Abstract
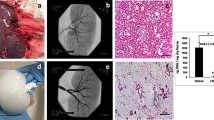
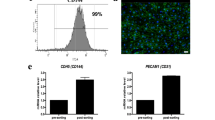
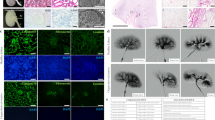
Approximately 100,000 individuals in the United States currently await kidney transplantation, and 400,000 individuals live with end-stage kidney disease requiring hemodialysis. The creation of a transplantable graft to permanently replace kidney function would address donor organ shortage and the morbidity associated with immunosuppression. Such a bioengineered graft must have the kidney's architecture and function and permit perfusion, filtration, secretion, absorption and drainage of urine. We decellularized rat, porcine and human kidneys by detergent perfusion, yielding acellular scaffolds with vascular, cortical and medullary architecture, a collecting system and ureters. To regenerate functional tissue, we seeded rat kidney scaffolds with epithelial and endothelial cells and perfused these cell-seeded constructs in a whole-organ bioreactor. The resulting grafts produced rudimentary urine in vitro when perfused through their intrinsic vascular bed. When transplanted in an orthotopic position in rat, the grafts were perfused by the recipient's circulation and produced urine through the ureteral conduit in vivo.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Change history
18 October 2013
In the version of this article initially published, the human kidneys in Figure 2a,b were incorrectly described as porcine and the porcine kidneys in Figure 2c,d were incorrectly described as human in the figure legend. The errors have been corrected in the HTML and PDF versions of the article.
References
Centers for Disease Control and Prevention. National chronic kidney disease fact sheet: general information and national estimates on chronic kidney disease in the United States, 2010. 〈http://www.cdc.gov/DIABETES//pubs/factsheets/kidney.htm〉 (2010).
US Department of Health and Human Services. National transplantation data report. OPTN: Data Organ Procurement and Transplantation Network. 〈http://optn.transplant.hrsa.gov/latestData/step2.asp?〉 (2013).
Kawai, T. et al. HLA-mismatched renal transplantation without maintenance immunosuppression. N. Engl. J. Med. 358, 353–361 (2008).
Humes, H.D., Krauss, J.C., Cieslinski, D.A. & Funke, A.J. Tubulogenesis from isolated single cells of adult mammalian kidney: clonal analysis with a recombinant retrovirus. Am. J. Physiol. 271, F42–F49 (1996).
Humes, H.D., MacKay, S.M., Funke, A.J. & Buffington, D.A. Tissue engineering of a bioartificial renal tubule assist device: in vitro transport and metabolic characteristics. Kidney Int. 55, 2502–2514 (1999).
Humes, H.D., Buffington, D.A., MacKay, S.M., Funke, A.J. & Weitzel, W.F. Replacement of renal function in uremic animals with a tissue-engineered kidney. Nat. Biotechnol. 17, 451–455 (1999).
Humes, H.D. et al. Initial clinical results of the bioartificial kidney containing human cells in ICU patients with acute renal failure. Kidney Int. 66, 1578–1588 (2004).
Humes, H.D., Weitzel, W.F. & Fissell, W.H. Renal cell therapy in the treatment of patients with acute and chronic renal failure. Blood Purif. 22, 60–72 (2004).
Rogers, S.A. & Hammerman, M.R. Prolongation of life in anephric rats following de novo renal organogenesis. Organogenesis 1, 22–25 (2004).
Gura, V., Macy, A.S., Beizai, M., Ezon, C. & Golper, T.A. Technical breakthroughs in the wearable artificial kidney (WAK). Clin. J. Am. Soc. Nephrol. 4, 1441–1448 (2009).
Fissell, W.H. & Roy, S. The implantable artificial kidney. Semin. Dial. 22, 665–670 (2009).
Atala, A., Bauer, S.B., Soker, S., Yoo, J.J. & Retik, A.B. Tissue-engineered autologous bladders for patients needing cystoplasty. Lancet 367, 1241–1246 (2006).
Ott, H.C. et al. Perfusion-decellularized matrix: using nature's platform to engineer a bioartificial heart. Nat. Med. 14, 213–221 (2008).
Ott, H.C. et al. Regeneration and orthotopic transplantation of a bioartificial lung. Nat. Med. 16, 927–933 (2010).
Nakayama, K.H., Batchelder, C.A., Lee, C.I. & Tarantal, A.F. Decellularized rhesus monkey kidney as a three-dimensional scaffold for renal tissue engineering. Tissue Eng. Part A 16, 2207–2216 (2010).
Ross, E.A. et al. Embryonic stem cells proliferate and differentiate when seeded into kidney scaffolds. J. Am. Soc. Nephrol. 20, 2338–2347 (2009).
Orlando, G. et al. Production and implantation of renal extracellular matrix scaffolds from porcine kidneys as a platform for renal bioengineering investigations. Ann. Surg. 256, 363–370 (2012).
Song, J.J. & Ott, H.C. Organ engineering based on decellularized matrix scaffolds. Trends Mol. Med. 17, 424–432 (2011).
Sullivan, D.C. et al. Decellularization methods of porcine kidneys for whole organ engineering using a high-throughput system. Biomaterials 33, 7756–7764 (2012).
Olivetti, G., Anversa, P., Rigamonti, W., Vitali-Mazza, L. & Loud, A.V. Morphometry of the renal corpuscle during normal postnatal growth and compensatory hypertrophy. A light microscope study. J. Cell Biol. 75, 573–585 (1977).
Welling, L.W. & Grantham, J.J. Physical properties of isolated perfused renal tubules and tubular basement membranes. J. Clin. Invest. 51, 1063–1075 (1972).
Falk, G. Maturation of renal function in infant rats. Am. J. Physiol. 181, 157–170 (1955).
Bray, J. & Robinson, G.B. Influence of charge on filtration across renal basement membrane films in vitro. Kidney Int. 25, 527–533 (1984).
Deen, W.M., Lazzara, M.J. & Myers, B.D. Structural determinants of glomerular permeability. Am. J. Physiol. Renal Physiol. 281, F579–F596 (2001).
Humes, H.D. Acute renal failure: prevailing challenges and prospects for the future. Kidney Int. Suppl. 50, S26–S32 (1995).
Quint, C. et al. Decellularized tissue-engineered blood vessel as an arterial conduit. Proc. Natl. Acad. Sci. USA 108, 9214–9219 (2011).
Elliott, M.J. et al. Stem-cell-based, tissue engineered tracheal replacement in a child: a 2-year follow-up study. Lancet 380, 994–1000 (2012).
Dandapani, S.V. et al. α–-actinin-4 is required for normal podocyte adhesion. J. Biol. Chem. 282, 467–477 (2007).
Kretzler, M. Regulation of adhesive interaction between podocytes and glomerular basement membrane. Microsc. Res. Tech. 57, 247–253 (2002).
Cybulsky, A.V., Carbonetto, S., Huang, Q., McTavish, A.J. & Cyr, M.D. Adhesion of rat glomerular epithelial cells to extracellular matrices: role of β1 integrins. Kidney Int. 42, 1099–1106 (1992).
Borza, C.M. et al. Human podocytes adhere to the KRGDS motif of the α3α4α5 collagen IV network. J. Am. Soc. Nephrol. 19, 677–684 (2008).
Acknowledgements
The present study was supported by the US National Institutes of Health (NIH) Director's New Innovator Award DP2 OD008749-01 and departmental funds. J.J.S. was supported by an AΩA Research Fellowship and an American Heart Association Predoctoral Fellowship. The Program in Membrane Biology Microscopy Core is supported by NIH grants DK43351 and DK57521. We further thank Q.C. Ott for critical review and editing of the manuscript. We thank C. Hoffman, J. Beagle and M. Duggan for technical support with urine sample analysis. We thank M. McKee for her expert support with transmission electron microscopy and A. Tisdale for her expert support with scanning electron microscopy.
Author information
Authors and Affiliations
Contributions
H.C.O. conceived, designed and oversaw all of the studies, collection of results, interpretation of the data and writing of the manuscript and was also responsible for the primary undertaking, completion and supervision of all experiments. J.J.S. and J.P.G. performed animal surgeries, conducted decellularization and whole-organ culture experiments and performed in vitro and in vivo testing. S.E.G. was responsible for cell culture, preparation of cell suspensions and matrix characterization. G.G. characterized fetal lung cells and scaffolds and regenerated constructs using various imaging techniques. J.P.V. provided input on tissue engineering aspects and reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
(PDF 297 kb)
Rights and permissions
About this article
Cite this article
Song, J., Guyette, J., Gilpin, S. et al. Regeneration and experimental orthotopic transplantation of a bioengineered kidney. Nat Med 19, 646–651 (2013). https://doi.org/10.1038/nm.3154
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.3154
This article is cited by
-
Optimization strategies of mesenchymal stem cell-based therapy for acute kidney injury
Stem Cell Research & Therapy (2023)
-
Effect of different decellularization protocols on reendothelialization with human cells for a perfused renal bioscaffold of the rat
BMC Biotechnology (2023)
-
In-vivo oogenesis of oogonial and mesenchymal stem cells seeded in transplanted ovarian extracellular matrix
Journal of Ovarian Research (2023)
-
Replacing renal function using bioengineered tissues
Nature Reviews Bioengineering (2023)
-
Directed self-assembly of a xenogeneic vascularized endocrine pancreas for type 1 diabetes
Nature Communications (2023)