Abstract
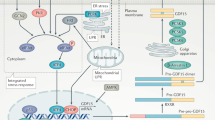
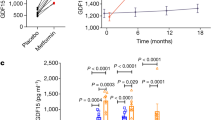
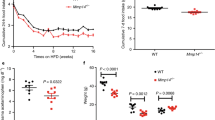
Growth differentiation factor 15 (GDF15; also known as MIC-1) is a divergent member of the TGF-β superfamily and is associated with body-weight regulation in humans and rodents. However, the cognate receptor of GDF15 is unknown. Here we show that GDF15 binds specifically to GDNF family receptor α-like (GFRAL) with high affinity, and that GFRAL requires association with the coreceptor RET to elicit intracellular signaling in response to GDF15 stimulation. We also found that GDF15-mediated reductions in food intake and body weight of mice with obesity were abolished in GFRAL-knockout mice. We further found that GFRAL expression was limited to hindbrain neurons and not present in peripheral tissues, which suggests that GDF15–GFRAL-mediated regulation of food intake is by a central mechanism. Lastly, given that GDF15 did not increase energy expenditure in treated mice with obesity, the anti-obesity actions of the cytokine are likely driven primarily by a reduction in food intake.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Johnen, H. et al. Tumor-induced anorexia and weight loss are mediated by the TGF-beta superfamily cytokine MIC-1. Nat. Med. 13, 1333–1340 (2007).
Macia, L. et al. Macrophage inhibitory cytokine 1 (MIC-1/GDF15) decreases food intake, body weight and improves glucose tolerance in mice on normal & obesogenic diets. PLoS One 7, e34868 (2012).
Chrysovergis, K. et al. NAG-1/GDF-15 prevents obesity by increasing thermogenesis, lipolysis and oxidative metabolism. Int. J. Obes. 38, 1555–1564 (2014).
Tsai, V.W. et al. Serum levels of human MIC-1/GDF15 vary in a diurnal pattern, do not display a profile suggestive of a satiety factor and are related to BMI. PLoS One 10, e0133362 (2015).
Tsai, V.W. et al. The anorectic actions of the TGFβ cytokine MIC-1/GDF15 require an intact brainstem area postrema and nucleus of the solitary tract. PLoS One 9, e100370 (2014).
Böttner, M. et al. Characterization of the rat, mouse, and human genes of growth/differentiation factor-15/macrophage inhibiting cytokine-1 (GDF-15/MIC-1). Gene 237, 105–111 (1999).
Bootcov, M.R. et al. MIC-1, a novel macrophage inhibitory cytokine, is a divergent member of the TGF-beta superfamily. Proc. Natl. Acad. Sci. USA 94, 11514–11519 (1997).
Valdimarsdottir, G. & Mummery, C. Functions of the TGFbeta superfamily in human embryonic stem cells. APMIS 113, 773–789 (2005).
Herpin, A., Lelong, C. & Favrel, P. Transforming growth factor-beta-related proteins: an ancestral and widespread superfamily of cytokines in metazoans. Dev. Comp. Immunol. 28, 461–485 (2004).
Chen, W. & Ten Dijke, P. Immunoregulation by members of the TGFβ superfamily. Nat. Rev. Immunol. 16, 723–740 (2016).
Airaksinen, M.S., Holm, L. & Hätinen, T. Evolution of the GDNF family ligands and receptors. Brain Behav. Evol. 68, 181–190 (2006).
Artz, A., Butz, S. & Vestweber, D. GDF-15 inhibits integrin activation and mouse neutrophil recruitment through the ALK-5/TGF-βRII heterodimer. Blood 128, 529–541 (2016).
Liu, D.D., Lu, J.M., Zhao, Q.R., Hu, C. & Mei, Y.A. Growth differentiation factor-15 promotes glutamate release in medial prefrontal cortex of mice through upregulation of T-type calcium channels. Sci. Rep. 6, 28653 (2016).
Li, Z. et al. Identification, expression and functional characterization of the GRAL gene. J. Neurochem. 95, 361–376 (2005).
Fairlie, W.D. et al. Epitope mapping of the transforming growth factor-beta superfamily protein, macrophage inhibitory cytokine-1 (MIC-1): identification of at least five distinct epitope specificities. Biochemistry 40, 65–73 (2001).
Baloh, R.H., Enomoto, H., Johnson, E.M. Jr. & Milbrandt, J. The GDNF family ligands and receptors—implications for neural development. Curr. Opin. Neurobiol. 10, 103–110 (2000).
Airaksinen, M.S. & Saarma, M. The GDNF family: signalling, biological functions and therapeutic value. Nat. Rev. Neurosci. 3, 383–394 (2002).
Airaksinen, M.S., Titievsky, A. & Saarma, M. GDNF family neurotrophic factor signaling: four masters, one servant? Mol. Cell. Neurosci. 13, 313–325 (1999).
Leppänen, V.M. et al. The structure of GFRalpha1 domain 3 reveals new insights into GDNF binding and RET activation. EMBO J. 23, 1452–1462 (2004).
Scott, R.P. & Ibanez, C.F. Determinants of ligand binding specificity in the glial cell line-derived neurotrophic factor family receptor alpha S. J. Biol. Chem. 276, 1450–1458 (2001).
Treanor, J.J. et al. Characterization of a multicomponent receptor for GDNF. Nature 382, 80–83 (1996).
Mazagova, M. et al. Genetic deletion of growth differentiation factor 15 augments renal damage in both type 1 and type 2 models of diabetes. Am. J. Physiol. Renal Physiol. 305, F1249–F1264 (2013).
Heger, J. et al. Growth differentiation factor 15 acts anti-apoptotic and pro-hypertrophic in adult cardiomyocytes. J. Cell. Physiol. 224, 120–126 (2010).
Kempf, T. et al. The transforming growth factor-beta superfamily member growth-differentiation factor-15 protects the heart from ischemia/reperfusion injury. Circ. Res. 98, 351–360 (2006).
Park, H., Kim, C.H., Jeong, J.H., Park, M. & Kim, K.S. GDF15 contributes to radiation-induced senescence through the ROS-mediated p16 pathway in human endothelial cells. Oncotarget 7, 9634–9644 (2016).
Wong, A. et al. Phosphotyrosine 1062 is critical for the in vivo activity of the Ret9 receptor tyrosine kinase isoform. Mol. Cell. Biol. 25, 9661–9673 (2005).
Strelau, J. et al. Progressive postnatal motoneuron loss in mice lacking GDF-15. J. Neurosci. 29, 13640–13648 (2009).
Tsai, V.W. et al. TGF-b superfamily cytokine MIC-1/GDF15 is a physiological appetite and body weight regulator. PLoS One 8, e55174 (2013).
Mwangi, S.M. et al. Glial cell line-derived neurotrophic factor protects against high-fat diet-induced obesity. Am. J. Physiol. Gastrointest. Liver Physiol. 306, G515–G525 (2014).
Lebrun, B., Bariohay, B., Moyse, E. & Jean, A. Brain-derived neurotrophic factor (BDNF) and food intake regulation: a minireview. Auton. Neurosci. 126-127, 30–38 (2006).
Quartu, M. et al. Tissue distribution of Ret, GFRalpha-1, GFRalpha-2 and GFRalpha-3 receptors in the human brainstem at fetal, neonatal and adult age. Brain Res. 1173, 36–52 (2007).
Yang, C., Hutto, D. & Sah, D.W. Distribution of GDNF family receptor alpha3 and RET in rat and human non-neural tissues. J. Mol. Histol. 37, 69–77 (2006).
Norgren, R. & Smith, G.P. A method for selective section of vagal afferent or efferent axons in the rat. Am. J. Physiol. 267, R1136–R1141 (1994).
Moran, T.H., Baldessarini, A.R., Salorio, C.F., Lowery, T. & Schwartz, G.J. Vagal afferent and efferent contributions to the inhibition of food intake by cholecystokinin. Am. J. Physiol. 272, R1245–R1251 (1997).
Acknowledgements
The monoclonal MIC-1 antibody (MAB26) was a kind gift of S. Breit (St Vincent's Hospital, Sydney, Australia). J. Jelsing and N. Vrang (Gubra, Denmark) are acknowledged for their skillful conduction of in vivo study of rats using selective vagal deafferentiation, as well as data analysis. Z. Wang (NN Research Centre, China) is acknowledged for generating GFRAL and RET cotransfected cells. X. Sun, X. Yang, X. Liu, and J. Chen (NN Research Centre, China) are acknowledged for skillful conduction of in vitro studies. The authors would also like to thank K.E. Pedersen, H. Andersen, P.G. Mortensen (NN Global Research, Denmark), L. Wang, W. Wang, T. Li, W. Liu, Y. Du, J. Xu, D. Liu, and Y. Zhao (NN Research Centre, China) for their excellent technical support.
Author information
Authors and Affiliations
Contributions
L.Y.: library screening, flow cytometry and SPR binding assay, data collection and analysis, manuscript preparation; S.B.J.: project leadership, in vivo experiments, data collection and analysis, manuscript preparation); C.-C.C.: study supervision, manuscript preparation; Z.S.: library cell transfection; H.Z.: molecular cloning and protein expression; X.W.: Protein purification; T.H.: SPR kinetics assay; J.W.: library DNA preparation; Z.Y.: study supervision; H.C.: library DNA preparation; X.L.: bioinformatics analysis; B.S.: library-protein purification; Z.H.: cell-signaling assay, data analysis, manuscript preparation; X.G.: MIC-1 protein generation; J.S.: study supervision; W.Y.: study supervision; K.T.H.: project leadership, manuscript preparation; A.B.: qPCR tissue analysis, data collection and data analysis; J.B.F. and P.N.: expression and purification of biotinylated MIC-1 protein); K.H. and A.S.: ISH/IHC, manuscript preparation, data collection and analysis; L.M.J. and J.F.J.: indirect calorimetry, data collection and analysis; S.J.P.: laser-capture microscopy and NanoString-analysis data collection and analysis; S.B.P.: NN6K library sequence collection and analysis; D.M.: established data management and information-flow system for NN6k production and screening campaign.
Corresponding author
Ethics declarations
Competing interests
All authors are or have been working for Novo Nordisk A/S, a pharmaceutical company producing and selling medicine for the treatment of diabetes and obesity.
Supplementary information
Supplementary Figures and Tables
Supplementary Figures 1–7 and Supplementary Tables 1–2. (PDF 1086 kb)
Rights and permissions
About this article
Cite this article
Yang, L., Chang, CC., Sun, Z. et al. GFRAL is the receptor for GDF15 and is required for the anti-obesity effects of the ligand. Nat Med 23, 1158–1166 (2017). https://doi.org/10.1038/nm.4394
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.4394
This article is cited by
-
Artesunate treats obesity in male mice and non-human primates through GDF15/GFRAL signalling axis
Nature Communications (2024)
-
Genetic determinants of plasma protein levels in the Estonian population
Scientific Reports (2024)
-
Obesity and the kidney: mechanistic links and therapeutic advances
Nature Reviews Endocrinology (2024)
-
Whole genome sequencing of mouse lines divergently selected for fatness (FLI) and leanness (FHI) revealed several genetic variants as candidates for novel obesity genes
Genes & Genomics (2024)
-
Poly-Agonist Pharmacotherapies for Metabolic Diseases: Hopes and New Challenges
Drugs (2024)