Abstract
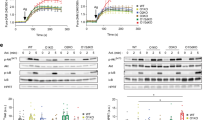
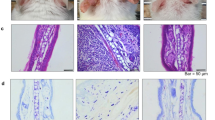
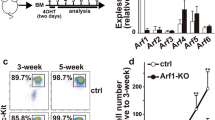
Critical to the function of mast cells in immune responses including allergy is their production of lipid mediators, among which only omega-6 (ω-6) arachidonate–derived eicosanoids have been well characterized. Here, by employing comprehensive lipidomics, we identify omega-3 (ω-3) fatty acid epoxides as new mast cell–derived lipid mediators and show that they are produced by PAF-AH2, an oxidized-phospholipid-selective phospholipase A2. Genetic or pharmacological deletion of PAF-AH2 reduced the steady-state production of ω-3 epoxides, leading to attenuated mast cell activation and anaphylaxis following FcɛRI cross-linking. Mechanistically, the ω-3 epoxides promote IgE-mediated activation of mast cells by downregulating Srcin1, a Src-inhibitory protein that counteracts FcɛRI signaling, through a pathway involving PPARg. Thus, the PAF-AH2–ω-3 epoxide–Srcin1 axis presents new potential drug targets for allergic diseases.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Accession codes
References
Devereux, G. The increase in the prevalence of asthma and allergy: food for thought. Nat. Rev. Immunol. 6, 869–874 (2006).
Kalesnikoff, J. & Galli, S.J. New developments in mast cell biology. Nat. Immunol. 9, 1215–1223 (2008).
Serhan, C.N. Pro-resolving lipid mediators are leads for resolution physiology. Nature 510, 92–101 (2014).
Nagakura, T., Matsuda, S., Shichijyo, K., Sugimoto, H. & Hata, K. Dietary supplementation with fish oil rich in omega-3 polyunsaturated fatty acids in children with bronchial asthma. Eur. Respir. J. 16, 861–865 (2000).
Peat, J.K. et al. Three-year outcomes of dietary fatty acid modification and house dust mite reduction in the Childhood Asthma Prevention Study. J. Allergy Clin. Immunol. 114, 807–813 (2004).
Murakami, M. et al. Recent progress in phospholipase A2 research: from cells to animals to humans. Prog. Lipid Res. 50, 152–192 (2011).
Sugiyama, H. et al. Peroxisome proliferator–activated receptors are expressed in mouse bone marrow–derived mast cells. FEBS Lett. 467, 259–262 (2000).
Murakami, M., Matsumoto, R., Urade, Y., Austen, K.F. & Arm, J.P. c-Kit ligand mediates increased expression of cytosolic phospholipase A2, prostaglandin endoperoxide synthase-1, and hematopoietic prostaglandin D2 synthase and increased IgE-dependent prostaglandin D2 generation in immature mouse mast cells. J. Biol. Chem. 270, 3239–3246 (1995).
Fujishima, H. et al. Cytosolic phospholipase A2 is essential for both the immediate and the delayed phases of eicosanoid generation in mouse bone marrow–derived mast cells. Proc. Natl. Acad. Sci. USA 96, 4803–4807 (1999).
Uozumi, N. et al. Role of cytosolic phospholipase A2 in allergic response and parturition. Nature 390, 618–622 (1997).
Bae, K. et al. Platelet-activating factor (PAF)-dependent transacetylase and its relationship with PAF acetylhydrolases. J. Biol. Chem. 275, 26704–26709 (2000).
Costello, P.S. et al. Critical role for the tyrosine kinase Syk in signalling through the high affinity IgE receptor of mast cells. Oncogene 13, 2595–2605 (1996).
Nishida, K. et al. FcɛRI-mediated mast cell degranulation requires calcium-independent microtubule-dependent translocation of granules to the plasma membrane. J. Cell Biol. 170, 115–126 (2005).
Lee, J.H. et al. The Src family kinase Fgr is critical for activation of mast cells and IgE-mediated anaphylaxis in mice. J. Immunol. 187, 1807–1815 (2011).
Hong, H. et al. The Src family kinase Hck regulates mast cell activation by suppressing an inhibitory Src family kinase Lyn. Blood 110, 2511–2519 (2007).
Suzuki, R. et al. Molecular editing of cellular responses by the high-affinity receptor for IgE. Science 343, 1021–1025 (2014).
Matsuzawa, A., Hattori, K., Aoki, J., Arai, H. & Inoue, K. Protection against oxidative stress–induced cell death by intracellular platelet-activating factor–acetylhydrolase II. J. Biol. Chem. 272, 32315–32320 (1997).
Morisseau, C. & Hammock, B.D. Impact of soluble epoxide hydrolase and epoxyeicosanoids on human health. Annu. Rev. Pharmacol. Toxicol. 53, 37–58 (2013).
Stafforini, D.M. et al. Release of free F2-isoprostanes from esterified phospholipids is catalyzed by intracellular and plasma platelet-activating factor acetylhydrolases. J. Biol. Chem. 281, 4616–4623 (2006).
Adibekian, A. et al. Click-generated triazole ureas as ultrapotent in vivo–active serine hydrolase inhibitors. Nat. Chem. Biol. 7, 469–478 (2011).
Di Stefano, P. et al. p140Cap protein suppresses tumour cell properties, regulating Csk and Src kinase activity. EMBO J. 26, 2843–2855 (2007).
Latour, S. & Veillette, A. Proximal protein tyrosine kinases in immunoreceptor signaling. Curr. Opin. Immunol. 13, 299–306 (2001).
Li, J.P. et al. The phosphatase JKAP/DUSP22 inhibits T-cell receptor signalling and autoimmunity by inactivating Lck. Nat. Commun. 5, 3618 (2014).
Yoshioka, K. et al. Endothelial PI3K-C2α, a class II PI3K, has an essential role in angiogenesis and vascular barrier function. Nat. Med. 18, 1560–1569 (2012).
Taketomi, Y. et al. Mast cell maturation is driven via a group III phospholipase A2–prostaglandin D2–DP1 receptor paracrine axis. Nat. Immunol. 14, 554–563 (2013).
Vadas, P. et al. Platelet-activating factor, PAF acetylhydrolase, and severe anaphylaxis. N. Engl. J. Med. 358, 28–35 (2008).
Hercule, H.C. et al. The vasodilator 17,18-epoxyeicosatetraenoic acid targets the pore-forming BKα channel subunit in rodents. Exp. Physiol. 92, 1067–1076 (2007).
López-Vicario, C. et al. Inhibition of soluble epoxide hydrolase modulates inflammation and autophagy in obese adipose tissue and liver: role for omega-3 epoxides. Proc. Natl. Acad. Sci. USA 112, 536–541 (2015).
Zhang, G. et al. Epoxy metabolites of docosahexaenoic acid (DHA) inhibit angiogenesis, tumor growth, and metastasis. Proc. Natl. Acad. Sci. USA 110, 6530–6535 (2013).
Aldrovandi, M. et al. Human platelets generate phospholipid-esterified prostaglandins via cyclooxygenase-1 that are inhibited by low dose aspirin supplementation. J. Lipid Res. 54, 3085–3097 (2013).
Sugiyama, H. et al. Peroxisome proliferator–activated receptors are expressed in human cultured mast cells: a possible role of these receptors in negative regulation of mast cell activation. Eur. J. Immunol. 30, 3363–3370 (2000).
Torres, R.C. et al. Activation of PPARγ by restores mast cell numbers and reactivity in alloxan-diabetic rats by reducing the systemic glucocorticoid levels. Eur. J. Pharmacol. 691, 261–267 (2012).
Ahmadian, M. et al. PPARγ signaling and metabolism: the good, the bad and the future. Nat. Med. 19, 557–566 (2013).
Wakabayashi, K. et al. The peroxisome proliferator–activated receptor γ/retinoid X receptor α heterodimer targets the histone modification enzyme PR-Set7/Setd8 gene and regulates adipogenesis through a positive feedback loop. Mol. Cell. Biol. 29, 3544–3555 (2009).
Szatmari, I. et al. PPARγ regulates the function of human dendritic cells primarily by altering lipid metabolism. Blood 110, 3271–3280 (2007).
Step, S.E. et al. Anti-diabetic rosiglitazone remodels the adipocyte transcriptome by redistributing transcription to PPARγ-driven enhancers. Genes Dev. 28, 1018–1028 (2014).
Ohtake, H., Ichikawa, N., Okada, M. & Yamashita, T. Transmembrane phosphoprotein Csk-binding protein/phosphoprotein associated with glycosphingolipid-enriched microdomains as a negative feedback regulator of mast cell signaling through the FcɛRI. J. Immunol. 168, 2087–2090 (2002).
Broadfield, E.C. et al. A case–control study of dietary and erythrocyte membrane fatty acids in asthma. Clin. Exp. Allergy 34, 1232–1236 (2004).
Woods, R.K., Raven, J.M., Walters, E.H., Abramson, M.J. & Thien, F.C. Fatty acid levels and risk of asthma in young adults. Thorax 59, 105–110 (2004).
Almqvist, C. et al. Omega-3 and omega-6 fatty acid exposure from early life does not affect atopy and asthma at age 5 years. J. Allergy Clin. Immunol. 119, 1438–1444 (2007).
Chiurchiù, V. et al. Proresolving lipid mediators resolvin D1, resolvin D2, and maresin 1 are critical in modulating T cell responses. Sci. Transl. Med. 8, 353ra111 (2016).
Colegio, O.R. et al. Functional polarization of tumour-associated macrophages by tumour-derived lactic acid. Nature 513, 559–563 (2014).
Miki, Y. et al. Dual roles of group IID phospholipase A2 in inflammation and cancer. J. Biol. Chem. 291, 15588–15601 (2016).
Rivera, J., Kinet, J.P., Kim, J., Pucillo, C. & Metzger, H. Studies with a monoclonal antibody to the β subunit of the receptor with high affinity for immunoglobulin E. Mol. Immunol. 25, 647–661 (1988).
Kono, N. et al. Protection against oxidative stress–induced hepatic injury by intracellular type II platelet-activating factor acetylhydrolase by metabolism of oxidized phospholipids in vivo. J. Biol. Chem. 283, 1628–1636 (2008).
Yoda, E. et al. Mitochondrial dysfunction and reduced prostaglandin synthesis in skeletal muscle of group VIB Ca2+-independent phospholipase A2γ-deficient mice. J. Lipid Res. 51, 3003–3015 (2010).
Bligh, E.G. & Dyer, W.J. A rapid method of total lipid extraction and purification. Can. J. Biochem. Physiol. 37, 911–917 (1959).
Fujisawa, D. et al. Expression of Mas-related gene X2 on mast cells is upregulated in the skin of patients with severe chronic urticaria. J. Allergy Clin. Immunol. 134, 622–633 (2014).
Lee, H. et al. Activation of human synovial mast cells from rheumatoid arthritis or osteoarthritis patients in response to aggregated IgG through Fcγ receptor I and Fcγ receptor II. Arthritis Rheum. 65, 109–119 (2013).
Acknowledgements
We thank K. Mukai, Y. Takada, T. Fusuma (University of Tokyo) and T. Sakamoto (Nihon University School of Medicine) for their assistance and Y. Nozaki, M. Mikamoto (Eisai Co. Ltd.) and T. Imai (Kumamoto University) for their suggestions on the in vivo pharmacokinetic studies in mice. This work was supported by Grants-in-Aid for JSPS Research Fellow (Y.S.), for Young Scientists (B) (grant number 21790060 to N.K.), for Scientific Research (A) (grant number 16H02613 to M.M.), for Challenging Research (16K15122 to Y.T.), for Scientific Research (S) (grant numbers 23227004 and 17H06164 to H.A.) and for Scientific Research on Innovative Areas (grant numbers 15H05905 to M.M. and 17H06418 to H.A.); by the Science and Technology Research Promotion Program for Agriculture, Forestry, Fisheries and Food Industry (H.A.); by AMED-CREST, AMED (H.A. and M.M.); by PRIME, AMED (N.K.); by the Ono Medical Research Foundation (N.K.); and by the Cosmetology Research Foundation (N.K.).
Author information
Authors and Affiliations
Contributions
Y.S. and N.K. designed and performed the experiments, analyzed the data, interpreted the results and wrote the manuscript. Y. Taketomi performed the experiments with mast cell engraftment and interpreted the results. M.A. performed comprehensive lipidomics analysis. Y.O. aided in experiments with human mast cells. Y. Tanaka performed experiments with electroporation-mediated gene silencing. Y.N. performed microarray analysis. T.M. and H.K. performed pharmacokinetic studies and interpreted the results. A.A. and B.C. supplied reagents and interpreted the results. M.M. and H.A. designed the experiments, interpreted the results and wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text, Figures and Tables
Supplementary Note, Supplementary Figures 1–7 and Supplementary Tables 1–5 (PDF 11256 kb)
Rights and permissions
About this article
Cite this article
Shimanaka, Y., Kono, N., Taketomi, Y. et al. Omega-3 fatty acid epoxides are autocrine mediators that control the magnitude of IgE-mediated mast cell activation. Nat Med 23, 1287–1297 (2017). https://doi.org/10.1038/nm.4417
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.4417
This article is cited by
-
Omega-3 fatty acid epoxides produced by PAF-AH2 in mast cells regulate pulmonary vascular remodeling
Nature Communications (2022)
-
LimeMap: a comprehensive map of lipid mediator metabolic pathways
npj Systems Biology and Applications (2021)
-
A chemical–genetic screen identifies ABHD12 as an oxidized-phosphatidylserine lipase
Nature Chemical Biology (2019)
-
Comprehensive analysis of the mouse cytochrome P450 family responsible for omega-3 epoxidation of eicosapentaenoic acid
Scientific Reports (2018)