mice lacking NF-κB1 and NF-κB2. Nat. Med. 3, 1285–1289 (1997).
Franzoso, G. et al. Requirement for NF-κB in osteoclast and B-cell development. Genes Dev. 11, 3482–3496 (1997).
Ghosh, S. & Karin, M. Missing pieces in the NF-κB puzzle. Cell 109 (suppl.), S81–S96 (2002).
Rothwarf, D.M. & Karin, M. The NF-κB activation pathway: a paradigm in information transfer from membrane to nucleus. Sci. STKE RE1 (1999).
Karin, M. & Delhase, M. The IκB kinase (IKK) and NF-κB: key elements of proinflammatory signalling. Semin. Immunol. 12, 85–98 (2000).
May, M.J. et al. Selective inhibition of NF-κB activation by a peptide that blocks the interaction of NEMO with the IκB kinase complex. Science 289, 1550–1554 (2000).
May, M.J., Marienfeld, R.B. & Ghosh, S. Characterization of the IκB-kinase NEMO binding domain. J. Biol. Chem. 277, 45992–46000 (2002).
Rehman, K.K. et al. Protection of islets by in situ peptide-mediated transduction of the IκB kinase inhibitor Nemo-binding domain peptide. J. Biol. Chem. 278, 9862–9868 (2003).
Choi, M. et al. Inhibition of NF-κB by a TAT-NEMO-binding domain peptide accelerates constitutive apoptosis and abrogates LPS-delayed neutrophil apoptosis. Blood 102, 2259–2267 (2003).
Voll, R.E. et al. NF-κB activation by the pre-T cell receptor serves as a selective survival signal in T lymphocyte development. Immunity 13, 677–689 (2000).
Yang, X. & Karsenty, G. Transcription factors in bone: developmental and pathological aspects. Trends Mol. Med. 8, 340–345 (2002).
Theill, L.E., Boyle, W.J. & Penninger, J.M. RANK-L and RANK: T cells, bone loss, and mammalian evolution. Annu. Rev. Immunol. 20, 795–823 (2002).
Nair, S.P. et al. Bacterially induced bone destruction: mechanisms and misconceptions. Infect. Immun. 64, 2371–2380 (1996).
Arai, F. et al. Commitment and differentiation of osteoclast precursor cells by the sequential expression of c-Fms and receptor activator of nuclear factor κB (RANK) receptors. J. Exp. Med. 190, 1741–1754 (1999).
Myers, L.K., Rosloniec, E.F., Cremer, M.A. & Kang, A.H. Collagen-induced arthritis, an animal model of autoimmunity. Life Sci. 61, 1861–1878 (1997).
Delgado, M., Abad, C., Martinez, C., Leceta, J. & Gomariz, R.P. Vasoactive intestinal peptide prevents experimental arthritis by downregulating both autoimmune and inflammatory components of the disease. Nat. Med. 7, 563–568 (2001).
Mauri, C., Williams, R.O., Walmsley, M. & Feldmann, M. Relationship between Th1/Th2 cytokine patterns and the arthritogenic response in collagen-induced arthritis. Eur. J. Immunol. 26, 1511–1518 (1996).
Luross, J.A. & Williams, N.A. The genetic and immunopathological processes underlying collagen-induced arthritis. Immunology 103, 407–416 (2001).
Seetharaman, R., Mora, A.L., Nabozny, G., Boothby, M. & Chen, J. Essential role of T cell NF-κB activation in collagen-induced arthritis. J. Immunol. 163, 1577–1583 (1999).
Cuzzocrea, S. et al. Pyrrolidine dithiocarbamate attenuates the development of acute and chronic inflammation. Br. J. Pharmacol. 135, 496–510 (2002).
Carlsen, H., Moskaug, J.O., Fromm, S.H. & Blomhoff, R. In vivo imaging of NF-κB activity. J. Immunol. 168, 1441–1446 (2002).
Chen, L.W. et al. The two faces of IKK and NF-κB inhibition: prevention of systemic inflammation but increased local injury following intestinal ischemia-reperfusion. Nat. Med. 9, 575–581 (2003).
Acknowledgements
This work was supported by the National Institutes of Health (R37-AI33443) and the Howard Hughes Medical Institute (S.G.), a Scientist Development Grant from the American Heart Association (M.J.M.), by Grants-in-Aid for Scientific Research (13557151) from the Ministry of Education, Culture, Sports Science and Technology of Japan (K.A.) and by the Takeda Science Foundation (E.J.).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Jimi, E., Aoki, K., Saito, H. et al. Selective inhibition of NF-κB blocks osteoclastogenesis and prevents inflammatory bone destruction in vivo. Nat Med 10, 617–624 (2004). https://doi.org/10.1038/nm1054
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm1054
This article is cited by
-
Bone mineral density and its predictors in a cohort of adults with type 1 diabetes attending a tertiary care institute in North India
International Journal of Diabetes in Developing Countries (2024)
-
Characterization of a small-molecule inhibitor targeting NEMO/IKKβ to suppress colorectal cancer growth
Signal Transduction and Targeted Therapy (2022)
-
Transcriptome-wide association study identifies novel genes associated with bone mineral density and lean body mass in children
Endocrine (2022)
-
miR-9-5p promotes wear-particle-induced osteoclastogenesis through activation of the SIRT1/NF-κB pathway
3 Biotech (2021)
-
Recent advances in design and applications of biomimetic self-assembled peptide hydrogels for hard tissue regeneration
Bio-Design and Manufacturing (2021)