Abstract
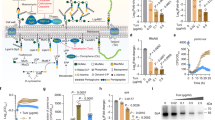
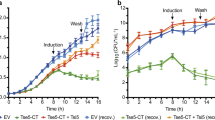
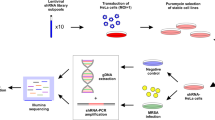
Widespread antibiotic resistance among important bacterial pathogens such as Staphylococcus aureus1 calls for alternative routes of drug development. Interfering with crucial virulence determinants is considered a promising new approach to control bacterial infection2. Phenol-soluble modulins (PSMs) are peptide toxins with multiple key roles in pathogenesis3,4,5 and have a major impact on the ability of highly virulent S. aureus to cause disease3,6. However, targeting PSMs for therapeutic intervention is hampered by their multitude and diversity. Here we report that an ATP-binding cassette transporter with previously unknown function is responsible for the export of all PSMs, thus representing a single target for complete obstruction of PSM production. The transporter had a strong effect on virulence phenotypes, such as neutrophil lysis, and the extent of its effect on the development of S. aureus infection was similar to that of the sum of all PSMs. Notably, the transporter was essential for bacterial growth. Furthermore, it contributed to producer immunity toward secreted PSMs and defense against PSM-mediated bacterial interference. Our study reveals a noncanonical, dedicated secretion mechanism for an important class of toxins and identifies this mechanism as a comprehensive potential target for the development of drugs to efficiently inhibit the growth and virulence of pathogenic staphylococci.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Lowy, F.D. Antimicrobial resistance: the example of Staphylococcus aureus. J. Clin. Invest. 111, 1265–1273 (2003).
Alksne, L.E. & Projan, S.J. Bacterial virulence as a target for antimicrobial chemotherapy. Curr. Opin. Biotechnol. 11, 625–636 (2000).
Wang, R. et al. Identification of novel cytolytic peptides as key virulence determinants for community-associated MRSA. Nat. Med. 13, 1510–1514 (2007).
Wang, R. et al. Staphylococcus epidermidis surfactant peptides promote biofilm maturation and dissemination of biofilm-associated infection in mice. J. Clin. Invest. 121, 238–248 (2011).
Kretschmer, D. et al. Human formyl peptide receptor 2 senses highly pathogenic Staphylococcus aureus. Cell Host Microbe 7, 463–473 (2010).
Kobayashi, S.D. et al. Comparative analysis of USA300 virulence determinants in a rabbit model of skin and soft tissue infection. J. Infect. Dis. 204, 937–941 (2011).
Cheung, G.Y. et al. Staphylococcus epidermidis strategies to avoid killing by human neutrophils. PLoS Pathog. 6, e1001133 (2010).
Cheung, G.Y., Duong, A.C. & Otto, M. Direct and synergistic hemolysis caused by Staphylococcus phenol-soluble modulins: implications for diagnosis and pathogenesis. Microbes Infect. 14, 380–386 (2012).
Periasamy, S. et al. How Staphylococcus aureus biofilms develop their characteristic structure. Proc. Natl. Acad. Sci. USA 109, 1281–1286 (2012).
Schwartz, K., Syed, A.K., Stephenson, R.E., Rickard, A.H. & Boles, B.R. Functional amyloids composed of phenol soluble modulins stabilize Staphylococcus aureus biofilms. PLoS Pathog. 8, e1002744 (2012).
Rautenberg, M., Joo, H.S., Otto, M. & Peschel, A. Neutrophil responses to staphylococcal pathogens and commensals via the formyl peptide receptor 2 relates to phenol-soluble modulin release and virulence. FASEB J. 25, 1254–1263 (2011).
Peterson, M.M. et al. Apolipoprotein B Is an innate barrier against invasive Staphylococcus aureus infection. Cell Host Microbe 4, 555–566 (2008).
Rothfork, J.M. et al. Inactivation of a bacterial virulence pheromone by phagocyte-derived oxidants: new role for the NADPH oxidase in host defense. Proc. Natl. Acad. Sci. USA 101, 13867–13872 (2004).
Surewaard, B.G. et al. Inactivation of staphylococcal phenol soluble modulins by serum lipoprotein particles. PLoS Pathog. 8, e1002606 (2012).
Forsman, H., Christenson, K., Bylund, J. & Dahlgren, C. Receptor-dependent and -independent immunomodulatory effects of phenol-soluble modulin peptides from Staphylococcus aureus on human neutrophils are abrogated through peptide inactivation by reactive oxygen species. Infect. Immun. 80, 1987–1995 (2012).
Mehlin, C., Headley, C.M. & Klebanoff, S.J. An inflammatory polypeptide complex from Staphylococcus epidermidis: isolation and characterization. J. Exp. Med. 189, 907–918 (1999).
Vuong, C. et al. Regulated expression of pathogen-associated molecular pattern molecules in Staphylococcus epidermidis: quorum-sensing determines pro-inflammatory capacity and production of phenol-soluble modulins. Cell Microbiol. 6, 753–759 (2004).
Yao, Y., Sturdevant, D.E. & Otto, M. Genomewide analysis of gene expression in Staphylococcus epidermidis biofilms: insights into the pathophysiology of S. epidermidis biofilms and the role of phenol-soluble modulins in formation of biofilms. J. Infect. Dis. 191, 289–298 (2005).
Queck, S.Y. et al. RNAIII-independent target gene control by the agr quorum-sensing system: insight into the evolution of virulence regulation in Staphylococcus aureus. Mol. Cell 32, 150–158 (2008).
Cheung, G.Y., Wang, R., Khan, B.A., Sturdevant, D.E. & Otto, M. Role of the accessory gene regulator agr in community-associated methicillin-resistant Staphylococcus aureus pathogenesis. Infect. Immun. 79, 1927–1935 (2011).
Otto, M. Basis of virulence in community-associated methicillin-resistant Staphylococcus aureus. Annu. Rev. Microbiol. 64, 143–162 (2010).
Zolnerciks, J.K., Andress, E.J., Nicolaou, M. & Linton, K.J. Structure of ABC transporters. Essays Biochem. 50, 43–61 (2011).
Walker, J.E., Saraste, M., Runswick, M.J. & Gay, N.J. Distantly related sequences in the α- and β-subunits of ATP synthase, myosin, kinases and other ATP-requiring enzymes and a common nucleotide binding fold. EMBO J. 1, 945–951 (1982).
Otto, M. Staphylococcus epidermidis—the 'accidental' pathogen. Nat. Rev. Microbiol. 7, 555–567 (2009).
Cogen, A.L. et al. Selective antimicrobial action is provided by phenol-soluble modulins derived from Staphylococcus epidermidis, a normal resident of the skin. J. Invest. Dermatol. 130, 192–200 (2010).
Joo, H.S., Cheung, G.Y. & Otto, M. Antimicrobial activity of community-associated methicillin-resistant Staphylococcus aureus is caused by phenol-soluble modulin derivatives. J. Biol. Chem. 286, 8933–8940 (2011).
Malachowa, N. et al. Global changes in Staphylococcus aureus gene expression in human blood. PLoS ONE 6, e18617 (2011).
Lowy, F.D. Staphylococcus aureus infections. N. Engl. J. Med. 339, 520–532 (1998).
Dorschner, R.A. et al. The mammalian ionic environment dictates microbial susceptibility to antimicrobial defense peptides. FASEB J. 20, 35–42 (2006).
Wells, J.M., Wilson, P.W. & Le Page, R.W. Improved cloning vectors and transformation procedure for Lactococcus lactis. J. Appl. Bacteriol. 74, 629–636 (1993).
Augustin, J. & Gotz, F. Transformation of Staphylococcus epidermidis and other staphylococcal species with plasmid DNA by electroporation. FEMS Microbiol. Lett. 54, 203–207 (1990).
Bae, T. & Schneewind, O. Allelic replacement in Staphylococcus aureus with inducible counter-selection. Plasmid 55, 58–63 (2006).
Vandecasteele, S.J., Peetermans, W.E., Merckx, R., Van Ranst, M. & Van Eldere, J. Use of gDNA as internal standard for gene expression in staphylococci in vitro and in vivo. Biochem. Biophys. Res. Commun. 291, 528–534 (2002).
Li, M. et al. The antimicrobial peptide-sensing system aps of Staphylococcus aureus. Mol. Microbiol. 66, 1136–1147 (2007).
Li, M. et al. Gram-positive three-component antimicrobial peptide-sensing system. Proc. Natl. Acad. Sci. USA 104, 9469–9474 (2007).
Acknowledgements
This work was supported by the Intramural Research Program of the National Institute of Allergy and Infectious Diseases (NIAID), US National Institutes of Health (NIH) (grant ZIA AI000904-10) to M.O. and the National Natural Science Foundation of China (grants 30900026, 81171623 and 81261120387) to M.L. We thank J. Kok (University of Groningen), F. Lowy (Columbia University) and G. Dunny (University of Minnesota) for lactococcal strains and plasmids and A. Peschel for critically reading the manuscript.
Author information
Authors and Affiliations
Contributions
H.-S.J. and M.O. performed HPLC measurements for PSM detection. E.R.F. performed electron microscopy. S.S.C., G.Y.C.C., A.C.D., V.Y.T. and T.D.D. performed mouse experiments. Y.S. and M.L. designed and performed experiments with neutrophils and human blood. S.S.C. performed all other experiments. S.S.C. and M.O. analyzed data, designed the study and wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–12 and Supplementary Tables 1–3 (PDF 3366 kb)
Rights and permissions
About this article
Cite this article
Chatterjee, S., Joo, HS., Duong, A. et al. Essential Staphylococcus aureus toxin export system. Nat Med 19, 364–367 (2013). https://doi.org/10.1038/nm.3047
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.3047
This article is cited by
-
High density lipoproteins mediate in vivo protection against staphylococcal phenol-soluble modulins
Scientific Reports (2021)
-
Rapid pathogen-specific recruitment of immune effector cells in the skin by secreted toxins
Nature Microbiology (2021)
-
Increased resistance of a methicillin-resistant Staphylococcus aureus Δagr mutant with modified control in fatty acid metabolism
AMB Express (2020)
-
Staphylococcus aureus induces DNA damage in host cell
Scientific Reports (2019)
-
Epistasis analysis uncovers hidden antibiotic resistance-associated fitness costs hampering the evolution of MRSA
Genome Biology (2018)