Abstract
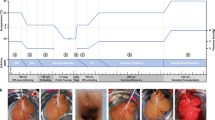
The realization of long-term human organ preservation will have groundbreaking effects on the current practice of transplantation. Herein we present a new technique based on subzero nonfreezing preservation and extracorporeal machine perfusion that allows transplantation of rat livers preserved for up to four days, thereby tripling the viable preservation duration.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
US Department of Health & Human Services Health Resources and Services Administration. Organ Procurement and Transplantation Network (OPTN), http://optn.transplant.hrsa.gov/ (2014; accessed 1 June 2014).
Belzer, F.O. & Southard, J.H. Principles of solid-organ preservation by cold storage. Transplantation 45, 673–676 (1988).
Ploeg, R.J. et al. Successful 72-hour cold storage kidney preservation with UW solution. Transplant. Proc. 20, 935–938 (1988).
Fahy, G.M. et al. Cryopreservation of organs by vitrification: perspectives and recent advances. Cryobiology 48, 157–178 (2004).
Song, Y.C., Khirabadi, B.S., Lightfoot, F., Brockbank, K.G. & Taylor, M.J. Vitreous cryopreservation maintains the function of vascular grafts. Nat. Biotechnol. 18, 296–299 (2000).
Fahy, G.M., Wowk, B. & Wu, J. Cryopreservation of complex systems: the missing link in the regenerative medicine supply chain. Rejuvenation Res. 9, 279–291 (2006).
Pegg, D.E. The relevance of ice crystal formation for the cryopreservation of tissues and organs. Cryobiology 60, S36–S44 (2010).
Rall, W.F. & Fahy, G.M. Ice-free cryopreservation of mouse embryos at −196 °C by vitrification. Nature 313, 573–575 (1985).
Fahy, G.M. et al. Physical and biological aspects of renal vitrification. Organogenesis 5, 167–175 (2009).
al-Abdulla, N.A. et al. The effects of supercooling chemicals on myocardial ultrastructure: a transmission electron microscopy case study. Conn. Med. 59, 387–399 (1995).
Banafsche, R., Pomer, S. & Staehler, G. Nonfreezing cryopreservation—a possible means of improving long-term transplant function? Transpl. Int. 11, 127–136 (1998).
Kato, H., Tomita, S., Yamaguchi, S., Ohtake, H. & Watanabe, G. Subzero 24-hr nonfreezing rat heart preservation: a novel preservation method in a variable magnetic field. Transplantation 94, 473–477 (2012).
Monzen, K. et al. The use of a supercooling refrigerator improves the preservation of organ grafts. Biochem. Biophys. Res. Commun. 337, 534–539 (2005).
Okamoto, T. et al. Successful sub-zero non-freezing preservation of rat lungs at −2 °C utilizing a new supercooling technology. J. Heart Lung Transplant. 27, 1150–1157 (2008).
Scotte, M. et al. Liver preservation below 0 °C with UW solution and 2,3-butanediol. Cryobiology 33, 54–61 (1996).
Yoshida, K. et al. A novel conception for liver preservation at a temperature just above freezing point. J. Surg. Res. 81, 216–223 (1999).
Ishine, N., Rubinsky, B. & Lee, C.Y. Transplantation of mammalian livers following freezing: vascular damage and functional recovery. Cryobiology 40, 84–89 (2000).
Guarrera, J.V. et al. Hypothermic machine preservation in human liver transplantation: the first clinical series. Am. J. Transplant. 10, 372–381 (2010).
Moers, C. et al. Machine perfusion or cold storage in deceased-donor kidney transplantation. N. Engl. J. Med. 360, 7–19 (2009).
Dutkowski, P., de Rougemont, O. & Clavien, P.A. Machine perfusion for 'Marginal' liver grafts. Am. J. Transplant. 8, 917–924 (2008).
Maathuis, M.H. et al. Improved kidney graft function after preservation using a novel hypothermic machine perfusion device. Ann. Surg. 246, 982–988, discussion 989–991 (2007).
Nowak, G., Ungerstedt, J., Wernerson, A., Ungerstedt, U. & Ericzon, B.G. Hepatic cell membrane damage during cold preservation sensitizes liver grafts to rewarming injury. J. Hepatobiliary Pancreat. Surg. 10, 200–205 (2003).
Usta, O.B. et al. Supercooling as a viable non-freezing cell preservation method of rat hepatocytes. PLoS ONE 8, e69334 (2013).
Taylor, M.J. in Advances in Biopreservation (eds. Baust, J.G., Baust, J.M. & Group, F.) Ch. 2 (CRC Press, Boca Raton, Florida, 2007).
Gao, W., Bentley, R.C., Madden, J.F. & Clavien, P.A. Apoptosis of sinusoidal endothelial cells is a critical mechanism of preservation injury in rat liver transplantation. Hepatology 27, 1652–1660 (1998).
Huet, P.M. et al. Sinusoidal endothelial cell and hepatocyte death following cold ischemia-warm reperfusion of the rat liver. Hepatology 39, 1110–1119 (2004).
Schlegel, A., Rougemont, O., Graf, R., Clavien, P.A. & Dutkowski, P. Protective mechanisms of end-ischemic cold machine perfusion in DCD liver grafts. J. Hepatol. 58, 278–286 (2013).
Mühlbacher, F., Langer, F. & Mittermayer, C. Preservation solutions for transplantation. Transplant Proc. 31, 2069–2070 (1999).
Dutheil, D., Underhaug Gjerde, A., Petit-Paris, I., Mauco, G. & Holmsen, H. Polyethylene glycols interact with membrane glycerophospholipids: is this part of their mechanism for hypothermic graft protection? J. Chem. Biol. 2, 39–49 (2009).
Oltean, M. et al. Intraluminal polyethylene glycol stabilizes tight junctions and improves intestinal preservation in the rat. Am. J. Transplant. 12, 2044–2051 (2012).
Bessems, M., Doorschodt, B.M., Hooijschuur, O., van Vliet, A.K. & van Gulik, T.M. Optimization of a new preservation solution for machine perfusion of the liver: which is the preferred colloid? Transplant. Proc. 37, 329–331 (2005).
Storey, K.B., Bischof, J. & Rubinsky, B. Cryomicroscopic analysis of freezing in liver of the freeze-tolerant wood frog. Am. J. Physiol. 263, R185–R194 (1992).
Klepper, J. et al. Erythrocyte 3-O-methyl-d-glucose uptake assay for diagnosis of glucose-transporter-protein syndrome. J. Clin. Lab. Anal. 13, 116–121 (1999).
Sugimachi, K., Roach, K.L., Rhoads, D.B., Tompkins, R.G. & Toner, M. Nonmetabolizable glucose compounds impart cryotolerance to primary rat hepatocytes. Tissue Eng. 12, 579–588 (2006).
Gok, M.A. et al. Improving the quality of kidneys from non-heart-beating donors, using streptokinase: an animal model. Transplantation 73, 1869–1874 (2002).
Minor, T. & Paul, A. Hypothermic reconditioning in organ transplantation. Curr. Opin. Organ Transplant. 18, 161–167 (2013).
Vogel, T., Brockmann, J.G. & Friend, P.J. Ex vivo normothermic liver perfusion: an update. Curr. Opin. Organ Transplant. 15, 167–172 (2010).
Berendsen, T.A. et al. A simplified subnormothermic machine perfusion system restores ischemically damaged liver grafts in a rat model of orthotopic liver transplantation. Transplant. Res. 1, 6 (2012).
Bruinsma, B.G. et al. Subnormothermic machine perfusion for ex vivo preservation and recovery of the human liver for transplantation. Am. J. Transplant. 14, 1400–1409 (2014).
Bruinsma, B.G., Berendsen, T.A., Izamis, M.L., Yarmush, M.L. & Uygun, K. Determination and extension of the limits to static cold storage using subnormothermic machine perfusion. Int. J. Artif. Organs 36, 775–780 (2013).
Tolboom, H. et al. A model for normothermic preservation of the rat liver. Tissue Eng. 13, 2143–2151 (2007).
Tolboom, H. et al. Recovery of warm ischemic rat liver grafts by normothermic extracorporeal perfusion. Transplantation 87, 170–177 (2009).
Izamis, M.-L. et al. Simple machine perfusion significantly enhances hepatocyte yields of ischemic and fresh rat livers. Cell Med. 4, 109–123 (2013).
Acknowledgements
Funding from the US National Institutes of Health (grants R01EB008678, R01DK096075, R01DK084053, R00DK080942, R00DK088962 and F32 DK095558) and the Shriners Hospitals for Children is gratefully acknowledged.
Author information
Authors and Affiliations
Contributions
T.A.B., B.G.B., M.-L.I., O.B.U., B.E.U., M.T., M.L.Y. and K.U. conceived and designed the study; T.A.B., B.G.B., C.F.P. and N.S. performed data acquisition; T.A.B., B.G.B., M.-L.I., M.L.Y. and K.U. analyzed and interpreted data; T.A.B. and B.G.B. performed rat liver procurement and transplantation; T.A.B., B.G.B. and M.-L.I. designed and constructed the supercooling device and container; T.A.B., B.G.B. and K.U. wrote the manuscript; T.A.B., B.G.B., C.F.P., N.S., O.B.U., B.E.U., M.-L.I., M.T., M.L.Y. and K.U. participated in critical revision of the manuscript for intellectual content; T.A.B., B.G.B. and K.U. performed statistical analysis; and M.L.Y. and K.U. obtained funding. All authors contributed to the preparation of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
M.L.I., B.E.U., K.U. and M.L.Y. are inventors on a pending international patent application that involves some of the perfusion technology used in this work (WO/2011/002926); T.A.B., M.L.I., B.E.U., M.L.Y. and K.U. are inventors on a pending international patent application that describes the supercooling protocol employed in this work (WO/2011/35223); and B.G.B., M.L.I. and K.U. have a provisional patent application related to this work that describes scale-up of the perfusion protocol to human livers. K.U. has a financial interest in Organ Solutions LLC, a company focused on developing organ preservation technology. K.U.'s interests are managed by the Massachusetts General Hospital and Partners HealthCare in accordance with their conflict-of-interest policies.
Source data
Rights and permissions
About this article
Cite this article
Berendsen, T., Bruinsma, B., Puts, C. et al. Supercooling enables long-term transplantation survival following 4 days of liver preservation. Nat Med 20, 790–793 (2014). https://doi.org/10.1038/nm.3588
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.3588
This article is cited by
-
Vitrification and nanowarming enable long-term organ cryopreservation and life-sustaining kidney transplantation in a rat model
Nature Communications (2023)
-
Cryopreservation of Whole Rat Livers by Vitrification and Nanowarming
Annals of Biomedical Engineering (2023)
-
Partial freezing of rat livers extends preservation time by 5-fold
Nature Communications (2022)
-
Winter is coming: the future of cryopreservation
BMC Biology (2021)
-
Contactless and spatially structured cooling by directing thermal radiation
Scientific Reports (2021)