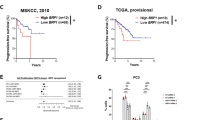
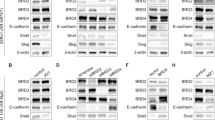
Abstract
Acquired resistance to anticancer treatments is a substantial barrier to reducing the morbidity and mortality that is attributable to malignant tumors. Components of tissue microenvironments are recognized to profoundly influence cellular phenotypes, including susceptibilities to toxic insults. Using a genome-wide analysis of transcriptional responses to genotoxic stress induced by cancer therapeutics, we identified a spectrum of secreted proteins derived from the tumor microenvironment that includes the Wnt family member wingless-type MMTV integration site family member 16B (WNT16B). We determined that WNT16B expression is regulated by nuclear factor of κ light polypeptide gene enhancer in B cells 1 (NF-κB) after DNA damage and subsequently signals in a paracrine manner to activate the canonical Wnt program in tumor cells. The expression of WNT16B in the prostate tumor microenvironment attenuated the effects of cytotoxic chemotherapy in vivo, promoting tumor cell survival and disease progression. These results delineate a mechanism by which genotoxic therapies given in a cyclical manner can enhance subsequent treatment resistance through cell nonautonomous effects that are contributed by the tumor microenvironment.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Accession codes
References
Longley, D.B. & Johnston, P.G. Molecular mechanisms of drug resistance. J. Pathol. 205, 275–292 (2005).
Wang, T.L. et al. Digital karyotyping identifies thymidylate synthase amplification as a mechanism of resistance to 5-fluorouracil in metastatic colorectal cancer patients. Proc. Natl. Acad. Sci. USA 101, 3089–3094 (2004).
Schmitt, C.A., Rosenthal, C.T. & Lowe, S.W. Genetic analysis of chemoresistance in primary murine lymphomas. Nat. Med. 6, 1029–1035 (2000).
Helmrich, A. et al. Recurrent chromosomal aberrations in INK4a/ARF defective primary lymphomas predict drug responses in vivo. Oncogene 24, 4174–4182 (2005).
Redmond, K.M., Wilson, T.R., Johnston, P.G. & Longley, D.B. Resistance mechanisms to cancer chemotherapy. Front. Biosci. 13, 5138–5154 (2008).
Wilson, T.R., Longley, D.B. & Johnston, P.G. Chemoresistance in solid tumours. Ann. Oncol. 17 (suppl. 10), x315–x324 (2006).
Lee, S. & Schmitt, C.A. Chemotherapy response and resistance. Curr. Opin. Genet. Dev. 13, 90–96 (2003).
Sakai, W. et al. Secondary mutations as a mechanism of cisplatin resistance in BRCA2-mutated cancers. Nature 451, 1116–1120 (2008).
Kobayashi, H. et al. Acquired multicellular-mediated resistance to alkylating agents in cancer. Proc. Natl. Acad. Sci. USA 90, 3294–3298 (1993).
Waldman, T. et al. Cell-cycle arrest versus cell death in cancer therapy. Nat. Med. 3, 1034–1036 (1997).
Samson, D.J., Seidenfeld, J., Ziegler, K. & Aronson, N. Chemotherapy sensitivity and resistance assays: a systematic review. J. Clin. Oncol. 22, 3618–3630 (2004).
Croix, B.S. et al. Reversal by hyaluronidase of adhesion-dependent multicellular drug resistance in mammary carcinoma cells. J. Natl. Cancer Inst. 88, 1285–1296 (1996).
Kerbel, R.S. Molecular and physiologic mechanisms of drug resistance in cancer: an overview. Cancer Metastasis Rev. 20, 1–2 (2001).
Wang, F. et al. Phenotypic reversion or death of cancer cells by altering signaling pathways in three-dimensional contexts. J. Natl. Cancer Inst. 94, 1494–1503 (2002).
Kim, J.J. & Tannock, I.F. Repopulation of cancer cells during therapy: an important cause of treatment failure. Nat. Rev. Cancer 5, 516–525 (2005).
Trédan, O., Galmarini, C.M., Patel, K. & Tannock, I.F. Drug resistance and the solid tumor microenvironment. J. Natl. Cancer Inst. 99, 1441–1454 (2007).
Garzotto, M., Myrthue, A., Higano, C.S. & Beer, T.M. Neoadjuvant mitoxantrone and docetaxel for high-risk localized prostate cancer. Urol. Oncol. 24, 254–259 (2006).
Beer, T.M. et al. Phase I study of weekly mitoxantrone and docetaxel before prostatectomy in patients with high-risk localized prostate cancer. Clin. Cancer Res. 10, 1306–1311 (2004).
Bavik, C. et al. The gene expression program of prostate fibroblast senescence modulates neoplastic epithelial cell proliferation through paracrine mechanisms. Cancer Res. 66, 794–802 (2006).
Coppé, J.P. et al. Senescence-associated secretory phenotypes reveal cell-nonautonomous functions of oncogenic RAS and the p53 tumor suppressor. PLoS Biol. 6, 2853–2868 (2008).
Clevers, H. Wnt/β-catenin signaling in development and disease. Cell 127, 469–480 (2006).
Binet, R. et al. WNT16B is a new marker of cellular senescence that regulates p53 activity and the phosphoinositide 3-kinase/AKT pathway. Cancer Res. 69, 9183–9191 (2009).
Acosta, J.C. et al. Chemokine signaling via the CXCR2 receptor reinforces senescence. Cell 133, 1006–1018 (2008).
Huang, S.M. et al. Tankyrase inhibition stabilizes axin and antagonizes Wnt signalling. Nature 461, 614–620 (2009).
Thiery, J.P., Acloque, H., Huang, R.Y. & Nieto, M.A. Epithelial-mesenchymal transitions in development and disease. Cell 139, 871–890 (2009).
Yook, J.I. et al. A Wnt-Axin2–GSK3β cascade regulates Snail1 activity in breast cancer cells. Nat. Cell Biol. 8, 1398–1406 (2006).
Vincan, E. & Barker, N. The upstream components of the Wnt signalling pathway in the dynamic EMT and MET associated with colorectal cancer progression. Clin. Exp. Metastasis 25, 657–663 (2008).
Wu, K. & Bonavida, B. The activated NF-κB–Snail–RKIP circuitry in cancer regulates both the metastatic cascade and resistance to apoptosis by cytotoxic drugs. Crit. Rev. Immunol. 29, 241–254 (2009).
Peinado, H., Olmeda, D. & Cano, A. Snail, Zeb and bHLH factors in tumour progression: an alliance against the epithelial phenotype? Nat. Rev. Cancer 7, 415–428 (2007).
Bernard, D. et al. Involvement of Rel/nuclear factor-κB transcription factors in keratinocyte senescence. Cancer Res. 64, 472–481 (2004).
Berchtold, C.M., Wu, Z.H., Huang, T.T. & Miyamoto, S. Calcium-dependent regulation of NEMO nuclear export in response to genotoxic stimuli. Mol. Cell. Biol. 27, 497–509 (2007).
Kuilman, T. et al. Oncogene-induced senescence relayed by an interleukin-dependent inflammatory network. Cell 133, 1019–1031 (2008).
Kuilman, T. & Peeper, D.S. Senescence-messaging secretome: SMS-ing cellular stress. Nat. Rev. Cancer 9, 81–94 (2009).
Fumagalli, M. & d'Adda di Fagagna, F. SASPense and DDRama in cancer and ageing. Nat. Cell Biol. 11, 921–923 (2009).
Michaloglou, C. et al. BRAFE600-associated senescence-like cell cycle arrest of human naevi. Nature 436, 720–724 (2005).
Xue, W. et al. Senescence and tumour clearance is triggered by p53 restoration in murine liver carcinomas. Nature 445, 656–660 (2007).
Wajapeyee, N., Serra, R.W., Zhu, X., Mahalingam, M. & Green, M.R. Oncogenic BRAF induces senescence and apoptosis through pathways mediated by the secreted protein IGFBP7. Cell 132, 363–374 (2008).
Coppé, J.P., Kauser, K., Campisi, J. & Beausejour, C.M. Secretion of vascular endothelial growth factor by primary human fibroblasts at senescence. J. Biol. Chem. 281, 29568–29574 (2006).
Gilbert, L.A. & Hemann, M.T. DNA damage–mediated induction of a chemoresistant niche. Cell 143, 355–366 (2010).
Biswas, S. et al. Inhibition of TGF-β with neutralizing antibodies prevents radiation-induced acceleration of metastatic cancer progression. J. Clin. Invest. 117, 1305–1313 (2007).
Davis, A.J. & Tannock, J.F. Repopulation of tumour cells between cycles of chemotherapy: a neglected factor. Lancet Oncol. 1, 86–93 (2000).
Meads, M.B., Hazlehurst, L.A. & Dalton, W.S. The bone marrow microenvironment as a tumor sanctuary and contributor to drug resistance. Clin. Cancer Res. 14, 2519–2526 (2008).
Shree, T. et al. Macrophages and cathepsin proteases blunt chemotherapeutic response in breast cancer. Genes Dev. 25, 2465–2479 (2011).
DeNardo, D.G. et al. Leukocyte complexity predicts breast cancer survival and functionally regulates response to chemotherapy. Cancer Discov. 1, 54–67 (2011).
Chien, Y. et al. Control of the senescence-associated secretory phenotype by NF-κB promotes senescence and enhances chemosensitivity. Genes Dev. 25, 2125–2136 (2011).
Alderton, P.M., Gross, J. & Green, M.D. Comparative study of doxorubicin, mitoxantrone, and epirubicin in combination with ICRF-187 (ADR-529) in a chronic cardiotoxicity animal model. Cancer Res. 52, 194–201 (1992).
Acknowledgements
We thank J. Dean and D. Bianchi-Frias for helpful comments, A. Moreno for administrative assistance and N. Clegg for bioinformatics support. S. Hayward, Vanderbilt University, and J. Ware, Medical College of Virginia, provided BPH1 and M12 cells, respectively. Primary human prostate (PSC27), ovarian (OVF28901) and breast (HBF1203) fibroblasts were provided by B. Knudsen, Cedars Sinai Medical Center, E. Swisher, University of Washington, and P. Porter through the Seattle Breast SPORE (P50 CA138293), Fred Hutchinson Cancer Research Center, respectively. B. Torok-Strorb, Fred Hutchinson Cancer Research Center, provided HS5 and HS27A HPV E6/E7 immortalized human bone marrow stromal cells. We thank the clinicians who participated in the trials of neoadjuvant chemotherapy: M. Garzotto, T. Takayama, P. Lange, W. Ellis, S. Lieberman and B.A. Lowe. We are also grateful for the participation of the patients and their families in these studies. Breast cancer specimens were obtained from the Fred Hutchinson Cancer Research Center/University of Washington Medical Center Breast Specimen Repository. We thank N. Urban, Fred Hutchinson Cancer Research Center, for providing ovarian cancer biospecimens funded through the POCRC SPORE grant P50CA83636. This work was supported by a fellowship from the Department of Defense (PC073217), R01CA119125, the National Cancer Institute Tumor Microenvironment Network U54126540, the Pacific Northwest Prostate Cancer SPORE P50CA097186 and the Prostate Cancer Foundation.
Author information
Authors and Affiliations
Contributions
Y.S. designed and conducted experiments, and wrote the manuscript. J.C. provided reagents and technical advice. C.H., T.M.B. and P.P. provided clinical materials for the assessments of treatment responses. I.C. analyzed data. L.T. analyzed tissue histology and immunohistochemical assays. P.S.N. designed experiments, analyzed data and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–7 and Supplementary Methods (PDF 10406 kb)
Rights and permissions
About this article
Cite this article
Sun, Y., Campisi, J., Higano, C. et al. Treatment-induced damage to the tumor microenvironment promotes prostate cancer therapy resistance through WNT16B. Nat Med 18, 1359–1368 (2012). https://doi.org/10.1038/nm.2890
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.2890
This article is cited by
-
Mitofusin 1 silencing decreases the senescent associated secretory phenotype, promotes immune cell recruitment and delays melanoma tumor growth after chemotherapy
Scientific Reports (2024)
-
Prognostic and therapeutic potential of senescent stromal fibroblasts in prostate cancer
Nature Reviews Urology (2024)
-
Residual ANTXR1+ myofibroblasts after chemotherapy inhibit anti-tumor immunity via YAP1 signaling pathway
Nature Communications (2024)
-
Mesoporous nanodrug delivery system: a powerful tool for a new paradigm of remodeling of the tumor microenvironment
Journal of Nanobiotechnology (2023)
-
PDK4-dependent hypercatabolism and lactate production of senescent cells promotes cancer malignancy
Nature Metabolism (2023)