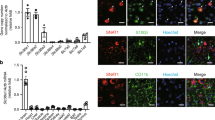
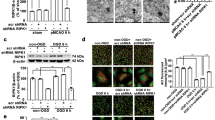
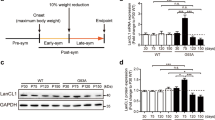
Abstract
Previous attempts to identify neuroprotective targets by studying the ischemic cascade and devising ways to suppress it have failed to translate to efficacious therapies for acute ischemic stroke1. We hypothesized that studying the molecular determinants of endogenous neuroprotection in two well-established paradigms, the resistance of CA3 hippocampal neurons to global ischemia2 and the tolerance conferred by ischemic preconditioning (IPC)3, would reveal new neuroprotective targets. We found that the product of the tuberous sclerosis complex 1 gene (TSC1), hamartin, is selectively induced by ischemia in hippocampal CA3 neurons. In CA1 neurons, hamartin was unaffected by ischemia but was upregulated by IPC preceding ischemia, which protects the otherwise vulnerable CA1 cells. Suppression of hamartin expression with TSC1 shRNA viral vectors both in vitro and in vivo increased the vulnerability of neurons to cell death following oxygen glucose deprivation (OGD) and ischemia. In vivo, suppression of TSC1 expression increased locomotor activity and decreased habituation in a hippocampal-dependent task. Overexpression of hamartin increased resistance to OGD by inducing productive autophagy through an mTORC1-dependent mechanism.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Accession codes
References
O'Collins, V.E. et al. 1,026 experimental treatments in acute stroke. Ann. Neurol. 59, 467–477 (2006).
Kirino, T. Delayed neuronal death in the gerbil hippocampus following ischemia. Brain Res. 239, 57–69 (1982).
Dirnagl, U., Becker, K. & Meisel, A. Preconditioning and tolerance against cerebral ischaemia: from experimental strategies to clinical use. Lancet Neurol. 8, 398–412 (2009).
Petito, C.K., Feldmann, E., Pulsinelli, W.A. & Plum, F. Delayed hippocampal damage in humans following cardiorespiratory arrest. Neurology 37, 1281–1286 (1987).
Chen, J. et al. Expression of the apoptosis-effector gene, Bax, is up-regulated in vulnerable hippocampal CA1 neurons following global ischemia. J. Neurochem. 67, 64–71 (1996).
Ouyang, Y.B., Voloboueva, L.A., Xu, L.J. & Giffard, R.G. Selective dysfunction of hippocampal CA1 astrocytes contributes to delayed neuronal damage after transient forebrain ischemia. J. Neurosci. 27, 4253–4260 (2007).
Sun, H.S. et al. Suppression of hippocampal TRPM7 protein prevents delayed neuronal death in brain ischemia. Nat. Neurosci. 12, 1300–1307 (2009).
Zhang, Q.G. et al. Akt inhibits MLK3/JNK3 signaling by inactivating Rac1: a protective mechanism against ischemic brain injury. J. Neurochem. 98, 1886–1898 (2006).
Shioda, N., Han, F., Morioka, M. & Fukunaga, K. Bis(1-oxy-2-pyridinethiolato)oxovanadium(IV) enhances neurogenesis via phosphatidylinositol 3-kinase/Akt and extracellular signal regulated kinase activation in the hippocampal subgranular zone after mouse focal cerebral ischemia. Neuroscience 155, 876–887 (2008).
Endo, H., Nito, C., Kamada, H., Yu, F. & Chan, P.H. Akt/GSK3β survival signaling is involved in acute brain injury after subarachnoid hemorrhage in rats. Stroke 37, 2140–2146 (2006).
Gozal, E. et al. Proteomic analysis of CA1 and CA3 regions of rat hippocampus and differential susceptibility to intermittent hypoxia. J. Neurochem. 83, 331–345 (2002).
Kawagoe, J. et al. Distributions of heat shock protein (HSP) 70 and heat shock cognate protein (HSC) 70 mRNAs after transient focal ischemia in rat brain. Brain Res. 587, 195–202 (1992).
Kinouchi, H. et al. Induction of 70-kDa heat shock protein and hsp70 mRNA following transient focal cerebral ischemia in the rat. J. Cereb. Blood Flow Metab. 13, 105–115 (1993).
Colbourne, F., Li, H., Buchan, A.M. & Clemens, J.A. Continuing postischemic neuronal death in CA1: influence of ischemia duration and cytoprotective doses of NBQX and SNX-111 in rats. Stroke 30, 662–668 (1999).
Choo, A.Y. et al. Glucose addiction of TSC null cells is caused by failed mTORC1-dependent balancing of metabolic demand with supply. Mol. Cell 38, 487–499 (2010).
Di Nardo, A. et al. Tuberous sclerosis complex activity is required to control neuronal stress responses in an mTOR-dependent manner. J. Neurosci. 29, 5926–5937 (2009).
Tavazoie, S.F., Alvarez, V.A., Ridenour, D.A., Kwiatkowski, D.J. & Sabatini, B.L. Regulation of neuronal morphology and function by the tumor suppressors Tsc1 and Tsc2. Nat. Neurosci. 8, 1727–1734 (2005).
Andersen, M.B., Zimmer, J. & Sams-Dodd, F. Postischemic hyperactivity in the Mongolian gerbil correlates with loss of hippocampal neurons. Behav. Neurosci. 111, 1205–1216 (1997).
Kesner, R.P. Behavioral functions of the CA3 subregion of the hippocampus. Learn. Mem. 14, 771–781 (2007).
Mileson, B.E. & Schwartz, R.D. The use of locomotor activity as a behavioral screen for neuronal damage following transient forebrain ischemia in gerbils. Neurosci. Lett. 128, 71–76 (1991).
Tee, A.R., Manning, B.D., Roux, P.P., Cantley, L.C. & Blenis, J. Tuberous sclerosis complex gene products, tuberin and hamartin, control mTOR signaling by acting as a GTPase-activating protein complex toward Rheb. Curr. Biol. 13, 1259–1268 (2003).
Malagelada, C., Jin, Z.H., Jackson-Lewis, V., Przedborski, S. & Greene, L.A. Rapamycin protects against neuron death in in vitro and in vivo models of Parkinson's disease. J. Neurosci. 30, 1166–1175 (2010).
Yang, S.S. et al. Rapamycin protects heart from ischemia/reperfusion injury independent of autophagy by activating PI3 kinase-Akt pathway and mitochondria K(ATP) channel. Pharmazie 65, 760–765 (2010).
Chauhan, A., Sharma, U., Jagannathan, N.R., Reeta, K.H. & Gupta, Y.K. Rapamycin protects against middle cerebral artery occlusion induced focal cerebral ischemia in rats. Behav. Brain Res. 225, 603–609 (2011).
Gabryel, B., Kost, A. & Kasprowska, D. Neuronal autophagy in cerebral ischemia—a potential target for neuroprotective strategies? Pharmacol. Rep. 64, 1–15 (2012).
Klionsky, D.J. et al. Guidelines for the use and interpretation of assays for monitoring autophagy in higher eukaryotes. Autophagy 4, 151–175 (2008).
Zheng, Y.Q., Liu, J.X., Li, X.Z., Xu, L. & Xu, Y.G. RNA interference-mediated downregulation of Beclin1 attenuates cerebral ischemic injury in rats. Acta Pharmacol. Sin. 30, 919–927 (2009).
Wen, Y.D. et al. Neuronal injury in rat model of permanent focal cerebral ischemia is associated with activation of autophagic and lysosomal pathways. Autophagy 4, 762–769 (2008).
Carloni, S., Buonocore, G. & Balduini, W. Protective role of autophagy in neonatal hypoxia-ischemia induced brain injury. Neurobiol. Dis. 32, 329–339 (2008).
Wang, P. et al. Induction of autophagy contributes to the neuroprotection of nicotinamide phosphoribosyltransferase in cerebral ischemia. Autophagy 8, 77–87 (2012).
Liu, C., Gao, Y., Barrett, J. & Hu, B. Autophagy and protein aggregation after brain ischemia. J. Neurochem. 115, 68–78 (2010).
Sheng, R. et al. Autophagy activation is associated with neuroprotection in a rat model of focal cerebral ischemic preconditioning. Autophagy 6, 482–494 (2010).
van den Ouweland, A.M. et al. Characterisation of TSC1 promoter deletions in tuberous sclerosis complex patients. Eur. J. Hum. Genet. 19, 157–163 (2011).
Pulsinelli, W.A. & Buchan, A.M. The four-vessel occlusion rat model: method for complete occlusion of vertebral arteries and control of collateral circulation. Stroke 19, 913–914 (1988).
Guillemin, I. et al. A subcellular prefractionation protocol for minute amounts of mammalian cell cultures and tissue. Proteomics 5, 35–45 (2005).
Xu, D. et al. Novel MMP-9 substrates in cancer cells revealed by a label-free quantitative proteomics approach. Mol. Cell. Proteomics 7, 2215–2228 (2008).
Vogiatzi, T., Xilouri, M., Vekrellis, K. & Stefanis, L. Wild type alpha-synuclein is degraded by chaperone-mediated autophagy and macroautophagy in neuronal cells. J. Biol. Chem. 283, 23542–23556 (2008).
Xilouri, M., Vogiatzi, T., Vekrellis, K., Park, D. & Stefanis, L. Abberant alpha-synuclein confers toxicity to neurons in part through inhibition of chaperone-mediated autophagy. PLoS ONE 4, e5515 (2009).
Kaur, J., Zhao, Z., Geransar, R.M., Papadakis, M. & Buchan, A.M. Prior deafferentation confers long term protection to CA1 against transient forebrain ischemia and sustains GluR2 expression. Brain Res. 1075, 201–212 (2006).
Jiang, Y., Deacon, R., Anthony, D.C. & Campbell, S.J. Inhibition of peripheral TNF can block the malaise associated with CNS inflammatory diseases. Neurobiol. Dis. 32, 125–132 (2008).
Acknowledgements
This work was supported by the UK Medical Research Council grant G0500495 and by the Dunhill Medical Trust. A.M.B. is a senior investigator of the UK National Institute for Health Research (NIHR) and received funding from Fondation Leducq for neurovascular coupling. G.H. was funded through the NIHR Integrated Academic Training Programme and Oxford University Clinical Academic Graduate School. B.K. and C.W.D. were supported by the NIHR Biomedical Research Centre. S.N. was supported by the Deutsche Forschungsgemeinschaft. G.T. and S.M.W. received funding from National Health Service Blood and Transplant and the NIHR under its Programme Grants Scheme (NIHR Programmes RP-PG-0310-10001 and -10003). We would also like to thank R. Deacon from the Department of Experimental Psychology, University of Oxford, for providing us with the open-field apparatus and for his guidance with the behavioral experiments. We thank E. Martin Rendon for her input concerning the lentiviral vectors and J. Peeling for his evaluation of the manuscript. We dedicate this paper to the memory of our colleague and mentor, John P. MacManus.
Author information
Authors and Affiliations
Contributions
M.P. initiated and designed the study, carried out the proteomic and biochemical analyses, performed part of the in vitro and in vivo shRNA lentiviral studies and part of the in vitro overexpression studies and their analysis, and wrote the manuscript. G.H. carried out the subcellular fractionation and immunoblotting experiments for Figures 1 and 4. M.X. produced the overexpression lentiviral particles and helped with the analysis of long-lived protein degradation assays. L.C.H. carried out the rat surgeries for the IPC studies and contributed to the development of the subcellular fractionation protocol. S.N. assisted with the rat surgeries and immunofluorescence experiments and contributed in the interpretation of the proteomic data sets. G.T. assisted with the cortical culture experiments. S.M.W. supervised the National Health Service Blood and Transplant collaborative studies and critically reviewed and edited the manuscript. C.W.D. assisted with the IPA. R.C. assisted in the behavioral testing. Z.Z. carried out the rat surgeries to generate the tissue for the proteomic experiments and time course studies. M.M.M. and M.J.A.W. assisted with the design and conduct of the lentiviral overexpression experiments. B.K. supervised and helped with the proteomic analysis. K.V. contributed to the primary culture lentiviral studies, assisted with the in vivo shRNA experiments and designed, carried out and assisted in the analysis of protein degradation, autophagy and necrosis assays and supervised the collaboration. A.M.B. initiated and supervised the whole project. All authors edited the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–13, Supplementary Tables 1–9 and Supplementary Results and Discussion (PDF 3125 kb)
Rights and permissions
About this article
Cite this article
Papadakis, M., Hadley, G., Xilouri, M. et al. Tsc1 (hamartin) confers neuroprotection against ischemia by inducing autophagy. Nat Med 19, 351–357 (2013). https://doi.org/10.1038/nm.3097
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.3097
This article is cited by
-
Hypoxic mesenchymal stem cell-derived exosomes promote the survival of skin flaps after ischaemia–reperfusion injury via mTOR/ULK1/FUNDC1 pathways
Journal of Nanobiotechnology (2023)
-
Reinventing the Penumbra — the Emerging Clockwork of a Multi-modal Mechanistic Paradigm
Translational Stroke Research (2023)
-
The Multiple Roles of Autophagy in Neural Function and Diseases
Neuroscience Bulletin (2023)
-
The interplay between oxidative stress and autophagy: focus on the development of neurological diseases
Behavioral and Brain Functions (2022)
-
The neurovascular unit and systemic biology in stroke — implications for translation and treatment
Nature Reviews Neurology (2022)