Abstract
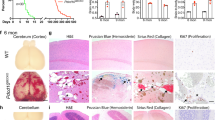
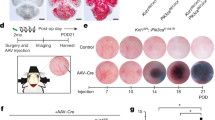
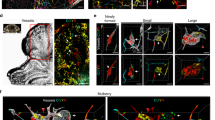
Cerebral cavernous malformations (CCMs) are vascular malformations that affect the central nervous system and result in cerebral hemorrhage, seizure and stroke. CCMs arise from loss-of-function mutations in one of three genes: KRIT1 (also known as CCM1), CCM2 or PDCD10 (also known as CCM3). PDCD10 mutations in humans often result in a more severe form of the disease relative to mutations in the other two CCM genes, and PDCD10-knockout mice show severe defects, the mechanistic basis for which is unclear. We have recently reported that CCM3 regulates exocytosis mediated by the UNC13 family of exocytic regulatory proteins. Here, in investigating the role of endothelial cell exocytosis in CCM disease progression, we found that CCM3 suppresses UNC13B- and vesicle-associated membrane protein 3 (VAMP3)-dependent exocytosis of angiopoietin 2 (ANGPT2) in brain endothelial cells. CCM3 deficiency in endothelial cells augments the exocytosis and secretion of ANGPT2, which is associated with destabilized endothelial cell junctions, enlarged lumen formation and endothelial cell–pericyte dissociation. UNC13B deficiency, which blunts ANGPT2 secretion from endothelial cells, or treatment with an ANGPT2-neutralizing antibody normalizes the defects in the brain and retina caused by endothelial-cell-specific CCM3 deficiency, including the disruption of endothelial cell junctions, vessel dilation and pericyte dissociation. Thus, enhanced secretion of ANGPT2 in endothelial cells contributes to the progression of CCM disease, providing a new therapeutic approach for treating this devastating pathology.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Change history
21 September 2016
In the version of this article initially published, labels for micrographs in Figure 2d and Figure 5b were omitted, and two grants were not acknowledged in the Acknowledgments section. The errors have been corrected in the HTML and PDF versions of the article.
References
Revencu, N. & Vikkula, M. Cerebral cavernous malformation: new molecular and clinical insights. J. Med. Genet. 43, 716–721 (2006).
Cavalcanti, D.D. et al. Cerebral cavernous malformations: from genes to proteins to disease. J. Neurosurg. 116, 122–132 (2012).
Tanriover, G. et al. Ultrastructural analysis of vascular features in cerebral cavernous malformations. Clin. Neurol. Neurosurg. 115, 438–444 (2013).
Riant, F., Bergametti, F., Ayrignac, X., Boulday, G. & Tournier-Lasserve, E. Recent insights into cerebral cavernous malformations: the molecular genetics of CCM. FEBS J. 277, 1070–1075 (2010).
Labauge, P., Denier, C., Bergametti, F. & Tournier-Lasserve, E. Genetics of cavernous angiomas. Lancet Neurol. 6, 237–244 (2007).
Sahoo, T. et al. Mutations in the gene encoding KRIT1, a Krev-1/rap1a binding protein, cause cerebral cavernous malformations (CCM1). Hum. Mol. Genet. 8, 2325–2333 (1999).
Liquori, C.L. et al. Mutations in a gene encoding a novel protein containing a phosphotyrosine-binding domain cause type 2 cerebral cavernous malformations. Am. J. Hum. Genet. 73, 1459–1464 (2003).
Bergametti, F. et al. Mutations within the programmed cell death 10 gene cause cerebral cavernous malformations. Am. J. Hum. Genet. 76, 42–51 (2005).
Gault, J., Shenkar, R., Recksiek, P. & Awad, I.A. Biallelic somatic and germ line CCM1 truncating mutations in a cerebral cavernous malformation lesion. Stroke 36, 872–874 (2005).
Akers, A.L., Johnson, E., Steinberg, G.K., Zabramski, J.M. & Marchuk, D.A. Biallelic somatic and germline mutations in cerebral cavernous malformations (CCMs): evidence for a two-hit mechanism of CCM pathogenesis. Hum. Mol. Genet. 18, 919–930 (2009).
Pagenstecher, A., Stahl, S., Sure, U. & Felbor, U. A two-hit mechanism causes cerebral cavernous malformations: complete inactivation of CCM1, CCM2 or CCM3 in affected endothelial cells. Hum. Mol. Genet. 18, 911–918 (2009).
McDonald, D.A. et al. A novel mouse model of cerebral cavernous malformations based on the two-hit mutation hypothesis recapitulates the human disease. Hum. Mol. Genet. 20, 211–222 (2011).
Chan, A.C. et al. Mutations in 2 distinct genetic pathways result in cerebral cavernous malformations in mice. J. Clin. Invest. 121, 1871–1881 (2011).
Cunningham, K. et al. Conditional deletion of Ccm2 causes hemorrhage in the adult brain: a mouse model of human cerebral cavernous malformations. Hum. Mol. Genet. 20, 3198–3206 (2011).
Boulday, G. et al. Tissue-specific conditional CCM2 knockout mice establish the essential role of endothelial CCM2 in angiogenesis: implications for human cerebral cavernous malformations. Dis. Model. Mech. 2, 168–177 (2009).
Maddaluno, L. et al. EndMT contributes to the onset and progression of cerebral cavernous malformations. Nature 498, 492–496 (2013).
Shenkar, R. et al. Exceptional aggressiveness of cerebral cavernous malformation disease associated with PDCD10 mutations. Genet. Med. 17, 188–196 (2015).
Whitehead, K.J. et al. The cerebral cavernous malformation signaling pathway promotes vascular integrity via Rho GTPases. Nat. Med. 15, 177–184 (2009).
Stockton, R.A., Shenkar, R., Awad, I.A. & Ginsberg, M.H. Cerebral cavernous malformations proteins inhibit Rho kinase to stabilize vascular integrity. J. Exp. Med. 207, 881–896 (2010).
Zhou, Z. et al. Cerebral cavernous malformations arise from endothelial gain of MEKK3–KLF2/4 signalling. Nature 532, 122–126 (2016).
Denier, C. et al. Genotype-phenotype correlations in cerebral cavernous malformations patients. Ann. Neurol. 60, 550–556 (2006).
Zheng, X. et al. CCM3 signaling through sterile 20-like kinases plays an essential role during zebrafish cardiovascular development and cerebral cavernous malformations. J. Clin. Invest. 120, 2795–2804 (2010).
Yoruk, B., Gillers, B.S., Chi, N.C. & Scott, I.C. Ccm3 functions in a manner distinct from Ccm1 and Ccm2 in a zebrafish model of CCM vascular disease. Dev. Biol. 362, 121–131 (2012).
Zhang, Y. et al. A network of interactions enables CCM3 and STK24 to coordinate UNC13D-driven vesicle exocytosis in neutrophils. Dev. Cell 27, 215–226 (2013).
Feldmann, J. et al. Munc13-4 is essential for cytolytic granules fusion and is mutated in a form of familial hemophagocytic lymphohistiocytosis (FHL3). Cell 115, 461–473 (2003).
Jahn, R. & Südhof, T.C. Membrane fusion and exocytosis. Annu. Rev. Biochem. 68, 863–911 (1999).
Lowenstein, C.J., Morrell, C.N. & Yamakuchi, M. Regulation of Weibel-Palade body exocytosis. Trends Cardiovasc. Med. 15, 302–308 (2005).
Fiedler, U. et al. Angiopoietin-2 sensitizes endothelial cells to TNF-α and has a crucial role in the induction of inflammation. Nat. Med. 12, 235–239 (2006).
Gaengel, K., Genové, G., Armulik, A. & Betsholtz, C. Endothelial-mural cell signaling in vascular development and angiogenesis. Arterioscler. Thromb. Vasc. Biol. 29, 630–638 (2009).
Eklund, L. & Olsen, B.R. Tie receptors and their angiopoietin ligands are context-dependent regulators of vascular remodeling. Exp. Cell Res. 312, 630–641 (2006).
Maisonpierre, P.C. et al. Angiopoietin-2, a natural antagonist for Tie2 that disrupts in vivo angiogenesis. Science 277, 55–60 (1997).
He, Y. et al. Stabilization of VEGFR2 signaling by cerebral cavernous malformation 3 is critical for vascular development. Sci. Signal. 3, ra26 (2010).
Fidalgo, M. et al. CCM3/PDCD10 stabilizes GCKIII proteins to promote Golgi assembly and cell orientation. J. Cell Sci. 123, 1274–1284 (2010).
Zhou, H.J. et al. AIP1 mediates vascular endothelial cell growth factor receptor-3-dependent angiogenic and lymphangiogenic responses. Arterioscler. Thromb. Vasc. Biol. 34, 603–615 (2014).
Kluger, M.S., Clark, P.R., Tellides, G., Gerke, V. & Pober, J.S. Claudin-5 controls intercellular barriers of human dermal microvascular but not human umbilical vein endothelial cells. Arterioscler. Thromb. Vasc. Biol. 33, 489–500 (2013).
Nakatsu, M.N. & Hughes, C.C. An optimized three-dimensional in vitro model for the analysis of angiogenesis. Methods Enzymol. 443, 65–82 (2008).
Abraham, S. et al. A Rac/Cdc42 exchange factor complex promotes formation of lateral filopodia and blood vessel lumen morphogenesis. Nat. Commun. 6, 7286 (2015).
Felcht, M. et al. Angiopoietin-2 differentially regulates angiogenesis through TIE2 and integrin signaling. J. Clin. Invest. 122, 1991–2005 (2012).
Chang, W.G., Andrejecsk, J.W., Kluger, M.S., Saltzman, W.M. & Pober, J.S. Pericytes modulate endothelial sprouting. Cardiovasc. Res. 100, 492–500 (2013).
Varoqueaux, F. et al. Total arrest of spontaneous and evoked synaptic transmission but normal synaptogenesis in the absence of Munc13-mediated vesicle priming. Proc. Natl. Acad. Sci. USA 99, 9037–9042 (2002).
Holopainen, T. et al. Effects of angiopoietin-2-blocking antibody on endothelial cell-cell junctions and lung metastasis. J. Natl. Cancer Inst. 104, 461–475 (2012).
Gale, N.W. et al. Angiopoietin-2 is required for postnatal angiogenesis and lymphatic patterning, and only the latter role is rescued by Angiopoietin-1. Dev. Cell 3, 411–423 (2002).
Thomson, B.R. et al. A lymphatic defect causes ocular hypertension and glaucoma in mice. J. Clin. Invest. 124, 4320–4324 (2014).
Saharinen, P. et al. Angiopoietins assemble distinct Tie2 signalling complexes in endothelial cell-cell and cell-matrix contacts. Nat. Cell Biol. 10, 527–537 (2008).
Fukuhara, S. et al. Differential function of Tie2 at cell-cell contacts and cell-substratum contacts regulated by angiopoietin-1. Nat. Cell Biol. 10, 513–526 (2008).
Daly, C. et al. Angiopoietin-2 functions as an autocrine protective factor in stressed endothelial cells. Proc. Natl. Acad. Sci. USA 103, 15491–15496 (2006).
Marchi, S. et al. Defective autophagy is a key feature of cerebral cavernous malformations. EMBO Mol. Med. 7, 1403–1417 (2015).
Kümpers, P. et al. Time course of angiopoietin-2 release during experimental human endotoxemia and sepsis. Crit. Care 13, R64 (2009).
Gingras, A.R., Liu, J.J. & Ginsberg, M.H. Structural basis of the junctional anchorage of the cerebral cavernous malformations complex. J. Cell Biol. 199, 39–48 (2012).
Cuttano, R. et al. KLF4 is a key determinant in the development and progression of cerebral cavernous malformations. EMBO Mol. Med. 8, 6–24 (2015).
Sako, K. et al. Angiopoietin-1 induces Kruppel-like factor 2 expression through a phosphoinositide 3-kinase/AKT-dependent activation of myocyte enhancer factor 2. J. Biol. Chem. 284, 5592–5601 (2009).
Zawistowski, J.S. et al. CCM1 and CCM2 protein interactions in cell signaling: implications for cerebral cavernous malformations pathogenesis. Hum. Mol. Genet. 14, 2521–2531 (2005).
Pouwels, J., Nevo, J., Pellinen, T., Ylänne, J. & Ivaska, J. Negative regulators of integrin activity. J. Cell Sci. 125, 3271–3280 (2012).
Brütsch, R. et al. Integrin cytoplasmic domain-associated protein-1 attenuates sprouting angiogenesis. Circ. Res. 107, 592–601 (2010).
Lampugnani, M.G. et al. CCM1 regulates vascular-lumen organization by inducing endothelial polarity. J. Cell Sci. 123, 1073–1080 (2010).
Carmeliet, P. et al. Targeted deficiency or cytosolic truncation of the VE-cadherin gene in mice impairs VEGF-mediated endothelial survival and angiogenesis. Cell 98, 147–157 (1999).
Gory-Fauré, S. et al. Role of vascular endothelial-cadherin in vascular morphogenesis. Development 126, 2093–2102 (1999).
Sigurbjörnsdóttir, S., Mathew, R. & Leptin, M. Molecular mechanisms of de novo lumen formation. Nat. Rev. Mol. Cell Biol. 15, 665–676 (2014).
Song, Y., Eng, M. & Ghabrial, A.S. Focal defects in single-celled tubes mutant for cerebral cavernous malformation 3, GCKIII, or NSF2. Dev. Cell 25, 507–519 (2013).
Li, S. et al. Retro-orbital injection of FITC-dextran is an effective and economical method for observing mouse retinal vessels. Mol. Vis. 17, 3566–3573 (2011).
Acknowledgements
Unc13B-deficient mice were a gift from N. Brose (Max Planck Institute of Experimental Medicine, Germany). We thank J. Pober, R. Liu and T. Manes for reagents and discussion. This work was partly supported by US National Institutes of Health (NIH) grants R01 HL109420 (W.M.), HL115148 (W.M.), GM109487 (D.W.), National Natural Science Foundation of China (no. 91539110) (W.M.), CT Stem Cell Innovation (Established Investigator Grant) award no. 14-SCB-YALE-17 (W.M.) and 2016YFC1300600 (W.M.), Scientific Grants of Guangdong (no. 2015B020225002 and 2015A050502018) (W.M.), and American Heart Association grants 13SDG17110045 (H.Z.) and 14SDG20490020 (W.T.).
Author information
Authors and Affiliations
Contributions
H.J.Z., L.Q., H.Z. and W.M. conceived the study, designed experiments and wrote the manuscript; H.J.Z., L.Q., H.Z., W.T., W.J., Y.H., Z.W., Q.Y. and M.S.K. performed experiments; W.J., G.F. and M.Y. generated the anti-angiopoietin-2 antibodies; X.L. interpreted retinal data; A.V. provided the human CCM blocks; and H.J.Z., L.Q., H.Z., D.T., D.W. and W.M. interpreted data. M.S.K. edited the manuscript.
Corresponding authors
Ethics declarations
Competing interests
G.F. and M.Y. are employees of Genentech. W.J. is an employee of Guangzhou Darron Medscience.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–8 (PDF 4939 kb)
Rights and permissions
About this article
Cite this article
Zhou, H., Qin, L., Zhang, H. et al. Endothelial exocytosis of angiopoietin-2 resulting from CCM3 deficiency contributes to cerebral cavernous malformation. Nat Med 22, 1033–1042 (2016). https://doi.org/10.1038/nm.4169
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.4169
This article is cited by
-
Comparative transcriptome findings reveal the neuroinflammatory network and potential biomarkers to early detection of ischemic stroke
Journal of Biological Engineering (2023)
-
Mitochondrial dysfunction induces ALK5-SMAD2-mediated hypovascularization and arteriovenous malformations in mouse retinas
Nature Communications (2022)
-
ATPIF1 maintains normal mitochondrial structure which is impaired by CCM3 deficiency in endothelial cells
Cell & Bioscience (2021)
-
Imaging and optogenetic modulation of vascular mural cells in the live brain
Nature Protocols (2021)
-
Caveolae-mediated Tie2 signaling contributes to CCM pathogenesis in a brain endothelial cell-specific Pdcd10-deficient mouse model
Nature Communications (2021)