Abstract
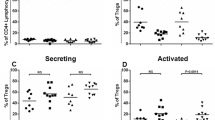
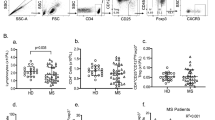
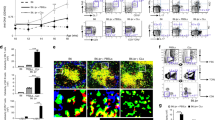
Human CD4+CD25highCD127−FoxP3+ regulatory T (Treg) cells suppress immune responses in vitro and in vivo1. Reduced suppressive function and/or number of peripheral Treg cells has been previously reported in autoimmune disorders2,3. Treg cells represent the most actively replicating compartment within the CD4+ cells in vivo, but they are hyporesponsive to classical T cell receptor (TCR) stimulation in vitro, a condition that is secondary to their overactive metabolic state4,5. Here we report that proliferation of Treg cells after TCR stimulation is impaired in subjects with relapsing-remitting multiple sclerosis (RRMS) because of altered interleukin-2 (IL-2) secretion and IL-2 receptor (IL-2R)–signal transducer and activator of transcription 5 (STAT5) signaling. This is associated with decreased expression of the forkhead box P3 (FoxP3) 44- and 47-kDa splicing forms, overactivation of S6 ribosomal protein (a downstream target of the mammalian target of rapamycin, mTOR) and altered activity of the cyclin-dependent kinase inhibitor p27 (p27kip1) and extracellular signal–related kinases 1 and 2 (ERK1/2). The impaired capacity of Treg cells to proliferate in RRMS correlates with the clinical state of the subject, where increasing disease severity is associated with a decline in Treg cell expansion. These results suggest a previously unrecognized mechanism that may account for the progressive loss of Treg cells in autoimmune disease.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Change history
17 January 2014
In the version of this article published, the data points on the graph in Figure 2g were incorrectly labeled. The black data points should be Controls and the gray data points should be RRMS. The error has been corrected in the HTML and PDF versions of the article.
References
Nylander, A. & Hafler, D.A. Multiple sclerosis. J. Clin. Invest. 122, 1180–1188 (2012).
Sakaguchi, S. et al. Foxp3+ CD25+ CD4+ natural regulatory T cells in dominant self-tolerance and autoimmune disease. Immunol. Rev. 212, 8–27 (2006).
Viglietta, V., Baecher-Allan, C., Weiner, H.L. & Hafler, D.A. Loss of functional suppression by CD4+CD25+ regulatory T cells in patients with multiple sclerosis. J. Exp. Med. 199, 971–979 (2004).
Haas, J. et al. Prevalence of newly generated naive regulatory T cells (Treg) is critical for Treg suppressive function and determines Treg dysfunction in multiple sclerosis. J. Immunol. 179, 1322–1330 (2007).
Matarese, G. et al. Leptin increase in multiple sclerosis associates with reduced number of CD4+CD25+ regulatory T cells. Proc. Natl. Acad. Sci. USA 102, 5150–5155 (2005).
Borsellino, G. et al. Expression of ectonucleotidase CD39 by Foxp3+ Treg cells: hydrolysis of extracellular ATP and immune suppression. Blood 110, 1225–1232 (2007).
Long, S.A. & Buckner, J.H. CD4+FOXP3+ T regulatory cells in human autoimmunity: more than a numbers game. J. Immunol. 187, 2061–2066 (2011).
Fisson, S. et al. Continuous activation of autoreactive CD4+ CD25+ regulatory T cells in the steady state. J. Exp. Med. 198, 737–746 (2003).
Vukmanovic-Stejic, M. et al. Human CD4+ CD25hi Foxp3+ regulatory T cells are derived by rapid turnover of memory populations in vivo. J. Clin. Invest. 116, 2423–2433 (2006).
Booth, N.J. et al. Different proliferative potential and migratory characteristics of human CD4+ regulatory T cells that express either CD45RA or CD45RO. J. Immunol. 184, 4317–4326 (2010).
Lombardi, G., Sidhu, S., Batchelor, R. & Lechler, R. Anergic T cells as suppressor cells in vitro. Science 264, 1587–1589 (1994).
Lechler, R., Chai, J.G., Marelli-Berg, F. & Lombardi, G. The contributions of T-cell anergy to peripheral T-cell tolerance. Immunology 103, 262–269 (2001).
James, M.J. et al. Anergic T cells exert antigen-independent inhibition of cell-cell interactions via chemokine metabolism. Blood 102, 2173–2179 (2003).
Liu, W. et al. CD127 expression inversely correlates with FoxP3 and suppressive function of human CD4+ T reg cells. J. Exp. Med. 203, 1701–1711 (2006).
Seddiki, N. et al. Expression of interleukin (IL)-2 and IL-7 receptors discriminates between human regulatory and activated T cells. J. Exp. Med. 203, 1693–1700 (2006).
De Rosa, V. et al. A key role of leptin in the control of regulatory T cell proliferation. Immunity 26, 241–255 (2007).
Procaccini, C. et al. An oscillatory switch in mTOR kinase activity sets regulatory T cell responsiveness. Immunity 33, 929–941 (2010).
Procaccini, C., Galgani, M., De Rosa, V. & Matarese, G. Intracellular metabolic pathways control immune tolerance. Trends Immunol. 33, 1–7 (2012).
Batocchi, A.P. et al. Leptin as a marker of multiple sclerosis activity in patients treated with interferon-β. J. Neuroimmunol. 139, 150–154 (2003).
Tang, Q. et al. Central role of defective interleukin-2 production in the triggering of islet autoimmune destruction. Immunity 28, 687–697 (2008).
Long, S.A. et al. Defects in IL-2R signaling contribute to diminished maintenance of FOXP3 expression in CD4+CD25+ regulatory T-cells of type 1 diabetic subjects. Diabetes 59, 407–415 (2010).
Cheng, G., Yu, A. & Malek, T.R. T-cell tolerance and the multi-functional role of IL-2R signaling in T-regulatory cells. Immunol. Rev. 241, 63–76 (2011).
Cheng, G., Yu, A., Dee, M.J. & Malek, T.R. IL-2R signaling is essential for functional maturation of regulatory T cells during thymic development. J. Immunol. 190, 1567–1575 (2013).
Maruyama, T., Konkel, J.E., Zamarron, B.F. & Chen, W. The molecular mechanisms of Foxp3 gene regulation. Semin. Immunol. 23, 418–423 (2011).
Kaur, G., Goodall, J.C., Jarvis, L.B. & Hill Gaston, J.S. Characterization of Foxp3 splice variants in human CD4+ and CD8+ cells—identification od Foxp3 Δ7 in human regulatory T cell. Mol. Immunol. 48, 321–332 (2010).
Smith, E.L., Finney, H.M., Nesbitt, A.M., Ramsdell, F. & Robinson, M.K. Splice variants of human Foxp3 are functional inhibitors of human CD4+ T cell activation. Immunology 119, 203–211 (2006).
Allan, S.E. et al. The role of two Foxp3 isoforms in the generation of human CD4+ Tregs. J. Clin. Invest. 115, 3276–3284 (2005).
Mailer, R.K.W., Falk, K. & Rötzschke, O. Absence of leucine zipper in the natural Foxp3Δ2Δ7 isoform does not affect dimerization but abrogates suppressive capacity. PLoS ONE 4, e6104 (2009).
Lord, J.D., Valliant-Saunders, K., Hahn, H., Thirlby, R.C. & Ziegler, S.F. Paradoxically increased Foxp3+ T cells in IBD do not preferentially express the isoform of Foxp3 lacking exon2. Dig. Dis. Sci. 57, 2846–2855 (2012).
Suzuki, K. et al. Decreased mRNA expression of two Foxp3 isoforms in peripheral blood mononuclear cells from patients with rheumatoid arthritis and systemic lupus erythematosus. Int. J. Immunopathol. Pharmacol. 24, 7–14 (2011).
Murdoch, D.J. & Chow, E.D. A graphical display of large correlation matrices. Am. Stat. 50, 178–180 (1996).
Dendrou, C.A. et al. Cell-specific protein phenotypes for the autoimmune locus IL2RA using a genotype-selectable human bioresurce. Nat. Genet. 41, 1011–1015 (2009).
Cavanillas, M.L. et al. Polymorphisms in the IL2, IL2RA and IL2RB genes in multiple sclerosis risk. Eur. J. Hum. Genet. 18, 794–799 (2010).
Kofler, D.M., Severson, C.A., Mousissian, N., Jager, P.L. & Hafler, D.A. The CD6 multiple sclerosis susceptibility allele is associated with alterations in CD4+ T cell proliferation. J. Immunol. 187, 3286–3291 (2011).
Guo, W. A note on adaptive Bonferroni and Holm procedures under dependence. Biometrika 96, 1012–1018 (2009).
Acknowledgements
This work is supported by grants from Fondazione Italiana Sclerosi Multipla (FISM) 2012/R/11, the European Union IDEAS Programme European Research Council Starting Grant “menTORingTregs” 310496, the Ministero della Salute Grant GR-2010-2315414 and the Fondo per gli Investimenti della Ricerca di Base (FIRB) grant RBFR12I3UB_004; A.L.C. is supported by the US National Institutes of Health grant AI95921. The authors thank S. De Simone and M. Galgani from the MoFlo Cell Sorting Facility and C. La Rocca for assistance in the Treg cell isolations. This work is dedicated to the memory of E. Papa and S. Zappacosta.
Author information
Authors and Affiliations
Contributions
G.M. designed the research; F.C., V.D.R. and C.P. performed the research; G.M., F.C., V.D.R., P.B.C., S.M., A.P., D.B. and A.L.C. analyzed the data and interpreted results; D.B. and A.P. performed statistical analyses; P.B.C. and S.M. recruited subjects with RRMS and analyzed clinical data and G.M., F.C., V.D.R. and A.L.C. wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–14 and Supplementary Tables 1–4 (PDF 2319 kb)
Rights and permissions
About this article
Cite this article
Carbone, F., De Rosa, V., Carrieri, P. et al. Regulatory T cell proliferative potential is impaired in human autoimmune disease. Nat Med 20, 69–74 (2014). https://doi.org/10.1038/nm.3411
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.3411
This article is cited by
-
The regulation and differentiation of regulatory T cells and their dysfunction in autoimmune diseases
Nature Reviews Immunology (2024)
-
Lrig1-expression confers suppressive function to CD4+ cells and is essential for averting autoimmunity via the Smad2/3/Foxp3 axis
Nature Communications (2023)
-
CD19+CD24highCD27+ B cell and interleukin 35 as potential biomarkers of disease activity in systemic lupus erythematosus patients
Advances in Rheumatology (2022)
-
Anti-neoplastic sulfonamides alter the metabolic homeostasis and disrupt the suppressor activity of regulatory T cells
Scientific Reports (2022)
-
A validated generally applicable approach using the systematic assessment of disease modules by GWAS reveals a multi-omic module strongly associated with risk factors in multiple sclerosis
BMC Genomics (2021)