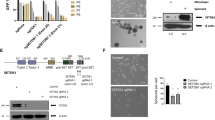
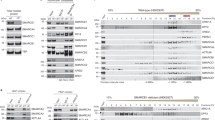
Abstract
Human cancer genome sequencing has recently revealed that genes that encode subunits of SWI/SNF chromatin remodeling complexes are frequently mutated across a wide variety of cancers, and several subunits of the complex have been shown to have bona fide tumor suppressor activity1. However, whether mutations in SWI/SNF subunits result in shared dependencies is unknown. Here we show that EZH2, a catalytic subunit of the polycomb repressive complex 2 (PRC2), is essential in all tested cancer cell lines and xenografts harboring mutations of the SWI/SNF subunits ARID1A, PBRM1, and SMARCA4, which are several of the most frequently mutated SWI/SNF subunits in human cancer, but that co-occurrence of a Ras pathway mutation is correlated with abrogation of this dependence. Notably, we demonstrate that SWI/SNF-mutant cancer cells are primarily dependent on a non-catalytic role of EZH2 in the stabilization of the PRC2 complex, and that they are only partially dependent on EZH2 histone methyltransferase activity. These results not only reveal a shared dependency of cancers with genetic alterations in SWI/SNF subunits, but also suggest that EZH2 enzymatic inhibitors now in clinical development may not fully suppress the oncogenic activity of EZH2.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Kadoch, C. et al. Proteomic and bioinformatic analysis of mammalian SWI/SNF complexes identifies extensive roles in human malignancy. Nat. Genet. 45, 592–601 (2013).
Wu, J.I., Lessard, J. & Crabtree, G.R. Understanding the words of chromatin regulation. Cell 136, 200–206 (2009).
Wilson, B.G. & Roberts, C.W. SWI/SNF nucleosome remodellers and cancer. Nat. Rev. Cancer 11, 481–492 (2011).
Weissman, B. & Knudsen, K.E. Hijacking the chromatin remodeling machinery: impact of SWI/SNF perturbations in cancer. Cancer Res. 69, 8223–8230 (2009).
Guan, B. et al. Roles of deletion of Arid1a, a tumor suppressor, in mouse ovarian tumorigenesis. J. Natl. Cancer Inst. 106, dju46 (2014).
Bultman, S.J. et al. Characterization of mammary tumors from Brg1 heterozygous mice. Oncogene 27, 460–468 (2008).
Roberts, C.W., Leroux, M.M., Fleming, M.D. & Orkin, S.H. Highly penetrant, rapid tumorigenesis through conditional inversion of the tumor suppressor gene Snf5. Cancer Cell 2, 415–425 (2002).
Jones, S. et al. Frequent mutations of chromatin remodeling gene ARID1A in ovarian clear cell carcinoma. Science 330, 228–231 (2010).
Wiegand, K.C. et al. ARID1A mutations in endometriosis-associated ovarian carcinomas. N. Engl. J. Med. 363, 1532–1543 (2010).
Fujimoto, A. et al. Whole-genome sequencing of liver cancers identifies etiological influences on mutation patterns and recurrent mutations in chromatin regulators. Nat. Genet. 44, 760–764 (2012).
Li, M. et al. Inactivating mutations of the chromatin remodeling gene ARID2 in hepatocellular carcinoma. Nat. Genet. 43, 828–829 (2011).
Gui, Y. et al. Frequent mutations of chromatin remodeling genes in transitional cell carcinoma of the bladder. Nat. Genet. 43, 875–878 (2011).
Varela, I. et al. Exome sequencing identifies frequent mutation of the SWI/SNF complex gene PBRM1 in renal carcinoma. Nature 469, 539–542 (2011).
Parsons, D.W. et al. The genetic landscape of the childhood cancer medulloblastoma. Science 331, 435–439 (2011).
Robinson, G. et al. Novel mutations target distinct subgroups of medulloblastoma. Nature 488, 43–48 (2012).
Sausen, M. et al. Integrated genomic analyses identify ARID1A and ARID1B alterations in the childhood cancer neuroblastoma. Nat. Genet. 45, 12–17 (2013).
Shain, A.H. et al. Convergent structural alterations define SWItch/Sucrose NonFermentable (SWI/SNF) chromatin remodeler as a central tumor suppressive complex in pancreatic cancer. Proc. Natl. Acad. Sci. USA 109, E252–E259 (2012).
Le Gallo, M. et al. Exome sequencing of serous endometrial tumors identifies recurrent somatic mutations in chromatin-remodeling and ubiquitin ligase complex genes. Nat. Genet. 44, 1310–1315 (2012).
Kennison, J.A. The Polycomb and trithorax group proteins of Drosophila: trans-regulators of homeotic gene function. Annu. Rev. Genet. 29, 289–303 (1995).
Shao, Z. et al. Stabilization of chromatin structure by PRC1, a Polycomb complex. Cell 98, 37–46 (1999).
Tamkun, J.W. et al. brahma: a regulator of Drosophila homeotic genes structurally related to the yeast transcriptional activator SNF2/SWI2. Cell 68, 561–572 (1992).
Ringrose, L. & Paro, R. Epigenetic regulation of cellular memory by the Polycomb and Trithorax group proteins. Annu. Rev. Genet. 38, 413–443 (2004).
Pasini, D., Bracken, A.P., Hansen, J.B., Capillo, M. & Helin, K. The polycomb group protein Suz12 is required for embryonic stem cell differentiation. Mol. Cell. Biol. 27, 3769–3779 (2007).
Cao, R. & Zhang, Y. SUZ12 is required for both the histone methyltransferase activity and the silencing function of the EED-EZH2 complex. Mol. Cell 15, 57–67 (2004).
Margueron, R. et al. Role of the polycomb protein EED in the propagation of repressive histone marks. Nature 461, 762–767 (2009).
Qi, W. et al. Selective inhibition of Ezh2 by a small-molecule inhibitor blocks tumor cells proliferation. Proc. Natl. Acad. Sci. USA 109, 21360–21365 (2012).
McCabe, M.T. et al. EZH2 inhibition as a therapeutic strategy for lymphoma with EZH2-activating mutations. Nature 492, 108–112 (2012).
McCabe, M.T. et al. Mutation of A677 in histone methyltransferase EZH2 in human B-cell lymphoma promotes hypertrimethylation of histone H3 on lysine 27 (H3K27). Proc. Natl. Acad. Sci. USA 109, 2989–2994 (2012).
Knutson, S.K. et al. A selective inhibitor of EZH2 blocks H3K27 methylation and kills mutant lymphoma cells. Nat. Chem. Biol. 8, 890–896 (2012).
Kia, S.K., Gorski, M.M., Giannakopoulos, S. & Verrijzer, C.P. SWI/SNF mediates polycomb eviction and epigenetic reprogramming of the INK4b-ARF-INK4a locus. Mol. Cell. Biol. 28, 3457–3464 (2008).
Knutson, S.K. et al. Durable tumor regression in genetically altered malignant rhabdoid tumors by inhibition of methyltransferase EZH2. Proc. Natl. Acad. Sci. USA 110, 7922–7927 (2013).
Wilson, B.G. et al. Epigenetic antagonism between polycomb and SWI/SNF complexes during oncogenic transformation. Cancer Cell 18, 316–328 (2010).
Medina, P.P. et al. Frequent BRG1/SMARCA4-inactivating mutations in human lung cancer cell lines. Hum. Mutat. 29, 617–622 (2008).
Cheung, H.W. et al. Systematic investigation of genetic vulnerabilities across cancer cell lines reveals lineage-specific dependencies in ovarian cancer. Proc. Natl. Acad. Sci. USA 108, 12372–12377 (2011).
Shao, D.D. et al. ATARiS: computational quantification of gene suppression phenotypes from multisample RNAi screens. Genome Res. 23, 665–678 (2013).
Cowley, G.S. et al. Parallel genome-scale loss of function screens in 216 cancer cell lines for the identification of context-specific genetic dependencies. Sci. Data 1, 140035 (2014).
MacConaill, L.E. et al. Profiling critical cancer gene mutations in clinical tumor samples. PLoS ONE 4, e7887 (2009).
De Raedt, T. et al. PRC2 loss amplifies Ras-driven transcription and confers sensitivity to BRD4-based therapies. Nature 514, 247–251 (2014).
Kim, W. et al. Targeted disruption of the EZH2-EED complex inhibits EZH2-dependent cancer. Nat. Chem. Biol. 9, 643–650 (2013).
Wei, Y. et al. CDK1-dependent phosphorylation of EZH2 suppresses methylation of H3K27 and promotes osteogenic differentiation of human mesenchymal stem cells. Nat. Cell Biol. 13, 87–94 (2011).
Xu, K. et al. EZH2 oncogenic activity in castration-resistant prostate cancer cells is Polycomb-independent. Science. 338, 1465–1469 (2012).
Acknowledgements
EZ005 was provided courtesy of J. Bradner (Dana-Farber Cancer Institute). Plasmids for wild-type and SET domain–truncated (SET) EZH2 were provided courtesy of M. Brown (Dana-Farber Cancer Institute). K.H.K. was supported by an award from National Cancer Center. This work was supported by US National Institutes of Health grants R01CA172152 (C.W.M.R.), R01CA113794 (C.W.M.R.) and U01CA176058 (W.C.H.). W.K. was supported by a Claudia Adams Barr grant. T.P.H. was supported by an award from the National Institute of General Medical Sciences (T32GM007753). The Cure AT/RT Now foundation, the Avalanna Fund, the Garrett B. Smith Foundation, Miles for Mary (C.W.M.R.), a Leukemia & Lymphoma Society Specialized Center of Research Award Project Grant (L.D.W.) and the Todd J. Schwartz Memorial Fund (L.D.W.) provided additional support.
Author information
Authors and Affiliations
Contributions
K.H.K. and C.W.M.R. designed the study; K.H.K., W.W. and J.R.H. performed the experiments with the help of J.N.W.; W.K., S.H.O. and L.D.W. provided stapled peptides and contributed to data analysis; T.P.H., W.C.H., F.V. and A.T. analyzed Achilles data; K.H.K. and C.W.M.R. wrote the manuscript with comments from all authors.
Corresponding author
Ethics declarations
Competing interests
L.D.W. is a scientific advisory board member and consultant for Aileron Therapeutics. Via the Dana-Farber Cancer Institute Novartis Drug Discovery Program, C.W.M.R. and W.C.H. receive research support and consulting fees from the Novartis Institutes for Biomedical Research.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–5 & Supplementary Table 1 (PDF 1940 kb)
Supplementary Data 1
Project Achilles data and mutation annotations (XLSX 39 kb)
Supplementary Data 2
Mutations in cell lines used in this study (XLSX 47 kb)
Rights and permissions
About this article
Cite this article
Kim, K., Kim, W., Howard, T. et al. SWI/SNF-mutant cancers depend on catalytic and non-catalytic activity of EZH2. Nat Med 21, 1491–1496 (2015). https://doi.org/10.1038/nm.3968
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.3968
This article is cited by
-
CUL4B-DDB1-COP1-mediated UTX downregulation promotes colorectal cancer progression
Experimental Hematology & Oncology (2023)
-
Context-defined cancer co-dependency mapping identifies a functional interplay between PRC2 and MLL-MEN1 complex in lymphoma
Nature Communications (2023)
-
Polycomb deficiency drives a FOXP2-high aggressive state targetable by epigenetic inhibitors
Nature Communications (2023)
-
UHRF1/UBE2L6/UBR4-mediated ubiquitination regulates EZH2 abundance and thereby melanocytic differentiation phenotypes in melanoma
Oncogene (2023)
-
Methylation across the central dogma in health and diseases: new therapeutic strategies
Signal Transduction and Targeted Therapy (2023)