Abstract
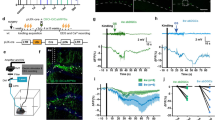
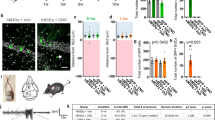
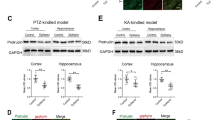
Temporal lobe epilepsy (TLE) is accompanied by an abnormal location of granule cells in the dentate gyrus. Using a rat model of complex febrile seizures, which are thought to be a precipitating insult of TLE later in life, we report that aberrant migration of neonatal-generated granule cells results in granule cell ectopia that persists into adulthood. Febrile seizures induced an upregulation of GABAA receptors (GABAA-Rs) in neonatally generated granule cells, and hyperactivation of excitatory GABAA-Rs caused a reversal in the direction of granule cell migration. This abnormal migration was prevented by RNAi-mediated knockdown of the Na+K+2Cl− co-transporter (NKCC1), which regulates the excitatory action of GABA. NKCC1 inhibition with bumetanide after febrile seizures rescued the granule cell ectopia, susceptibility to limbic seizures and development of epilepsy. Thus, this work identifies a previously unknown pathogenic role of excitatory GABAA-R signaling and highlights NKCC1 as a potential therapeutic target for preventing granule cell ectopia and the development of epilepsy after febrile seizures.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Chevassus-au-Louis, N., Baraban, S.C., Gaiarsa, J.L. & Ben Ari, Y. Cortical malformations and epilepsy: new insights from animal models. Epilepsia 40, 811–821 (1999).
Jacobs, K.M., Kharazia, V.N. & Prince, D.A. Mechanisms underlying epileptogenesis in cortical malformations. Epilepsy Res. 36, 165–188 (1999).
Lee, K.S. et al. A genetic animal model of human neocortical heterotopia associated with seizures. J. Neurosci. 17, 6236–6242 (1997).
Chevassus-Au-Louis, N., Rafiki, A., Jorquera, I., Ben Ari, Y. & Represa, A. Neocortex in the hippocampus: an anatomical and functional study of CA1 heterotopias after prenatal treatment with methylazoxymethanol in rats. J. Comp. Neurol. 394, 520–536 (1998).
Roper, S.N., Gilmore, R.L. & Houser, C.R. Experimentally induced disorders of neuronal migration produce an increased propensity for electrographic seizures in rats. Epilepsy Res. 21, 205–219 (1995).
Manent, J.B., Wang, Y., Chang, Y., Paramasivam, M. & LoTurco, J.J. Dcx reexpression reduces subcortical band heterotopia and seizure threshold in an animal model of neuronal migration disorder. Nat. Med. 15, 84–90 (2009).
Houser, C.R. Granule cell dispersion in the dentate gyrus of humans with temporal lobe epilepsy. Brain Res. 535, 195–204 (1990).
Lurton, D., El Bahh, B., Sundstrom, L. & Rougier, A. Granule cell dispersion is correlated with early epileptic events in human temporal lobe epilepsy. J. Neurol. Sci. 154, 133–136 (1998).
Parent, J.M. & Murphy, G.G. Mechanisms and functional significance of aberrant seizure-induced hippocampal neurogenesis. Epilepsia 49 (suppl. 5), 19–25 (2008).
Scharfman, H., Goodman, J. & McCloskey, D. Ectopic granule cells of the rat dentate gyrus. Dev. Neurosci. 29, 14–27 (2007).
Riban, V. et al. Evolution of hippocampal epileptic activity during the development of hippocampal sclerosis in a mouse model of temporal lobe epilepsy. Neuroscience 112, 101–111 (2002).
Scharfman, H.E., Goodman, J.H. & Sollas, A.L. Granule-like neurons at the hilar/CA3 border after status epilepticus and their synchrony with area CA3 pyramidal cells: functional implications of seizure-induced neurogenesis. J. Neurosci. 20, 6144–6158 (2000).
Scharfman, H.E., Sollas, A.L. & Goodman, J.H. Spontaneous recurrent seizures after pilocarpine-induced status epilepticus activate calbindin-immunoreactive hilar cells of the rat dentate gyrus. Neuroscience 111, 71–81 (2002).
Hauser, W.A. The prevalence and incidence of convulsive disorders in children. Epilepsia 35 (suppl. 2), S1–S6 (1994).
Berg, A.T. & Shinnar, S. Complex febrile seizures. Epilepsia 37, 126–133 (1996).
Nelson, K.B. & Ellenberg, J.H. Predictors of epilepsy in children who have experienced febrile seizures. N. Engl. J. Med. 295, 1029–1033 (1976).
Cendes, F. et al. Early childhood prolonged febrile convulsions, atrophy and sclerosis of mesial structures, and temporal lobe epilepsy: an MRI volumetric study. Neurology 43, 1083–1087 (1993).
French, J.A. et al. Characteristics of medial temporal lobe epilepsy: I. Results of history and physical examination. Ann. Neurol. 34, 774–780 (1993).
Bender, R.A., Dubé, C. & Baram, T.Z. Febrile seizures and mechanisms of epileptogenesis: insights from an animal model. Adv. Exp. Med. Biol. 548, 213–225 (2004).
Koyama, R. & Matsuki, N. Novel etiological and therapeutic strategies for neurodiseases: mechanisms and consequences of febrile seizures: lessons From animal models. J. Pharmacol. Sci. 113, 14–22 (2010).
Chen, K. et al. Persistently modified h-channels after complex febrile seizures convert the seizure-induced enhancement of inhibition to hyperexcitability. Nat. Med. 7, 331–337 (2001).
Dubé, C. et al. Prolonged febrile seizures in the immature rat model enhance hippocampal excitability long term. Ann. Neurol. 47, 336–344 (2000).
Dubé, C. et al. Temporal lobe epilepsy after experimental prolonged febrile seizures: prospective analysis. Brain 129, 911–922 (2006).
Muramatsu, R., Ikegaya, Y., Matsuki, N. & Koyama, R. Early-life status epilepticus induces ectopic granule cells in adult mice dentate gyrus. Exp. Neurol. 211, 503–510 (2008).
Muramatsu, R., Ikegaya, Y., Matsuki, N. & Koyama, R. Neonatally born granule cells numerically dominate adult mice dentate gyrus. Neuroscience 148, 593–598 (2007).
Behar, T.N. et al. GABA stimulates chemotaxis and chemokinesis of embryonic cortical neurons via calcium-dependent mechanisms. J. Neurosci. 16, 1808–1818 (1996).
Ben-Ari, Y. Excitatory actions of gaba during development: the nature of the nurture. Nat. Rev. Neurosci. 3, 728–739 (2002).
Bolteus, A.J. & Bordey, A. GABA release and uptake regulate neuronal precursor migration in the postnatal subventricular zone. J. Neurosci. 24, 7623–7631 (2004).
Heck, N. et al. GABA-A receptors regulate neocortical neuronal migration in vitro and in vivo. Cereb. Cortex 17, 138–148 (2007).
Manent, J.B. et al. A noncanonical release of GABA and glutamate modulates neuronal migration. J. Neurosci. 25, 4755–4765 (2005).
Eckenhoff, M.F. & Rakic, P. Radial organization of the hippocampal dentate gyrus: a Golgi, ultrastructural, and immunocytochemical analysis in the developing rhesus monkey. J. Comp. Neurol. 223, 1–21 (1984).
Frotscher, M., Haas, C.A. & Forster, E. Reelin controls granule cell migration in the dentate gyrus by acting on the radial glial scaffold. Cereb. Cortex 13, 634–640 (2003).
Tozuka, Y., Fukuda, S., Namba, T., Seki, T. & Hisatsune, T. GABAergic excitation promotes neuronal differentiation in adult hippocampal progenitor cells. Neuron 47, 803–815 (2005).
Hatanaka, Y. & Murakami, F. In vitro analysis of the origin, migratory behavior, and maturation of cortical pyramidal cells. J. Comp. Neurol. 454, 1–14 (2002).
Wang, D.D., Krueger, D.D. & Bordey, A. GABA depolarizes neuronal progenitors of the postnatal subventricular zone via GABAA receptor activation. J. Physiol. (Lond.) 550, 785–800 (2003).
Guan, C.B., Xu, H.T., Jin, M., Yuan, X.B. & Poo, M.M. Long-range Ca2+ signaling from growth cone to soma mediates reversal of neuronal migration induced by slit-2. Cell 129, 385–395 (2007).
Komuro, H. & Rakic, P. Intracellular Ca2+ fluctuations modulate the rate of neuronal migration. Neuron 17, 275–285 (1996).
Fukuda, A. et al. Changes in intracellular Ca2+ induced by GABAA receptor activation and reduction in Cl− gradient in neonatal rat neocortex. J. Neurophysiol. 79, 439–446 (1998).
Payne, J.A., Rivera, C., Voipio, J. & Kaila, K. Cation-chloride co-transporters in neuronal communication, development and trauma. Trends Neurosci. 26, 199–206 (2003).
Ge, S. et al. GABA regulates synaptic integration of newly generated neurons in the adult brain. Nature 439, 589–593 (2006).
Peng, Z. & Houser, C.R. Temporal patterns of fos expression in the dentate gyrus after spontaneous seizures in a mouse model of temporal lobe epilepsy. J. Neurosci. 25, 7210–7220 (2005).
McCloskey, D.P., Hintz, T.M., Pierce, J.P. & Scharfman, H.E. Stereological methods reveal the robust size and stability of ectopic hilar granule cells after pilocarpine-induced status epilepticus in the adult rat. Eur. J. Neurosci. 24, 2203–2210 (2006).
Ibarz, J.M., Foffani, G., Cid, E., Inostroza, M. & Menendez, P. Emergent dynamics of fast ripples in the epileptic hippocampus. J. Neurosci. 30, 16249–16261 (2010).
Bragin, A., Engel, J. Jr., Wilson, C.L., Fried, I. & Mathern, G.W. Hippocampal and entorhinal cortex high-frequency oscillations (100–500 Hz) in human epileptic brain and in kainic acid-treated rats with chronic seizures. Epilepsia 40, 127–137 (1999).
Worrell, G.A. et al. High-frequency oscillations in human temporal lobe: simultaneous microwire and clinical macroelectrode recording. Brain 131, 928–937 (2008).
Engel, J. Jr., Bragin, A., Staba, R. & Mody, I. High-frequency oscillations: what is normal and what is not? Epilepsia 50, 598–604 (2009).
Behar, T.N., Schaffner, A.E., Scott, C.A., Greene, C.L. & Barker, J.L. GABA receptor antagonists modulate postmitotic cell migration in slice cultures of embryonic rat cortex. Cereb. Cortex 10, 899–909 (2000).
Förster, E., Zhao, S. & Frotscher, M. Laminating the hippocampus. Nat. Rev. Neurosci. 7, 259–267 (2006).
Safiulina, V.F., Fattorini, G., Conti, F. & Cherubini, E. GABAergic signaling at mossy fiber synapses in neonatal rat hippocampus. J. Neurosci. 26, 597–608 (2006).
Romo-Parra, H., Vivar, C., Maqueda, J., Morales, M.A. & Gutierrez, R. Activity-dependent induction of multitransmitter signaling onto pyramidal cells and interneurons of hippocampal area CA3. J. Neurophysiol. 89, 3155–3167 (2003).
Sullivan, J.E., Witte, M.K., Yamashita, T.S., Myers, C.M. & Blumer, J.L. Pharmacokinetics of bumetanide in critically ill infants. Clin. Pharmacol. Ther. 60, 405–413 (1996).
Dzhala, V.I. et al. NKCC1 transporter facilitates seizures in the developing brain. Nat. Med. 11, 1205–1213 (2005).
Kahle, K.T., Barnett, S.M., Sassower, K.C. & Staley, K.J. Decreased seizure activity in a human neonate treated with bumetanide, an inhibitor of the Na+-K+-2Cl− cotransporter NKCC1. J. Child Neurol. 24, 572–576 (2009).
Dyhrfjeld-Johnsen, J., Morgan, R.J., Foldy, C. & Soltesz, I. Upregulated H-current in hyperexcitable CA1 dendrites after febrile seizures. Front. Cell Neurosci. 2, 2 (2008).
Koyama, R. et al. A low-cost method for brain slice cultures. J Pharmacol. Sci. 104, 191–194 (2007).
Yamada, R.X. et al. Long-range axonal calcium sweep induces axon retraction. J. Neurosci. 28, 4613–4618 (2008).
Sasaki, T., Matsuki, N. & Ikegaya, Y. Action-potential modulation during axonal conduction. Science 331, 599–601 (2011).
Patel, S., Meldrum, B.S. & Fine, A. Susceptibility to pilocarpine-induced seizures in rats increases with age. Behav. Brain Res. 31, 165–167 (1988).
Acknowledgements
This work was supported by a Grant-in-Aid for Science Research on Young Scientists (B) (19790048) and on Innovative Areas 'Mesoscopic Neurocircuitry' (22115003) from The Ministry of Education, Culture, Sports, Science and Technology of Japan; the Research Foundation for Pharmaceutical Sciences; and the Funding Program for Next Generation World-Leading Researchers (LS023).
Author information
Authors and Affiliations
Contributions
R.K. designed the project, conducted and analyzed the experiments, and wrote the manuscript. K.T. did in vivo electrophysiology, long-term video monitoring and helped with preparation of retroviruses. T.S. did in vitro electrophysiology and time-lapse imaging in explant cultures. J.I. did injection of retroviruses. D.M. helped with in vivo electrophysiology. R.M. helped with perfusion of mice. N.M. and Y.I. are senior authors and were responsible for project planning. All authors analyzed and discussed the results and commented on the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Methods and Supplementary Figures 1–9 (PDF 1062 kb)
Supplementary Video 1
Radial migration of a GFP+ GC (Supplementary Fig. 4a). The images were acquired every 1 h for 24 h during the 3 to 4 DIV. (MOV 349 kb)
Supplementary Video 2
Somal translocation of a GFP+ GC (Supplementary Fig. 4b). The images were acquired every 1 h for 24 h during the 3 to 4 DIV. (MOV 343 kb)
Supplementary Video 3
Aberrant migration of a GC from a HT rat in Figure 3c. The images were acquired every 1 h for 6 h beginning at 3 DIV. (MOV 92 kb)
Supplementary Video 4
A migrating GC from the explant culture prepared from a NT rat (Fig. 4b, left). The images were acquired at 1 frame per 2 min for 60 min. Muscimol was focally applied to the leading growth cone for 1 min at the time point of 0 min. (MOV 545 kb)
Supplementary Video 5
A migrating GC from the explant culture prepared from a HT rat (Fig. 4b, right). The images were acquired at 1 frame per 2 min for 60 min. Muscimol was focally applied to the leading growth cone for 1 min at the time point of 0 min. (MOV 261 kb)
Rights and permissions
About this article
Cite this article
Koyama, R., Tao, K., Sasaki, T. et al. GABAergic excitation after febrile seizures induces ectopic granule cells and adult epilepsy. Nat Med 18, 1271–1278 (2012). https://doi.org/10.1038/nm.2850
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.2850
This article is cited by
-
Molecular Mechanisms of Epilepsy: The Role of the Chloride Transporter KCC2
Journal of Molecular Neuroscience (2022)
-
Hippocampal granule cell dispersion: a non-specific finding in pediatric patients with no history of seizures
Acta Neuropathologica Communications (2020)
-
Non-radial tortuous migration with cell polarity alterations of newly generated granule neurons in the neonatal rat dentate gyrus
Brain Structure and Function (2019)
-
GABAa excitation and synaptogenesis after Status Epilepticus – A computational study
Scientific Reports (2018)
-
Canonical Transient Receptor Potential Channel 3 Contributes to Febrile Seizure Inducing Neuronal Cell Death and Neuroinflammation
Cellular and Molecular Neurobiology (2018)