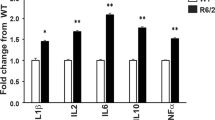
Abstract
Huntington disease is an autosomal dominant neurodegenerative disease with no effective treatment. Minocycline is a tetracycline derivative with proven safety. After ischemia, minocycline inhibits caspase-1 and inducible nitric oxide synthetase upregulation, and reduces infarction. As caspase-1 and nitric oxide seem to play a role in Huntington disease, we evaluated the therapeutic efficacy of minocycline in the R6/2 mouse model of Huntington disease. We report that minocycline delays disease progression, inhibits caspase-1 and caspase-3 mRNA upregulation, and decreases inducible nitric oxide synthetase activity. In addition, effective pharmacotherapy in R6/2 mice requires caspase-1 and caspase-3 inhibition. This is the first demonstration of caspase-1 and caspase-3 transcriptional regulation in a Huntington disease model.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
The Huntington's Disease Collaborative Research Group. A novel gene containing a trinucleotide repeat that is expanded and unstable on Huntington's disease chromosomes. Cell 72, 971–983 (1993).
Alnemri, E.S. et al. Human ICE/CED-3 protease nomenclature. Cell 87, 171 (1996).
Friedlander, R.M. & Yuan, J. ICE, neuronal apoptosis and neurodegeneration. Cell Death Differ. 5, 823–831 (1998).
Ona, V.O. et al. Inhibition of caspase-1 slows disease progression in a mouse model of Huntington's disease. Nature 399, 263–267 (1999).
Goldberg, Y.P. et al. Cleavage of huntingtin by apopain, a proapoptotic cysteine protease, is modulated by the polyglutamine tract. Nature Genet. 13, 442–449 ( 1996).
Wellington, C.L. et al. Caspase cleavage of gene products associated with triplet expansion disorders generates truncated fragments containing the polyglutamine tract. J. Biol. Chem. 273, 9158– 9167 (1998).
Aronson, A.L. Pharmacotherapeutics of the newer tetracyclines. J. Am. Vet. Med. Assoc. 176, 1061–1068 ( 1980).
Yrjanheikki, J., Keinanen, R., Pellikka, M., Hokfelt, T. & Koistinaho, J. Tetracyclines inhibit microglial activation and are neuroprotective in global brain ischemia. Proc. Natl. Acad. Sci. USA 95, 15769–15774 (1998).
Yrjanheikki, J. et al. A tetracycline derivative, minocycline, reduces inflammation and protects against focal cerebral ischemia with a wide therapeutic window . Proc. Natl. Acad. Sci. USA 96, 13496– 13500 (1999).
Tabrizi, S.J. et al. Mitochondrial dysfunction and free radical damage in the Huntington R6/2 transgenic mouse. Ann. Neurol. 47, 80–86 (2000).
Tabrizi, S.J. et al. Biochemical abnormalities and excitotoxicity in Huntington's disease brain. Ann. Neurol. 45, 25– 32 (1999).
Mangiarini, L. et al. Exon 1 of the HD gene with an expanded CAG repeat is sufficient to cause a progressive neurological phenotype in transgenic mice. Cell 87, 493–506 ( 1996).
Davies, S.W. et al. Formation of neuronal intranuclear inclusions underlies the neurological dysfunction in mice transgenic for the HD mutation. Cell 90, 537–548 ( 1997).
Cha, J.H. et al. Altered brain neurotransmitter receptors in transgenic mice expressing a portion of an abnormal human huntington disease gene. Proc. Natl. Acad. Sci. USA 95, 6480–6485 (1998).
Li, P. et al. Mice deficient in IL-1 beta-converting enzyme are defective in production of mature IL-1 beta and resistant to endotoxic shock. Cell 80, 401–411 ( 1995).
Kuida, K. et al. Altered cytokine export and apoptosis in mice deficient in interleukin-1 beta converting enzyme. Science 267, 2000 –2003 (1995).
Friedlander, R.M. et al. Expression of a dominant negative mutant of interleukin-1 beta converting enzyme in transgenic mice prevents neuronal cell death induced by trophic factor withdrawal and ischemic brain injury. J. Exp. Med. 185, 933–940 ( 1997).
Hara, H. et al. Inhibition of interleukin 1beta converting enzyme family proteases reduces ischemic and excitotoxic neuronal damage. Proc. Natl. Acad. Sci. USA 94, 2007–2012 ( 1997).
White, J.K. et al. Huntingtin is required for neurogenesis and is not impaired by the Huntington's disease CAG expansion. Nature Genet. 17, 404–410 (1997).
DiFiglia, M. et al. Aggregation of huntingtin in neuronal intranuclear inclusions and dystrophic neurites in brain. Science 277, 1990–1993 (1997).
Forss-Petter, S. et al. Transgenic mice expressing beta-galactosidase in mature neurons under neuron-specific enolase promoter control. Neuron 5, 187–197 (1990).
Enari, M., Talanian, R.V., Wong, W.W. & Nagata, S. Sequential activation of ICE-like and CPP32-like proteases during Fas-mediated apoptosis. Nature 380, 723– 736 (1996).
Li, M. et al. Functional role of caspase-1 and caspase-3 in an ALS transgenic mouse model. Science 288, 335– 339 (2000).
Browne, S.E. et al. Oxidative damage and metabolic dysfunction in Huntington's disease: selective vulnerability of the basal ganglia. Ann. Neurol. 41, 646–653 ( 1997).
Schapira, A.H. Mitochondrial dysfunction in neurodegenerative disorders. Biochim. Biophys. Acta. 1366, 225–233 (1998).
Tewari, M. et al. Yama/CPP32 beta, a mammalian homolog of CED-3, is a CrmA-inhibitable protease that cleaves the death substrate poly(ADP-ribose) polymerase. Cell 81, 801–809 ( 1995).
Goulden, V., Glass, D. & Cunliffe, W.J. Safety of long-term high-dose minocycline in the treatment of acne. Br. J. Dermatol. 134, 693– 695 (1996).
Gottlieb, A. Safety of minocycline for acne. Lancet 349, 374 (1997).
Bredt, D.S. & Snyder, S.H. Isolation of nitric oxide synthetase, a calmodulin-requiring enzyme. Proc. Natl. Acad. Sci. USA 87, 682–685 (1990).
Yoshida, T., Waeber, C., Huang, Z. & Moskowitz, M.A. Induction of nitric oxide synthase activity in rodent brain following middle cerebral artery occlusion. Neurosci. Lett. 194, 214 –218 (1995).
Ferrante, R.J. et al. Heterogeneous topographic and cellular distribution of huntingtin expression in the normal human neostriatum. J. Neurosci. 17, 3052–3063 (1997).
Acknowledgements
The authors thank E. Friedlander for editorial assistance. This work was supported by a grant from the Hereditary Disease Foundation (to R.M.F. and J.H.J.C), the Huntington Disease Society of America (to R.M.F. and J.H.J.C), the National Institutes of Health (to R.M.F., J.H.J.C, R.J.F and S.M.H) and the Veterans Administration (R.J.F.). R.M.F. is a member of the Cure Huntington's Disease Initiative of the Hereditary Disease Foundation and of the Coalition for the Cure from the Huntington's Disease Society of America.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chen, M., Ona, V., Li, M. et al. Minocycline inhibits caspase-1 and caspase-3 expression and delays mortality in a transgenic mouse model of Huntington disease. Nat Med 6, 797–801 (2000). https://doi.org/10.1038/77528
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/77528
This article is cited by
-
Neuromodulatory effect of 4-(methylthio)butyl isothiocyanate against 3-nitropropionic acid induced oxidative impairments in human dopaminergic SH-SY5Y cells via BDNF/CREB/TrkB pathway
Scientific Reports (2023)
-
Repurposing sarecycline for osteoinductive therapies: an in vitro and ex vivo assessment
Journal of Bone and Mineral Metabolism (2023)
-
Tetracycline ameliorates silica-induced pulmonary inflammation and fibrosis via inhibition of caspase-1
Respiratory Research (2022)
-
Inflammasome Signaling in the Aging Brain and Age-Related Neurodegenerative Diseases
Molecular Neurobiology (2022)
-
Challenges of repurposing tetracyclines for the treatment of Alzheimer’s and Parkinson’s disease
Journal of Neural Transmission (2022)