Abstract
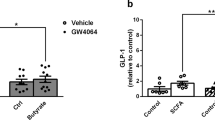
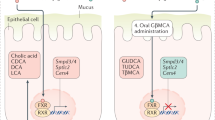
The systemic expression of the bile acid (BA) sensor farnesoid X receptor (FXR) has led to promising new therapies targeting cholesterol metabolism, triglyceride production, hepatic steatosis and biliary cholestasis. In contrast to systemic therapy, bile acid release during a meal selectively activates intestinal FXR. By mimicking this tissue-selective effect, the gut-restricted FXR agonist fexaramine (Fex) robustly induces enteric fibroblast growth factor 15 (FGF15), leading to alterations in BA composition, but does so without activating FXR target genes in the liver. However, unlike systemic agonism, we find that Fex reduces diet-induced weight gain, body-wide inflammation and hepatic glucose production, while enhancing thermogenesis and browning of white adipose tissue (WAT). These pronounced metabolic improvements suggest tissue-restricted FXR activation as a new approach in the treatment of obesity and metabolic syndrome.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Accession codes
References
Forman, B.M. et al. Identification of a nuclear receptor that is activated by farnesol metabolites. Cell 81, 687–693 (1995).
Lee, F.Y., Lee, H., Hubbert, M.L., Edwards, P.A. & Zhang, Y. FXR, a multipurpose nuclear receptor. Trends Biochem. Sci. 31, 572–580 (2006).
Repa, J.J. et al. Regulation of absorption and ABC1-mediated efflux of cholesterol by RXR heterodimers. Science 289, 1524–1529 (2000).
Zollner, G. et al. Role of nuclear bile acid receptor, FXR, in adaptive ABC transporter regulation by cholic and ursodeoxycholic acid in mouse liver, kidney and intestine. J. Hepatol. 39, 480–488 (2003).
Fang, S. et al. The p300 acetylase is critical for ligand-activated farnesoid X receptor (FXR) induction of SHP. J. Biol. Chem. 283, 35086–35095 (2008).
Kemper, J.K. et al. FXR acetylation is normally dynamically regulated by p300 and SIRT1 but constitutively elevated in metabolic disease states. Cell Metab. 10, 392–404 (2009).
Makishima, M. et al. Identification of a nuclear receptor for bile acids. Science 284, 1362–1365 (1999).
Stedman, C. et al. Benefit of farnesoid X receptor inhibition in obstructive cholestasis. Proc. Natl. Acad. Sci. USA 103, 11323–11328 (2006).
Sinal, C.J. et al. Targeted disruption of the nuclear receptor FXR/BAR impairs bile acid and lipid homeostasis. Cell 102, 731–744 (2000).
Prawitt, J. et al. Farnesoid X receptor deficiency improves glucose homeostasis in mouse models of obesity. Diabetes 60, 1861–1871 (2011).
Zhang, Y. et al. Activation of the nuclear receptor FXR improves hyperglycemia and hyperlipidemia in diabetic mice. Proc. Natl. Acad. Sci. USA 103, 1006–1011 (2006).
Watanabe, M. et al. Lowering bile acid pool size with a synthetic farnesoid X receptor (FXR) agonist induces obesity and diabetes through reduced energy expenditure. J. Biol. Chem. 286, 26913–26920 (2011).
Wang, L. et al. Redundant pathways for negative feedback regulation of bile acid production. Dev. Cell 2, 721–731 (2002).
Fang, S. et al. Coordinated recruitment of histone methyltransferase G9a and other chromatin-modifying enzymes in SHP-mediated regulation of hepatic bile acid metabolism. Mol. Cell. Biol. 27, 1407–1424 (2007).
Lu, T.T. et al. Molecular basis for feedback regulation of bile acid synthesis by nuclear receptors. Mol. Cell 6, 507–515 (2000).
Huang, W. et al. Nuclear receptor-dependent bile acid signaling is required for normal liver regeneration. Science 312, 233–236 (2006).
Zhang, Y., Castellani, L.W., Sinal, C.J., Gonzalez, F.J. & Edwards, P.A. Peroxisome proliferator-activated receptor-γ coactivator 1α (PGC-1α) regulates triglyceride metabolism by activation of the nuclear receptor FXR. Genes Dev. 18, 157–169 (2004).
Ma, K., Saha, P.K., Chan, L. & Moore, D.D. Farnesoid X receptor is essential for normal glucose homeostasis. J. Clin. Invest. 116, 1102–1109 (2006).
Stanimirov, B., Stankov, K. & Mikov, M. Pleiotropic functions of bile acids mediated by the farnesoid X receptor. Acta Gastroenterol. Belg. 75, 389–398 (2012).
Mudaliar, S. et al. Efficacy and safety of the farnesoid X receptor agonist obeticholic acid in patients with type 2 diabetes and nonalcoholic fatty liver disease. Gastroenterology 145, 574–582 e571 (2013).
Kim, I. et al. Differential regulation of bile acid homeostasis by the farnesoid X receptor in liver and intestine. J. Lipid Res. 48, 2664–2672 (2007).
Song, K.H., Li, T., Owsley, E., Strom, S. & Chiang, J.Y. Bile acids activate fibroblast growth factor 19 signaling in human hepatocytes to inhibit cholesterol 7α-hydroxylase gene expression. Hepatology 49, 297–305 (2009).
Inagaki, T. et al. Fibroblast growth factor 15 functions as an enterohepatic signal to regulate bile acid homeostasis. Cell Metab. 2, 217–225 (2005).
Li, F. et al. Microbiome remodelling leads to inhibition of intestinal farnesoid X receptor signalling and decreased obesity. Nat. Commun. 4, 2384 (2013).
Inagaki, T. et al. Regulation of antibacterial defense in the small intestine by the nuclear bile acid receptor. Proc. Natl. Acad. Sci. USA 103, 3920–3925 (2006).
Downes, M. et al. A chemical, genetic, and structural analysis of the nuclear bile acid receptor FXR. Mol. Cell 11, 1079–1092 (2003).
Cao, W. et al. p38 mitogen-activated protein kinase is the central regulator of cyclic AMP-dependent transcription of the brown fat uncoupling protein 1 gene. Mol. Cell. Biol. 24, 3057–3067 (2004).
Thomas, A.M. et al. Genome-wide tissue-specific farnesoid X receptor binding in mouse liver and intestine. Hepatology 51, 1410–1419 (2010).
Fu, L. et al. Fibroblast growth factor 19 increases metabolic rate and reverses dietary and leptin-deficient diabetes. Endocrinology 145, 2594–2603 (2004).
Stroeve, J.H. et al. Intestinal FXR-mediated FGF15 production contributes to diurnal control of hepatic bile acid synthesis in mice. Lab. Invest. 90, 1457–1467 (2010).
Wu, A.L. et al. FGF19 regulates cell proliferation, glucose and bile acid metabolism via FGFR4-dependent and independent pathways. PLoS ONE 6, e17868 (2011).
Gadaleta, R.M. et al. Farnesoid X receptor activation inhibits inflammation and preserves the intestinal barrier in inflammatory bowel disease. Gut 60, 463–472 (2011).
Ullmer, C. et al. Systemic bile acid sensing by G protein–coupled bile acid receptor 1 (GPBAR1) promotes PYY and GLP-1 release. Br. J. Pharmacol. 169, 671–684 (2013).
Reilly, S.M. et al. An inhibitor of the protein kinases TBK1 and IKK-varepsilon improves obesity-related metabolic dysfunctions in mice. Nat. Med. 19, 313–321 (2013).
Carmen, G.Y. & Victor, S.M. Signalling mechanisms regulating lipolysis. Cell. Signal. 18, 401–408 (2006).
Song, Y. et al. CRTC3 links catecholamine signalling to energy balance. Nature 468, 933–939 (2010).
Tsukiyama-Kohara, K. et al. Adipose tissue reduction in mice lacking the translational inhibitor 4E–BP1. Nat. Med. 7, 1128–1132 (2001).
Fisher, F.M. et al. FGF21 regulates PGC-1α and browning of white adipose tissues in adaptive thermogenesis. Genes Dev. 26, 271–281 (2012).
Hansen, J.B. et al. Retinoblastoma protein functions as a molecular switch determining white versus brown adipocyte differentiation. Proc. Natl. Acad. Sci. USA 101, 4112–4117 (2004).
Wang, H. et al. Liver X receptor α is a transcriptional repressor of the uncoupling protein 1 gene and the brown fat phenotype. Mol. Cell. Biol. 28, 2187–2200 (2008).
Cohen, J.C., Horton, J.D. & Hobbs, H.H. Human fatty liver disease: old questions and new insights. Science 332, 1519–1523 (2011).
Bhatnagar, S., Damron, H.A. & Hillgartner, F.B. Fibroblast growth factor-19, a novel factor that inhibits hepatic fatty acid synthesis. J. Biol. Chem. 284, 10023–10033 (2009).
Foster-Schubert, K.E. & Cummings, D.E. Emerging therapeutic strategies for obesity. Endocr. Rev. 27, 779–793 (2006).
Watanabe, M. et al. Bile acids induce energy expenditure by promoting intracellular thyroid hormone activation. Nature 439, 484–489 (2006).
Ryan, K.K. et al. FXR is a molecular target for the effects of vertical sleeve gastrectomy. Nature 509, 183–188 (2014).
Dobin, A. et al. STAR: ultrafast universal RNA-seq aligner. Bioinformatics 29, 15–21 (2013).
Trapnell, C. et al. Differential analysis of gene regulation at transcript resolution with RNA-seq. Nat. Biotechnol. 31, 46–53 (2013).
Roberts, A., Pimentel, H., Trapnell, C. & Pachter, L. Identification of novel transcripts in annotated genomes using RNA-seq. Bioinformatics 27, 2325–2329 (2011).
Vergnes, L., Lee, J.M., Chin, R.G., Auwerx, J. & Reue, K. Diet1 functions in the FGF15/19 enterohepatic signaling axis to modulate bile acid and lipid levels. Cell Metab. 17, 916–928 (2013).
Osborn, O. et al. G protein–coupled receptor 21 deletion improves insulin sensitivity in diet-induced obese mice. J. Clin. Invest. 122, 2444–2453 (2012).
Acknowledgements
We thank H. Juguilon, L. Chong, Y. Yin, J. Alvarez, Y. Dai, S. Kaufman and B. Collins for technical assistance, and L. Ong and C. Brondos for administrative assistance. R.M.E. is an Investigator of the Howard Hughes Medical Institute (HHMI) at the Salk Institute and March of Dimes Chair in Molecular and Developmental Biology, and is supported by National Institutes of Health (NIH) grants (DK057978, DK090962, HL088093, HL105278 and ES010337), the Glenn Foundation for Medical Research, the Leona M. and Harry B. Helmsley Charitable Trust, Ipsen/Biomeasure, California Institute for Regenerative Medicine and The Ellison Medical Foundation. C.L. and M.D. are funded by grants from the National Health and Medical Research Council of Australia Project Grants 512354, 632886 and 1043199; J.M.O. is supported by NIH grants (DK033651, DK074868, T32-DK007494, DK063491 and P01-DK054441-14A1) and by the Eunice Kennedy Shriver National Institute of Child Health and Human Development/NIH through cooperative agreement of U54-HD-012303-25 as part of the specialized Cooperative Centers Program in Reproduction and Infertility Research; A.R.S. is supported by NIH grants (DK60597 and DK61618). R.M.E., J.M.O., A.R.S. and D.A.B. are supported by NIH grant R24DK090962.
Author information
Authors and Affiliations
Contributions
S.F., J.M.S., M.D. and R.M.E. designed and supervised the research. S.F., J.M.S., S.M.R., E. Yoshihara, O.O., D.L., E. Yu, S.J., S.C. and Y.L. performed research. K.S., A.K., and A.P. performed research and analyzed data. S.F., J.M.S., S.M.R., E. Yoshihara, O.O., D.L., E. Yu, R.T.Y., S.C., C.L., A.R.A., B.S., D.A.B., J.M.O., A.R.S., M.D. and R.M.E. analyzed data. S.F., J.M.S., A.R.A., B.S., D.A.B., C.L., J.M.O., A.R.S., M.D. and R.M.E. wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
S.F., J.M.S., A.R.A., A.R.S., M.D. and R.M.E. are co-inventors of FXR molecules and methods of use and may be entitled to royalties.
Supplementary information
Supplementary Figures and Table
Supplementary Figures 1–14 and Supplementary Table 1 (PDF 4256 kb)
Rights and permissions
About this article
Cite this article
Fang, S., Suh, J., Reilly, S. et al. Intestinal FXR agonism promotes adipose tissue browning and reduces obesity and insulin resistance. Nat Med 21, 159–165 (2015). https://doi.org/10.1038/nm.3760
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.3760
This article is cited by
-
Pharmacological modulation of adaptive thermogenesis: new clues for obesity management?
Journal of Endocrinological Investigation (2023)
-
An update on the secretory functions of brown, white, and beige adipose tissue: Towards therapeutic applications
Reviews in Endocrine and Metabolic Disorders (2023)
-
The transcription factor hepatocyte nuclear factor 4A acts in the intestine to promote white adipose tissue energy storage
Nature Communications (2022)
-
Bile acid and receptors: biology and drug discovery for nonalcoholic fatty liver disease
Acta Pharmacologica Sinica (2022)
-
Metabolic Messengers: bile acids
Nature Metabolism (2022)