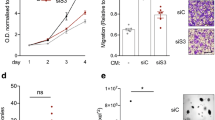
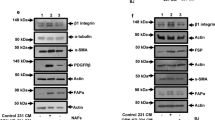
Abstract
Breast tumors of the basal-like, hormone receptor–negative subtype remain an unmet clinical challenge, as there is high rate of recurrence and poor survival in patients following treatment. Coevolution of the malignant mammary epithelium and its underlying stroma instigates cancer-associated fibroblasts (CAFs) to support most, if not all, hallmarks of cancer progression. Here we delineate a previously unappreciated role for CAFs as determinants of the molecular subtype of breast cancer. We identified paracrine crosstalk between cancer cells expressing platelet-derived growth factor (PDGF)-CC and CAFs expressing the cognate receptors in human basal-like mammary carcinomas. Genetic or pharmacological intervention of PDGF-CC activity in mouse models of cancer resulted in conversion of basal-like breast cancers into a hormone receptor-positive state that enhanced sensitivity to endocrine therapy in previously resistant tumors. We conclude that specification of breast cancer to the basal-like subtype is under microenvironmental control and is therapeutically actionable.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Accession codes
References
Perou, C.M. et al. Molecular portraits of human breast tumours. Nature 406, 747–752 (2000).
Goldhirsch, A. et al. Thresholds for therapies: highlights of the St Gallen International Expert Consensus on the primary therapy of early breast cancer 2009. Ann. Oncol. 20, 1319–1329 (2009).
Sørlie, T. et al. Gene expression patterns of breast carcinomas distinguish tumor subclasses with clinical implications. Proc. Natl. Acad. Sci. USA 98, 10869–10874 (2001).
Goldhirsch, A. et al. Personalizing the treatment of women with early breast cancer: highlights of the St Gallen International Expert Consensus on the Primary Therapy of Early Breast Cancer 2013. Ann. Oncol. 24, 2206–2223 (2013).
Goldhirsch, A. et al. Strategies for subtypes—dealing with the diversity of breast cancer: highlights of the St. Gallen International Expert Consensus on the Primary Therapy of Early Breast Cancer 2011. Ann. Oncol. 22, 1736–1747 (2011).
Prat, A. et al. Clinical implications of the intrinsic molecular subtypes of breast cancer. Breast 24 (Suppl. 2), S26–S35 (2015).
Ignatiadis, M. & Sotiriou, C. Luminal breast cancer: from biology to treatment. Nat. Rev. Clin. Oncol. 10, 494–506 (2013).
Voduc, K.D. et al. Breast cancer subtypes and the risk of local and regional relapse. J. Clin. Oncol. 28, 1684–1691 (2010).
Prat, A. & Perou, C.M. Deconstructing the molecular portraits of breast cancer. Mol. Oncol. 5, 5–23 (2011).
Hanahan, D. & Coussens, L.M. Accessories to the crime: functions of cells recruited to the tumor microenvironment. Cancer Cell 21, 309–322 (2012).
Pietras, K. & Ostman, A. Hallmarks of cancer: interactions with the tumor stroma. Exp. Cell Res. 316, 1324–1331 (2010).
Kalluri, R. & Zeisberg, M. Fibroblasts in cancer. Nat. Rev. Cancer 6, 392–401 (2006).
Cortez, E., Roswall, P. & Pietras, K. Functional subsets of mesenchymal cell types in the tumor microenvironment. Semin. Cancer Biol. 25, 3–9 (2014).
Augsten, M. Cancer-associated fibroblasts as another polarized cell type of the tumor microenvironment. Front. Oncol. 4, 62 (2014).
Kim, H.M., Jung, W.H. & Koo, J.S. Expression of cancer-associated fibroblast related proteins in metastatic breast cancer: an immunohistochemical analysis. J. Transl. Med. 13, 222 (2015).
Anderberg, C. et al. Paracrine signaling by platelet-derived growth factor-CC promotes tumor growth by recruitment of cancer-associated fibroblasts. Cancer Res. 69, 369–378 (2009).
Pietras, K., Pahler, J., Bergers, G. & Hanahan, D. Functions of paracrine PDGF signaling in the proangiogenic tumor stroma revealed by pharmacological targeting. PLoS Med. 5, e19 (2008).
Li, X. et al. PDGF-C is a new protease-activated ligand for the PDGF α-receptor. Nat. Cell Biol. 2, 302–309 (2000).
Cao, R. et al. Angiogenesis stimulated by PDGF-CC, a novel member in the PDGF family, involves activation of PDGFR-αα and -αβ receptors. FASEB J. 16, 1575–1583 (2002).
Theurillat, J.P. et al. NY-ESO-1 protein expression in primary breast carcinoma and metastases: correlation with CD8+ T-cell and CD79a+ plasmacytic/B-cell infiltration. Int. J. Cancer 120, 2411–2417 (2007).
Falck, A.K. et al. Biomarker expression and St Gallen molecular subtype classification in primary tumours, synchronous lymph node metastases and asynchronous relapses in primary breast cancer patients with 10 years' follow-up. Breast Cancer Res. Treat. 140, 93–104 (2013).
Falck, A.K. et al. Analysis of and prognostic information from disseminated tumour cells in bone marrow in primary breast cancer: a prospective observational study. BMC Cancer 12, 403 (2012).
Guy, C.T., Cardiff, R.D. & Muller, W.J. Induction of mammary tumors by expression of polyomavirus middle T oncogene: a transgenic mouse model for metastatic disease. Mol. Cell. Biol. 12, 954–961 (1992).
Lin, E.Y. et al. Progression to malignancy in the polyoma middle T oncoprotein mouse breast cancer model provides a reliable model for human diseases. Am. J. Pathol. 163, 2113–2126 (2003).
Ding, H. et al. A specific requirement for PDGF-C in palate formation and PDGFR-alpha signaling. Nat. Genet. 36, 1111–1116 (2004).
Cancer Genome Atlas, N.; Cancer Genome Atlas Network. Comprehensive molecular portraits of human breast tumours. Nature 490, 61–70 (2012).
Badve, S. et al. FOXA1 expression in breast cancer—correlation with luminal subtype A and survival. Clin. Cancer Res. 13, 4415–4421 (2007).
Thorat, M.A. et al. Forkhead box A1 expression in breast cancer is associated with luminal subtype and good prognosis. J. Clin. Pathol. 61, 327–332 (2008).
Neve, R.M. et al. A collection of breast cancer cell lines for the study of functionally distinct cancer subtypes. Cancer Cell 10, 515–527 (2006).
Kong, S.L., Li, G., Loh, S.L., Sung, W.K. & Liu, E.T. Cellular reprogramming by the conjoint action of ERα, FOXA1, and GATA3 to a ligand-inducible growth state. Mol. Syst. Biol. 7, 526 (2011).
Kojima, Y. et al. Autocrine TGF-β and stromal cell-derived factor-1 (SDF-1) signaling drives the evolution of tumor-promoting mammary stromal myofibroblasts. Proc. Natl. Acad. Sci. USA 107, 20009–20014 (2010).
Ray, P.S. et al. FOXC1 is a potential prognostic biomarker with functional significance in basal-like breast cancer. Cancer Res. 70, 3870–3876 (2010).
Chebil, G., Bendahl, P.O., Idvall, I. & Fernö, M. Comparison of immunohistochemical and biochemical assay of steroid receptors in primary breast cancer—clinical associations and reasons for discrepancies. Acta Oncol. 42, 719–725 (2003).
Hammond, M.E. et al. American Society of Clinical Oncology/College Of American Pathologists guideline recommendations for immunohistochemical testing of estrogen and progesterone receptors in breast cancer. J. Clin. Oncol. 28, 2784–2795 (2010).
Polyak, K. Breast cancer: origins and evolution. J. Clin. Invest. 117, 3155–3163 (2007).
Gupta, P.B. et al. Stochastic state transitions give rise to phenotypic equilibrium in populations of cancer cells. Cell 146, 633–644 (2011).
Lim, E. et al. Aberrant luminal progenitors as the candidate target population for basal tumor development in BRCA1 mutation carriers. Nat. Med. 15, 907–913 (2009).
Molyneux, G. & Smalley, M.J. The cell of origin of BRCA1 mutation-associated breast cancer: a cautionary tale of gene expression profiling. J. Mammary Gland Biol. Neoplasia 16, 51–55 (2011).
Liu, S. et al. BRCA1 regulates human mammary stem/progenitor cell fate. Proc. Natl. Acad. Sci. USA 105, 1680–1685 (2008).
Molyneux, G. et al. BRCA1 basal-like breast cancers originate from luminal epithelial progenitors and not from basal stem cells. Cell Stem Cell 7, 403–417 (2010).
Su, Y. et al. Somatic cell fusions reveal extensive heterogeneity in basal-like breast cancer. Cell Rep. 11, 1549–1563 (2015).
Yamamoto, S. et al. JARID1B is a luminal lineage-driving oncogene in breast cancer. Cancer Cell 25, 762–777 (2014).
Bernardo, G.M. et al. FOXA1 represses the molecular phenotype of basal breast cancer cells. Oncogene 32, 554–563 (2013).
Sflomos, G. et al. A preclinical model for ERα-positive breast cancer points to the epithelial microenvironment as determinant of luminal phenotype and hormone response. Cancer Cell 29, 407–422 (2016).
Tam, W.L. et al. Protein kinase C α is a central signaling node and therapeutic target for breast cancer stem cells. Cancer Cell 24, 347–364 (2013).
Meng, F. et al. PDGFRα and β play critical roles in mediating Foxq1-driven breast cancer stemness and chemoresistance. Cancer Res. 75, 584–593 (2015).
Jansson, S. et al. The three receptor tyrosine kinases c-KIT, VEGFR2 and PDGFRα, closely spaced at 4q12, show increased protein expression in triple-negative breast cancer. PLoS One 9, e102176 (2014).
Horikawa, S. et al. PDGFRα plays a crucial role in connective tissue remodeling. Sci. Rep. 5, 17948 (2015).
Kim, Y.J. et al. MET is a potential target for use in combination therapy with EGFR inhibition in triple-negative/basal-like breast cancer. Int. J. Cancer 134, 2424–2436 (2014).
Ho-Yen, C.M. et al. C-Met in invasive breast cancer: is there a relationship with the basal-like subtype? Cancer 120, 163–171 (2014).
Ponzo, M.G. et al. Met induces mammary tumors with diverse histologies and is associated with poor outcome and human basal breast cancer. Proc. Natl. Acad. Sci. USA 106, 12903–12908 (2009).
Graveel, C.R. et al. Met induces diverse mammary carcinomas in mice and is associated with human basal breast cancer. Proc. Natl. Acad. Sci. USA 106, 12909–12914 (2009).
Gastaldi, S. et al. Met signaling regulates growth, repopulating potential and basal cell-fate commitment of mammary luminal progenitors: implications for basal-like breast cancer. Oncogene 32, 1428–1440 (2013).
Marzec, K.A., Baxter, R.C. & Martin, J.L. Targeting insulin-like growth factor binding protein-3 signaling in triple-negative breast cancer. BioMed Res. Int. 2015, 638526 (2015).
Bissell, M.J. & Hines, W.C. Why don't we get more cancer? A proposed role of the microenvironment in restraining cancer progression. Nat. Med. 17, 320–329 (2011).
Marsh, T., Pietras, K. & McAllister, S.S. Fibroblasts as architects of cancer pathogenesis. Biochim. Biophys. Acta 1832, 1070–1078 (2013).
Gascard, P. & Tlsty, T.D. Carcinoma-associated fibroblasts: orchestrating the composition of malignancy. Genes Dev. 30, 1002–1019 (2016).
Brechbuhl, H.M. et al. Fibroblast subtypes regulate responsiveness of luminal breast cancer to estrogen. Clin. Cancer Res. 23, 1710–1721 (2017).
Özdemir, B.C. et al. Depletion of carcinoma-associated fibroblasts and fibrosis induces immunosuppression and accelerates pancreas cancer with reduced survival. Cancer Cell 25, 719–734 (2014).
Rhim, A.D. et al. Stromal elements act to restrain, rather than support, pancreatic ductal adenocarcinoma. Cancer Cell 25, 735–747 (2014).
Parker, J.S. et al. Supervised risk predictor of breast cancer based on intrinsic subtypes. J. Clin. Oncol. 27, 1160–1167 (2009).
Bray, N.L., Pimentel, H., Melsted, P. & Pachter, L. Near-optimal probabilistic RNA-seq quantification. Nat. Biotechnol. 34, 525–527 (2016).
Pimentel, H. et al. Differential analysis of RNA-seq incorporating quantification uncertainty. Nat. Methods 14, 687–690 (2017).
Mei, S. et al. Cistrome Data Browser: a data portal for ChIP–seq and chromatin accessibility data in human and mouse. Nucleic Acids Res. 45 D1, D658–D662 (2017).
Sikora-Wohlfeld, W., Ackermann, M., Christodoulou, E.G., Singaravelu, K. & Beyer, A. Assessing computational methods for transcription factor target gene identification based on ChIP–seq data. PLOS Comput. Biol. 9, e1003342 (2013).
Subramanian, A. et al. Gene set enrichment analysis: a knowledge-based approach for interpreting genome-wide expression profiles. Proc. Natl. Acad. Sci. USA 102, 15545–15550 (2005).
Mootha, V.K. et al. PGC-1α-responsive genes involved in oxidative phosphorylation are coordinately downregulated in human diabetes. Nat. Genet. 34, 267–273 (2003).
Acknowledgements
We gratefully acknowledge expert help with pathology assessments from the late D. Grabau and provision of the Tam2Y cohort by M. Fernö. Further, we would like to thank D. Cao, M. O'Brien and C. Murone for their technical support and P.-O. Bendahl for statistical assistance. K.P. is the Göran & Birgitta Grosskopf Professor at Lund University. The research presented herein was supported by grants from the following agencies to K.P.: a Consolidator Grant from the European Research Council (the TUMORGAN project, grant 309322), the Swedish Research Council, the Swedish Cancer Society, the STARGET consortium (a Swedish Research Council Linnaeus network), BioCARE and Lund University. U.E. acknowledges funding support from the Swedish Research Council, the Swedish Cancer Society, Karolinska Institutet and Ludwig Institute for Cancer Research. A.M.S. acknowledges funding support from National Health and Medical Research Council (NHMRC) Fellowship 1084178 and Grant 10927888 and the Operational Infrastructure Support Program provided by the Victorian Government, Australia.
Author information
Authors and Affiliations
Contributions
P.R. generated and analyzed data and conceived the study. M. Bocci, M. Bartoschek, H.L., G.K., S.J., S.L., J.S., C.L., P.E., S.R., C.A., E. Cortez, L.H.S., C.O.-P., B.K.H. and J.H. generated and analyzed data. E. Cordero, I.J.G.B. and E.L. generated data. A.O. provided exclusive reagents. M.H. analyzed data. L.R., H.M. and A.M.S. analyzed data and provided exclusive reagents. U.E. analyzed data, provided exclusive reagents and conceived the study. K.P. generated and analyzed data, conceived the study, managed the study and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
K.P., U.E. and P.R. are named inventors on Patent Cooperation Treaty (PCT) application no. PCT/EP2016/077295, which is related to the findings of the current study. K.P., U.E. and A.M.S. are shareholders of Paracrine Therapeutics, which develops inhibitory agents to PDGF-CC.
Supplementary information
Supplementary Figures & Tables
Supplementary Figures 1–12 & Supplementary Tables 1–8 (PDF 4066 kb)
Rights and permissions
About this article
Cite this article
Roswall, P., Bocci, M., Bartoschek, M. et al. Microenvironmental control of breast cancer subtype elicited through paracrine platelet-derived growth factor-CC signaling. Nat Med 24, 463–473 (2018). https://doi.org/10.1038/nm.4494
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.4494
This article is cited by
-
The interaction of platelet-related factors with tumor cells promotes tumor metastasis
Journal of Translational Medicine (2024)
-
Cancer-associated fibroblasts rewire the estrogen receptor response in luminal breast cancer, enabling estrogen independence
Oncogene (2024)
-
The role of stromal cells in epithelial–mesenchymal plasticity and its therapeutic potential
Discover Oncology (2024)
-
From complexity to clarity: unravelling tumor heterogeneity through the lens of tumor microenvironment for innovative cancer therapy
Histochemistry and Cell Biology (2024)
-
Modeling stress-induced responses: plasticity in continuous state space and gradual clonal evolution
Theory in Biosciences (2024)