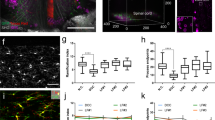
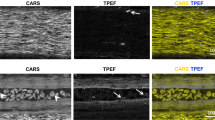
Abstract
Studying regeneration in the central nervous system (CNS) is hampered by current histological and imaging techniques because they provide only partial information about axonal and glial reactions. Here we developed a tetrahydrofuran-based clearing procedure that renders fixed and unsectioned adult CNS tissue transparent and fully penetrable for optical imaging. In large spinal cord segments, we imaged fluorescently labeled cells by 'ultramicroscopy' and two-photon microscopy without the need for histological sectioning. We found that more than a year after injury growth-competent axons regenerated abundantly through the injury site. A few growth-incompetent axons could also regenerate when they bypassed the lesion. Moreover, we accurately determined quantitative changes of glial cells after spinal cord injury. Thus, clearing CNS tissue enables an unambiguous evaluation of axon regeneration and glial reactions. Our clearing procedure also renders other organs transparent, which makes this approach useful for a large number of preclinical paradigms.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Chen, M.S. et al. Nogo-A is a myelin-associated neurite outgrowth inhibitor and an antigen for monoclonal antibody IN-1. Nature 403, 434–439 (2000).
Bradbury, E.J. et al. Chondroitinase ABC promotes functional recovery after spinal cord injury. Nature 416, 636–640 (2002).
GrandPré, T., Li, S. & Strittmatter, S.M. Nogo-66 receptor antagonist peptide promotes axonal regeneration. Nature 417, 547–551 (2002).
Neumann, S. & Woolf, C.J. Regeneration of dorsal column fibers into and beyond the lesion site following adult spinal cord injury. Neuron 23, 83–91 (1999).
Ramer, M.S., Priestley, J.V. & McMahon, S.B. Functional regeneration of sensory axons into the adult spinal cord. Nature 403, 312–316 (2000).
Steward, O., Zheng, B. & Tessier-Lavigne, M. False resurrections: distinguishing regenerated from spared axons in the injured central nervous system. J. Comp. Neurol. 459, 1–8 (2003).
Silver, J. & Miller, J.H. Regeneration beyond the glial scar. Nat. Rev. Neurosci. 5, 146–156 (2004).
Labombarda, F. et al. Modulation of NADPH-diaphorase and glial fibrillary acidic protein by progesterone in astrocytes from normal and injured rat spinal cord. J. Steroid Biochem. Mol. Biol. 73, 159–169 (2000).
Wang, L. et al. Glial and axonal responses in areas of Wallerian degeneration of the corticospinal and dorsal ascending tracts after spinal cord dorsal funiculotomy. Neuropathology 29, 230–241 (2009).
Stuart, D.A. & Oorschot, D.E. Embedding, sectioning, immunocytochemical and stereological methods that optimise research on the lesioned adult rat spinal cord. J. Neurosci. Methods 61, 5–14 (1995).
Ylera, B. et al. Chronically CNS-injured adult sensory neurons gain regenerative competence upon a lesion of their peripheral axon. Curr. Biol. 19, 930–936 (2009).
Kerschensteiner, M., Schwab, M.E., Lichtman, J.W. & Misgeld, T. In vivo imaging of axonal degeneration and regeneration in the injured spinal cord. Nat. Med. 11, 572–577 (2005).
Enes, J. et al. Electrical activity suppresses axon growth through Cav1.2 channels in adult primary sensory neurons. Curr. Biol. 20, 1154–1164 (2010).
Spalteholz, W. Über das Durchsichtigmachen von menschlichen und tierischen Präparaten (S. Hierzel, Leipzig, Germany, 1914).
Dodt, H.U. et al. Ultramicroscopy: three-dimensional visualization of neuronal networks in the whole mouse brain. Nat. Methods 4, 331–336 (2007).
Feng, G. et al. Imaging neuronal subsets in transgenic mice expressing multiple spectral variants of GFP. Neuron 28, 41–51 (2000).
Ertürk, A., Hellal, F., Enes, J. & Bradke, F. Disorganized microtubules underlie the formation of retraction bulbs and the failure of axonal regeneration. J. Neurosci. 27, 9169–9180 (2007).
Faulkner, J.R. et al. Reactive astrocytes protect tissue and preserve function after spinal cord injury. J. Neurosci. 24, 2143–2155 (2004).
Jung, S. et al. Analysis of fractalkine receptor CX(3)CR1 function by targeted deletion and green fluorescent protein reporter gene insertion. Mol. Cell. Biol. 20, 4106–4114 (2000).
Nolte, C. et al. GFAP promoter–controlled EGFP-expressing transgenic mice: a tool to visualize astrocytes and astrogliosis in living brain tissue. Glia 33, 72–86 (2001).
Neher, R. & Neher, E. Optimizing imaging parameters for the separation of multiple labels in a fluorescence image. J. Microsc. 213, 46–62 (2004).
Donald Starr, R.M.H. Tetrahydrofuran. Org. Synth. 2, 566 (1943).
Díaz, R.S., Monreal, J., Regueiro, P. & Lucas, M. Preparation of a protein-free total brain white matter lipid fraction: characterization of liposomes. J. Neurosci. Res. 31, 136–145 (1992).
Freund, P. et al. Nogo-A–specific antibody treatment enhances sprouting and functional recovery after cervical lesion in adult primates. Nat. Med. 12, 790–792 (2006).
Fouad, K., Klusman, I. & Schwab, M.E. Regenerating corticospinal fibers in the Marmoset (Callitrix jacchus) after spinal cord lesion and treatment with the anti–Nogo-A antibody IN-1. Eur. J. Neurosci. 20, 2479–2482 (2004).
Lichtman, J.W. & Sanes, J.R. Ome sweet ome: what can the genome tell us about the connectome? Curr. Opin. Neurobiol. 18, 346–353 (2008).
Zong, H., Espinosa, J.S., Su, H.H., Muzumdar, M.D. & Luo, L. Mosaic analysis with double markers in mice. Cell 121, 479–492 (2005).
Callaway, E.M. Transneuronal circuit tracing with neurotropic viruses. Curr. Opin. Neurobiol. 18, 617–623 (2008).
Wang, Y., Zhang, J., Mori, S. & Nathans, J. Axonal growth and guidance defects in Frizzled3 knock-out mice: a comparison of diffusion tensor magnetic resonance imaging, neurofilament staining, and genetically directed cell labeling. J. Neurosci. 26, 355–364 (2006).
Cuntz, H., Forstner, F., Borst, A. & Hausser, M. One rule to grow them all: a general theory of neuronal branching and its practical application. PLoS Comput. Biol. 6, e1000877 (2010).
Acknowledgements
We thank K. Dornmair, J. Enes, C. Hojer, A. Kania, D. Neukirchen, K. Olsen, M. Stiess and S. Tahirovic for critically reading the manuscript, V. Duc Ha for technical assistance and R. Brand for his help with Amira. We are indebted to A. Borst and M. Sheng for their support and to J. Sanes (Harvard University) for the GFP-M mice. A.E. was supported by the Marie Curie Association (European Union; RTN MRTN–CT–2,003–504,636). C.P.M. was supported by the Hertie Stiftung. N.J. was supported by the Theodor Körner Fonds. F.B. is a recipient of a Career Development Award from the Human Frontier Science Program. This work was supported by the Max Planck Society, the International Foundation for Research in Paraplegia and additional grants from the Deutsche Forschungsgemeinschaft and the Hertie Stiftung.
Author information
Authors and Affiliations
Contributions
A.E. initiated the project, designed the experiments, developed and performed the clearing protocol, performed the surgeries, performed the in vivo confocal and two-photon imaging, analyzed the data, made the figures and videos, performed the statistical tests, and wrote the paper. C.P.M. developed and performed the clearing protocol and performed ultramicroscopy imaging. F.H. performed surgeries and analysis for two-dimensional glia quantification and performed the rat tracing. F.F. developed the automated segmentation, tracking software and analyzed the 3D Sholl data. T.K. and M.H. performed initial two-photon experiments. M.R. and E.K. performed the deep-tissue antibody staining. H.S. and F.K. performed two-photon imaging on double- and triple-transgenic mice. K.B. and N.J. performed the histochemical screen and developed the clearing protocol. H.U.D. constructed ultramicroscopy and supervised C.P.M. F.B. initiated the project, designed experiments, coordinated and supervised the project and wrote the paper. All authors edited the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–14 and Supplementary Methods (PDF 2110 kb)
Supplementary Video 1
Uncleared (left) and cleared (right) spinal cords of GFP-M mice were imaged with two-photon microscopy. (MOV 6487 kb)
Supplementary Video 2
A cleared spinal cord tissue section of a GFP-M mouse imaged with confocal microscopy. (MOV 17618 kb)
Supplementary Video 3
3D rotation of the sample shown in Figure 2c. (MOV 13075 kb)
Supplementary Video 4
3D imaging of the unsectioned spinal cord and caudal section of the medulla from a GFP-M mouse in rostro-caudal direction. (MOV 10132 kb)
Supplementary Video 5
The CST of a rat after tracing with biotin dextran amine conjugated to rhodamine, cleared and imaged with two-photon microscopy. (MOV 3905 kb)
Supplementary Video 6
Visualization of a single injured spinal cord of a GFP-M mouse in three different orientations: horizontal, sagittal and cross. (MOV 8073 kb)
Supplementary Video 7
Two-photon stack of the injured spinal cord from a GFP-M mouse in its entire depth in dorsoventral orientation. (MOV 3250 kb)
Supplementary Video 8
3D reconstruction and animation of the spinal cord from a GFP-M mouse shown in Figure 3. (MOV 14446 kb)
Supplementary Video 9
3D reconstruction and animation of the spinal cord from a GFP-M mouse shown in Figure 4, 15 months after injury. (MOV 32719 kb)
Supplementary Video 10
3D reconstruction and animation of the unlesioned spinal cord from a (TgH(CX3CR1-EGFP)) mouse shown in Figure 5a. (MOV 35105 kb)
Supplementary Video 11
Two-photon scan of unlesioned spinal cord from a astrocyte-GFP mouse (TgN(hGFAP-EGFP)) in dorsoventral orientation. (MOV 10149 kb)
Supplementary Video 12
The astrocytes in the spinal cord of TgN(hGFAP-EGFP) mouse scanned by two-photon microscopy in high resolution. (MOV 3910 kb)
Supplementary Video 13
3D imaging of the cleared spinal cord from a double transgenic animal. (MOV 10763 kb)
Rights and permissions
About this article
Cite this article
Ertürk, A., Mauch, C., Hellal, F. et al. Three-dimensional imaging of the unsectioned adult spinal cord to assess axon regeneration and glial responses after injury. Nat Med 18, 166–171 (2012). https://doi.org/10.1038/nm.2600
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.2600
This article is cited by
-
Whole-mouse clearing and imaging at the cellular level with vDISCO
Nature Protocols (2023)
-
Effects of hyperbaric oxygen therapy on postoperative recovery after incomplete cervical spinal cord injury
Spinal Cord (2022)
-
A new protocol for whole-brain biodistribution analysis of AAVs by tissue clearing, light-sheet microscopy and semi-automated spatial quantification
Gene Therapy (2022)
-
Three-dimensional characterization of developing and adult ocular vasculature in mice using in toto clearing
Communications Biology (2022)
-
Antigen retrieval and clearing for whole-organ immunofluorescence by FLASH
Nature Protocols (2021)