Abstract
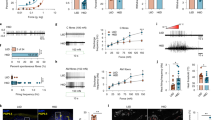
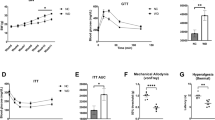
This study establishes a mechanism for metabolic hyperalgesia based on the glycolytic metabolite methylglyoxal. We found that concentrations of plasma methylglyoxal above 600 nM discriminate between diabetes-affected individuals with pain and those without pain. Methylglyoxal depolarizes sensory neurons and induces post-translational modifications of the voltage-gated sodium channel Nav1.8, which are associated with increased electrical excitability and facilitated firing of nociceptive neurons, whereas it promotes the slow inactivation of Nav1.7. In mice, treatment with methylglyoxal reduces nerve conduction velocity, facilitates neurosecretion of calcitonin gene-related peptide, increases cyclooxygenase-2 (COX-2) expression and evokes thermal and mechanical hyperalgesia. This hyperalgesia is reflected by increased blood flow in brain regions that are involved in pain processing. We also found similar changes in streptozotocin-induced and genetic mouse models of diabetes but not in Nav1.8 knockout (Scn10−/−) mice. Several strategies that include a methylglyoxal scavenger are effective in reducing methylglyoxal- and diabetes-induced hyperalgesia. This previously undescribed concept of metabolically driven hyperalgesia provides a new basis for the design of therapeutic interventions for painful diabetic neuropathy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Change history
06 July 2012
In the version of this article initially published, in Figure 1a, the bar for 3-deoxyglucosone was incorrectly labeled as 3-diacylglycerol. In Figure 6b, the labels for the control and methylglyoxal-treated samples were incorrectly switched. Also, Felix Lasitschka’s last name was misspelled as Lasischka. The errors have been corrected in the HTML and PDF versions of the article.
References
Tavee, J. & Zhou, L. Small fiber neuropathy: a burning problem. Cleve. Clin. J. Med. 76, 297–305 (2009).
Nawroth, P.P., Rudofsky, G. & Humpert, P.M. Have we understood diabetes? New tasks for diagnosis and therapy. Exp. Clin. Endocrinol. Diabetes 118, 1–3 (2010).
Calcutt, N.A. Potential mechanisms of neuropathic pain in diabetes. Int. Rev. Neurobiol. 50, 205–228 (2002).
Thornalley, P.J. Dicarbonyl intermediates in the maillard reaction. Ann. NY Acad. Sci. 1043, 111–117 (2005).
Brownlee, M. Biochemistry and molecular cell biology of diabetic complications. Nature 414, 813–820 (2001).
Karachalias, N., Babaei-Jadidi, R., Ahmed, N. & Thornalley, P.J. Accumulation of fructosyl-lysine and advanced glycation end products in the kidney, retina and peripheral nerve of streptozotocin-induced diabetic rats. Biochem. Soc. Trans. 31, 1423–1425 (2003).
Thornalley, P.J. Glyoxalase I—structure, function and a critical role in the enzymatic defence against glycation. Biochem. Soc. Trans. 31, 1343–1348 (2003).
Bierhaus, A. & Nawroth, P.P. Multiple levels of regulation determine the role of the receptor for AGE (RAGE) as common soil in inflammation, immune responses and diabetes mellitus and its complications. Diabetologia 52, 2251–2263 (2009).
Jack, M.M., Ryals, J.M. & Wright, D.E. Characterisation of glyoxalase I in a streptozocin-induced mouse model of diabetes with painful and insensate neuropathy. Diabetologia 54, 2174–2182 (2011).
Morcos, M. et al. Glyoxalase-1 prevents mitochondrial protein modification and enhances lifespan in Caenorhabditis elegans. Aging Cell 7, 260–269 (2008).
Suzuki, Y., Sato, J., Kawanishi, M. & Mizumura, K. Lowered response threshold and increased responsiveness to mechanical stimulation of cutaneous nociceptive fibers in streptozotocin-diabetic rat skin in vitro—correlates of mechanical allodynia and hyperalgesia observed in the early stage of diabetes. Neurosci. Res. 43, 171–178 (2002).
Misawa, S. et al. Axonal potassium conductance and glycemic control in human diabetic nerves. Clin. Neurophysiol. 116, 1181–1187 (2005).
Grafe, P., Bostock, H. & Schneider, U. The effects of hyperglycaemic hypoxia on rectification in rat dorsal root axons. J. Physiol. (Lond.) 480, 297–307 (1994).
Quasthoff, S. The role of axonal ion conductances in diabetic neuropathy: a review. Muscle Nerve 21, 1246–1255 (1998).
Craner, M.J., Klein, J.P., Renganathan, M., Black, J.A. & Waxman, S.G. Changes of sodium channel expression in experimental painful diabetic neuropathy. Ann. Neurol. 52, 786–792 (2002).
Hong, S., Morrow, T.J., Paulson, P.E., Isom, L.L. & Wiley, J.W. Early painful diabetic neuropathy is associated with differential changes in tetrodotoxin-sensitive and -resistant sodium channels in dorsal root ganglion neurons in the rat. J. Biol. Chem. 279, 29341–29350 (2004).
Shah, B.S. et al. Beta3, a novel auxiliary subunit for the voltage gated sodium channel is upregulated in sensory neurones following streptozocin induced diabetic neuropathy in rat. Neurosci. Lett. 309, 1–4 (2001).
Lampert, A., O'Reilly, A.O., Reeh, P. & Leffler, A. Sodium channelopathies and pain. Pflugers Arch. 460, 249–263 (2010).
Silos-Santiago, I. The role of tetrodotoxin-resistant sodium channels in pain states: are they the next target for analgesic drugs? Curr. Opin. Investig. Drugs 9, 83–89 (2008).
Hirade, M., Yasuda, H., Omatsu-Kanbe, M., Kikkawa, R. & Kitasato, H. Tetrodotoxin-resistant sodium channels of dorsal root ganglion neurons are readily activated in diabetic rats. Neuroscience 90, 933–939 (1999).
Konrade, I. et al. Verminderte glyoxalase-1 aktivitat bei patienten mit schmerzhafter diabetischer neuropathie. Diabetologie und Stoffwechsel 2, P98 (2007).
Thornalley, P.J. et al. Antitumour activity of S-p-bromobenzylglutathione cyclopentyl diester in vitro and in vivo. Inhibition of glyoxalase I and induction of apoptosis. Biochem. Pharmacol. 51, 1365–1372 (1996).
El-Osta, A. et al. Transient high glucose causes persistent epigenetic changes and altered gene expression during subsequent normoglycemia. J. Exp. Med. 205, 2409–2417 (2008).
Sullivan, K.A. et al. Mouse models of diabetic neuropathy. Neurobiol. Dis. 28, 276–285 (2007).
Kellogg, A.P., Cheng, H.T. & Pop-Busui, R. Cyclooxygenase-2 pathway as a potential therapeutic target in diabetic peripheral neuropathy. Curr. Drug Targets 9, 68–76 (2008).
Hong, S., Agresta, L., Guo, C. & Wiley, J.W. The TRPV1 receptor is associated with preferential stress in large dorsal root ganglion neurons in early diabetic sensory neuropathy. J. Neurochem. 105, 1212–1222 (2008).
Morrow, T.J., Paulson, P.E., Danneman, P.J. & Casey, K.L. Regional changes in forebrain activation during the early and late phase of formalin nociception: analysis using cerebral blood flow in the rat. Pain 75, 355–365 (1998).
Paulson, P.E., Wiley, J.W. & Morrow, T.J. Concurrent activation of the somatosensory forebrain and deactivation of periaqueductal gray associated with diabetes-induced neuropathic pain. Exp. Neurol. 208, 305–313 (2007).
Renganathan, M., Cummins, T.R. & Waxman, S.G. Contribution of Nav1.8 sodium channels to action potential electrogenesis in DRG neurons. J. Neurophysiol. 86, 629–640 (2001).
Fuchs, D., Birklein, F., Reeh, P.W. & Sauer, S.K. Sensitized peripheral nociception in experimental diabetes of the rat. Pain 151, 496–505 (2010).
Obrosova, I.G. Diabetic painful and insensate neuropathy: pathogenesis and potential treatments. Neurotherapeutics 6, 638–647 (2009).
Sandkühler, J. Models and mechanisms of hyperalgesia and allodynia. Physiol. Rev. 89, 707–758 (2009).
Rush, A.M., Cummins, T.R. & Waxman, S.G. Multiple sodium channels and their roles in electrogenesis within dorsal root ganglion neurons. J. Physiol. (Lond.) 579, 1–14 (2007).
Rush, A.M. et al. A single sodium channel mutation produces hyper- or hypoexcitability in different types of neurons. Proc. Natl. Acad. Sci. USA 103, 8245–8250 (2006).
Ahmed, N., Dobler, D., Dean, M. & Thornalley, P.J. Peptide mapping identifies hotspot site of modification in human serum albumin by methylglyoxal involved in ligand binding and esterase activity. J. Biol. Chem. 280, 5724–5732 (2005).
Bierhaus, A. et al. Loss of pain perception in diabetes is dependent on a receptor of the immunoglobulin superfamily. J. Clin. Invest. 114, 1741–1751 (2004).
Menéndez, L., Lastra, A., Hidalgo, A. & Baamonde, A. Unilateral hot plate test: a simple and sensitive method for detecting central and peripheral hyperalgesia in mice. J. Neurosci. Methods 113, 91–97 (2002).
Hargreaves, K., Dubner, R., Brown, F., Flores, C. & Joris, J. A new and sensitive method for measuring thermal nociception in cutaneous hyperalgesia. Pain 32, 77–88 (1988).
D'Amour, F.E., Erickson, B.R. & Smith, D.L. Effect of environmental temperature in traumatic shock. Proc. Soc. Exp. Biol. Med. 94, 1–4 (1957).
Vareniuk, I., Pavlov, I.A. & Obrosova, I.G. Inducible nitric oxide synthase gene deficiency counteracts multiple manifestations of peripheral neuropathy in a streptozotocin-induced mouse model of diabetes. Diabetologia 51, 2126–2133 (2008).
Miyoshi, T. & Goto, I. Serial in vivo determinations of nerve conduction velocity in rat tails. Physiological and pathological changes. Electroencephalogr. Clin. Neurophysiol. 35, 125–131 (1973).
Morrow, T.J., Paulson, P.E., Brewer, K.L., Yezierski, R.P. & Casey, K.L. Chronic, selective forebrain responses to excitotoxic dorsal horn injury. Exp. Neurol. 161, 220–226 (2000).
Zhang, Y. et al. Intravenous somatic gene transfer with antisense tissue factor restores blood flow by reducing tumor necrosis factor-induced tissue factor expression and fibrin deposition in mouse meth-A sarcoma. J. Clin. Invest. 97, 2213–2224 (1996).
Stucky, C.L. & Lewin, G.R. Isolectin B(4)-positive and -negative nociceptors are functionally distinct. J. Neurosci. 19, 6497–6505 (1999).
Bierhaus, A. et al. Diabetes-associated sustained activation of the transcription factor nuclear factor-κB. Diabetes. 50, 2792–2808 (2001).
Humpert, P.M. et al. External electric muscle stimulation improves burning sensations and sleeping disturbances in patients with type 2 diabetes and symptomatic neuropathy. Pain Med. 10, 413–419 (2009).
Acknowledgements
The authors thank S. Kaymak for immunohistochemistry, A. Buhl and K. Leotta for assistance in the cerebral blood flow experiments, L. Werner for support with bregma gradings and X. Du and A. Erhardt for the daily care of the mice. A. Bierhaus, J.F., M.E.C., M.B. and P.P.N. were supported by a Centre grant from the Juvenile Diabetes Research Foundation (JDRF). This work was also supported in part by grants from the Deutsche Forschungsgemeinschaft (BI-1281/3-1 to A. Bierhaus; LU728/3-1 and NA-350/3-2 in KFO 130 to P.W.R., C. Nau and A.L.), the US National Institutes of Health (2R56DK33861-21 to M.B.), the European Foundation for the Study of Diabetes (EFSD/Lilly-Programme, to A. Bierhaus), the German Diabetes Association (DDG, Christian-Hagedorn-Award to A. Bierhaus), the Manfred-Lautenschläger-Stiftung for Diabetes (LSD, to P.P.N.), the Dietmar-Hopp-Stiftung (to A. Bierhaus, P.M.H. and P.P.N.) and the Network Aging Research (NAR, to A. Bierhaus). A. Babes and C.N. were supported by grant PN2 164/2007 from the Romanian Research Council (CNCSIS). A. Babes also received support from the Alexander von Humboldt Foundation.
Author information
Authors and Affiliations
Contributions
A. Bierhaus planned, performed and supervised all experiments, was responsible for the data interpretation and wrote the manuscript. T.F. and S.S. performed most of the mouse experiments, and T.F. also did the biochemical analytics and cell culture experiments. A.L. and C. Nau performed the voltage clamp studies and were involved in data interpretation. A. Babes and C. Neacsu performed the current clamp studies and were involved in data interpretation. S.K.S., M.E. and T.I.K. performed CGRP-release experiments and single-fiber recordings in the skin-nerve preparation and were involved in data interpretation. M. Schnölzer, N.R. and P.J.T. were involved in the dicarbonyl analytics and data interpretation. F.L. provided biopsies of human sciatic nerves. W.L.N. performed electron microscopy. R.E. and I.K.L. performed the nerve conduction velocity experiments and the measurements of tactile allodynia. W.M., M. Schwaninger and U.H. supervised the cerebral blood flow measurements and were involved in data interpretation. T.D. was involved in the isolation and characterization of the DRG. D.E. generated the Glo1−/+ mice. J.F. and M.E.C. provided ALT-711 and were involved in data interpretation. I.K., V.P. and M.M. performed the clinical studies. P.M.H. supervised the clinical studies and was involved in data interpretation. D.M.S. was involved in data interpretation and the writing of the manuscript. D.Z. provided the human skin biopsies and was involved in data interpretation. M.B. provided the Glo1−/+ mice and was involved in data interpretation and the writing of the manuscript. P.W.R. supervised the electrophysiological part of the project and wrote the manuscript. P.P.N. supervised the project and wrote the manuscript. Senior coauthorship is shared by P.W.R. and P.P.N.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–8, Supplementary Tables 1 and 2 and Supplementary Methods (PDF 819 kb)
Rights and permissions
About this article
Cite this article
Bierhaus, A., Fleming, T., Stoyanov, S. et al. Methylglyoxal modification of Nav1.8 facilitates nociceptive neuron firing and causes hyperalgesia in diabetic neuropathy. Nat Med 18, 926–933 (2012). https://doi.org/10.1038/nm.2750
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.2750
This article is cited by
-
Methylglyoxal Induced Modifications to Stabilize Therapeutic Proteins: A Review
The Protein Journal (2024)
-
Glyoxal in hyperglycaemic ischemic stroke – a cohort study
Cardiovascular Diabetology (2023)
-
Reconsidering the role of protein glycation in disease
Nature Chemical Biology (2023)
-
Higher fasting blood glucose worsens knee symptoms in patients with radiographic knee osteoarthritis and comorbid central sensitization: an Iwaki cohort study
Arthritis Research & Therapy (2022)
-
CEBPβ regulation of endogenous IGF-1 in adult sensory neurons can be mobilized to overcome diabetes-induced deficits in bioenergetics and axonal outgrowth
Cellular and Molecular Life Sciences (2022)