Abstract
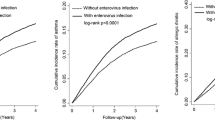
Immune tolerance is instituted early in life, during which time regulatory T (Treg) cells have an important role. Recurrent infections with respiratory syncytial virus (RSV) in early life increase the risk for asthma in adult life. Repeated infection of infant mice tolerized to ovalbumin (OVA) through their mother's milk with RSV induced allergic airway disease in response to OVA sensitization and challenge, including airway inflammation, hyper-reactivity and higher OVA-specific IgE, as compared to uninfected tolerized control mice. Virus infection induced GATA-3 expression and T helper type 2 (TH2) cytokine production in forkhead box P3 (FOXP3)+ Treg cells and compromised the suppressive function of pulmonary Treg cells in a manner that was dependent on interleukin-4 receptor α (IL-4Rα) expression in the host. Thus, by promoting a TH2-type inflammatory response in the lung, RSV induced a TH2-like effector phenotype in Treg cells and attenuated tolerance to an unrelated antigen (allergen). Our findings highlight a mechanism by which viral infection targets a host-protective mechanism in early life and increases susceptibility to allergic disease.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Levy, O. Innate immunity of the newborn: basic mechanisms and clinical correlates. Nat. Rev. Immunol. 7, 379–390 (2007).
Noble, V. et al. Respiratory status and allergy nine to 10 years after acute bronchiolitis. Arch. Dis. Child. 76, 315–319 (1997).
Sigurs, N., Bjarnason, R., Sigurbergsson, F. & Kjellman, B. Respiratory syncytial virus bronchiolitis in infancy is an important risk factor for asthma and allergy at age 7. Am. J. Respir. Crit. Care Med. 161, 1501–1507 (2000).
Sigurs, N. et al. Severe respiratory syncytial virus bronchiolitis in infancy and asthma and allergy at age 13. Am. J. Respir. Crit. Care Med. 171, 137–141 (2005).
Wu, P. et al. Evidence of a causal role of winter virus infection during infancy in early childhood asthma. Am. J. Respir. Crit. Care Med. 178, 1123–1129 (2008).
Sly, P.D., Kusel, M. & Holt, P.G. Do early-life viral infections cause asthma? J. Allergy Clin. Immunol. 125, 1202–1205 (2010).
Pullan, C.R. & Hey, E.N. Wheezing, asthma, and pulmonary dysfunction 10 years after infection with respiratory syncytial virus in infancy. Br. Med. J. (Clin. Res. Ed.) 284, 1665–1669 (1982).
Bacharier, L.B. et al. Determinants of asthma after severe respiratory syncytial virus bronchiolitis. J. Allergy Clin. Immunol. 91–100 (2012).
Culley, F.J., Pollott, J. & Openshaw, P.J. Age at first viral infection determines the pattern of T cell–mediated disease during reinfection in adulthood. J. Exp. Med. 196, 1381–1386 (2002).
Mold, J.E. et al. Maternal alloantigens promote the development of tolerogenic fetal regulatory T cells in utero. Science 322, 1562–1565 (2008).
Mold, J.E. et al. Fetal and adult hematopoietic stem cells give rise to distinct T cell lineages in humans. Science 330, 1695–1699 (2010).
Verhasselt, V. et al. Breast milk–mediated transfer of an antigen induces tolerance and protection from allergic asthma. Nat. Med. 14, 170–175 (2008).
Polte, T. & Hansen, G. Maternal tolerance achieved during pregnancy is transferred to the offspring via breast milk and persistently protects the offspring from allergic asthma. Clin. Exp. Allergy 38, 1950–1958 (2008).
Singleton, R., Etchart, N., Hou, S. & Hyland, L. Inability to evoke a long-lasting protective immune response to respiratory syncytial virus infection in mice correlates with ineffective nasal antibody responses. J. Virol. 77, 11303–11311 (2003).
Falsey, A.R., Hennessey, P.A., Formica, M.A., Cox, C. & Walsh, E.E. Respiratory syncytial virus infection in elderly and high-risk adults. N. Engl. J. Med. 352, 1749–1759 (2005).
Johnson, T.R. et al. Priming with secreted glycoprotein G of respiratory syncytial virus (RSV) augments interleukin-5 production and tissue eosinophilia after RSV challenge. J. Virol. 72, 2871–2880 (1998).
Tebbey, P.W., Hagen, M. & Hancock, G.E. Atypical pulmonary eosinophilia is mediated by a specific amino acid sequence of the attachment (G) protein of respiratory syncytial virus. J. Exp. Med. 188, 1967–1972 (1998).
Lukacs, N.W. et al. Differential immune responses and pulmonary pathophysiology are induced by two different strains of respiratory syncytial virus. Am. J. Pathol. 169, 977–986 (2006).
Curotto de Lafaille, M.A. et al. Adaptive Foxp3+ regulatory T cell–dependent and –independent control of allergic inflammation. Immunity 29, 114–126 (2008).
Ostroukhova, M. et al. Treg-mediated immunosuppression involves activation of the Notch-HESI axis by membrane-bound TGF-β. J. Clin. Invest. 116, 996–1004 (2006).
Ostroukhova, T.Y., Kulikov, A.V., Rozenkrants, A.A. & Smirnova, O.V. Overexpression of prolactin receptors during intrahepatic transplantation of RS1 rat cholangiocellular carcinoma cells. Bull. Exp. Biol. Med. 141, 364–367 (2006).
Zhou, X. et al. Instability of the transcription factor Foxp3 leads to the generation of pathogenic memory T cells in vivo. Nat. Immunol. 10, 1000–1007 (2009).
Oldenhove, G. et al. Decrease of Foxp3+ Treg cell number and acquisition of effector cell phenotype during lethal infection. Immunity 31, 772–786 (2009).
Zhang, D.H., Cohn, L., Ray, P., Bottomly, K. & Ray, A. Transcription factor GATA-3 is differentially expressed in murine TH1 and TH2 cells and controls TH2-specific expression of the interleukin-5 gene. J. Biol. Chem. 272, 21597–21603 (1997).
Zheng, W. & Flavell, R.A. The transcription factor GATA-3 is necessary and sufficient for TH2 cytokine gene expression in CD4 T cells. Cell 89, 587–596 (1997).
Bhattacharya, D. et al. Transcriptional profiling of antigen-dependent murine B cell differentiation and memory formation. J. Immunol. 179, 6808–6819 (2007).
Liu, W. et al. CD127 expression inversely correlates with FoxP3 and suppressive function of human CD4+ Treg cells. J. Exp. Med. 203, 1701–1711 (2006).
Mucida, D. et al. Reciprocal TH17 and regulatory T cell differentiation mediated by retinoic acid. Science 317, 256–260 (2007).
Benson, M.J., Pino-Lagos, K., Rosemblatt, M. & Noelle, R.J. All-trans retinoic acid mediates enhanced Treg cell growth, differentiation, and gut homing in the face of high levels of co-stimulation. J. Exp. Med. 204, 1765–1774 (2007).
Takaki, H. et al. STAT6 inhibits TGF-β1–mediated Foxp3 induction through direct binding to the Foxp3 promoter, which is reverted by retinoic acid receptor. J. Biol. Chem. 283, 14955–14962 (2008).
Lewkowich, I.P. et al. CD4+CD25+ T cells protect against experimentally induced asthma and alter pulmonary dendritic cell phenotype and function. J. Exp. Med. 202, 1549–1561 (2005).
Koch, M.A. et al. The transcription factor T-bet controls regulatory T cell homeostasis and function during type 1 inflammation. Nat. Immunol. 10, 595–602 (2009).
Chaudhry, A. et al. CD4+ regulatory T cells control TH17 responses in a Stat3-dependent manner. Science 326, 986–991 (2009).
Zheng, Y. et al. Regulatory T-cell suppressor program co-opts transcription factor IRF4 to control TH2 responses. Nature 458, 351–356 (2009).
Dominguez-Villar, M., Baecher-Allan, C.M. & Hafler, D.A. Identification of T helper type 1-like, Foxp3+ regulatory T cells in human autoimmune disease. Nat. Med. 17, 673–675 (2011).
Forsthuber, T., Yip, H.C. & Lehmann, P.V. Induction of TH1 and TH2 immunity in neonatal mice. Science 271, 1728–1730 (1996).
Openshaw, P.J. Immunity and immunopathology to respiratory syncytial virus. The mouse model. Am. J. Respir. Crit. Care Med. 152, S59–S62 (1995).
Graham, B.S., Johnson, T.R. & Peebles, R.S. Immune-mediated disease pathogenesis in respiratory syncytial virus infection. Immunopharmacology 48, 237–247 (2000).
Moore, M.L. et al. STAT1 negatively regulates lung basophil IL-4 expression induced by respiratory syncytial virus infection. J. Immunol. 183, 2016–2026 (2009).
Shirey, K.A. et al. Control of RSV-induced lung injury by alternatively activated macrophages is IL-4R α-, TLR4-, and IFN-β–dependent. Mucosal Immunol. 3, 291–300 (2010).
Yung, R.L. & Julius, A. Epigenetics, aging, and autoimmunity. Autoimmunity 41, 329–335 (2008).
Wenzel, S., Wilbraham, D., Fuller, R., Getz, E.B. & Longphre, M. Effect of an interleukin-4 variant on late phase asthmatic response to allergen challenge in asthmatic patients: results of two phase 2a studies. Lancet 370, 1422–1431 (2007).
Wang, Y., Su, M.A. & Wan, Y.Y. An essential role of the transcription factor GATA-3 for the function of regulatory T cells. Immunity 35, 337–348 (2011).
Wohlfert, E.A. et al. GATA3 controls Foxp3+ regulatory T cell fate during inflammation in mice. J. Clin. Invest. 121, 4503–4515 (2011).
Ray, A. & Cohn, L. TH2 cells and GATA-3 in asthma: new insights into the regulation of airway inflammation. J. Clin. Invest. 104, 985–993 (1999).
Mantel, P.Y. et al. GATA3-driven TH2 responses inhibit TGF-β1–induced FOXP3 expression and the formation of regulatory T cells. PLoS Biol. 5, e329 (2007).
Moore, M.L. et al. Cutting edge: oseltamivir decreases T cell GM1 expression and inhibits clearance of respiratory syncytial virus: potential role of endogenous sialidase in antiviral immunity. J. Immunol. 178, 2651–2654 (2007).
Domachowske, J.B. & Bonville, C.A. Overnight titration of human respiratory syncytial virus using quantitative shell vial amplification. Biotechniques 25, 644–647 (1998).
McMenamin, C., Pimm, C., McKersey, M. & Holt, P.G. Regulation of IgE responses to inhaled antigen in mice by antigen- specific γδ T cells. Science 265, 1869–1871 (1994).
Krishnamoorthy, N. et al. Activation of the c-kit–PI3 kinase axis induces the regulatory cytokine interleukin-6 in dendritic cells impacting allergic immune responses in the lung. Nat. Med. 14, 565–573 (2008).
Oriss, T.B. et al. Dynamics of dendritic cell phenotype and interactions with CD4+ T cells in airway inflammation and tolerance. J. Immunol. 174, 854–863 (2005).
Xu, H. et al. Indoleamine 2,3-dioxygenase in lung dendritic cells promotes TH2 responses and allergic inflammation. Proc. Natl. Acad. Sci. USA 105, 6690–6695 (2008).
Acknowledgements
We thank E. Shevach and G. Chattopadhyay (Laboratory of Immunology, National Institute of Allergy and Infectious Diseases, US National Institutes of Health (NIH)) for providing spleens of OT-II × FOXP3-eGFP knock-in mice and J. Zhu (Laboratory of Immunology, National Institute of Allergy and Infectious Diseases, NIH) for his efforts in identifying the source of these mice. DO11.10 transgenic mice were originally provided by K. Murphy (Washington University, St. Louis). This work was supported by NIH grants HL060207 and AI093116 (to P.R.), HL 077430 and AI048927 (to A.R.) and U19 AI095227 (to R.S.P.).
Author information
Authors and Affiliations
Contributions
N.K. designed and performed experiments, analyzed data and wrote the manuscript. A.K. and T.B.O. performed cell sorting and analyzed flow cytometry data. M.R. performed qRT-PCR, C.M. performed lung function tests and assisted in the analysis of ELISPOT data, and M.Y. performed mouse surgeries. M.L.M. and R.S.P. provided stocks of RSV line 19, as well as guidance regarding virus propagation and use in mice. S.E.W. analyzed data and edited the manuscript. A.R. designed experiments, analyzed data and wrote the manuscript. P.R. conceived of the study, designed experiments, analyzed data and wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–8 (PDF 1520 kb)
Rights and permissions
About this article
Cite this article
Krishnamoorthy, N., Khare, A., Oriss, T. et al. Early infection with respiratory syncytial virus impairs regulatory T cell function and increases susceptibility to allergic asthma. Nat Med 18, 1525–1530 (2012). https://doi.org/10.1038/nm.2896
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.2896
This article is cited by
-
Lung microbiome: new insights into the pathogenesis of respiratory diseases
Signal Transduction and Targeted Therapy (2024)
-
Treg plasticity and human diseases
Inflammation Research (2023)
-
Early life microbial exposures and allergy risks: opportunities for prevention
Nature Reviews Immunology (2021)
-
Evolutionary dynamics of group A and B respiratory syncytial virus in China, 2009-2018
Archives of Virology (2021)
-
Effects of Recombinant IL-35-BCG on Treg/Th17 Cell Imbalance and Inflammatory Response in Asthmatic Newborn Mice Induced by RSV
Inflammation (2021)