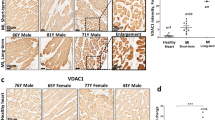
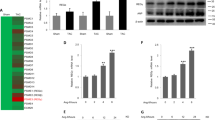
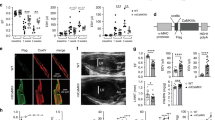
Abstract
Excessive activation of the β-adrenergic, angiotensin II (Ang II) and aldosterone signaling pathways promotes mortality after myocardial infarction, and antagonists targeting these pathways are core therapies for treating this condition. Catecholamines and Ang II activate the multifunctional Ca2+/calmodulin-dependent protein kinase II (CaMKII), the inhibition of which prevents isoproterenol-mediated and Ang II–mediated cardiomyopathy. Here we show that aldosterone exerts direct toxic actions on myocardium by oxidative activation of CaMKII, causing cardiac rupture and increased mortality in mice after myocardial infarction. Aldosterone induces CaMKII oxidation by recruiting NADPH oxidase, and this oxidized and activated CaMKII promotes matrix metalloproteinase 9 (MMP9) expression in cardiomyocytes. Myocardial CaMKII inhibition, overexpression of methionine sulfoxide reductase A (an enzyme that reduces oxidized CaMKII) or NADPH oxidase deficiency prevented aldosterone-enhanced cardiac rupture after myocardial infarction. These findings show that oxidized myocardial CaMKII mediates the cardiotoxic effects of aldosterone on the cardiac matrix and establish CaMKII as a nodal signal for the neurohumoral pathways associated with poor outcomes after myocardial infarction.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Lopez, A.D., Mathers, C.D., Ezzati, M., Jamison, D.T. & Murray, C.J. Global and regional burden of disease and risk factors, 2001: systematic analysis of population health data. Lancet 367, 1747–1757 (2006).
Zhang, R. et al. Calmodulin kinase II inhibition protects against structural heart disease. Nat. Med. 11, 409–417 (2005).
Erickson, J.R. et al. A dynamic pathway for calcium-independent activation of CaMKII by methionine oxidation. Cell 133, 462–474 (2008).
Gutierrez-Marcos, F.M. et al. Atrial natriuretic peptide in patients with acute myocardial infarction without functional heart failure. Eur. Heart J. 12, 503–507 (1991).
Beygui, F. et al. High plasma aldosterone levels on admission are associated with death in patients presenting with acute ST-elevation myocardial infarction. Circulation 114, 2604–2610 (2006).
Pitt, B. et al. Eplerenone, a selective aldosterone blocker, in patients with left ventricular dysfunction after myocardial infarction. N. Engl. J. Med. 348, 1309–1321 (2003).
Pitt, B. et al. The effect of spironolactone on morbidity and mortality in patients with severe heart failure. Randomized Aldactone Evaluation Study Investigators. N. Engl. J. Med. 341, 709–717 (1999).
Reilly, R.F. & Ellison, D.H. Mammalian distal tubule: physiology, pathophysiology, and molecular anatomy. Physiol. Rev. 80, 277–313 (2000).
Rude, M.K. et al. Aldosterone stimulates matrix metalloproteinases and reactive oxygen species in adult rat ventricular cardiomyocytes. Hypertension 46, 555–561 (2005).
Johar, S., Cave, A.C., Narayanapanicker, A., Grieve, D.J. & Shah, A.M. Aldosterone mediates angiotensin II-induced interstitial cardiac fibrosis via a Nox2-containing NADPH oxidase. FASEB J. 20, 1546–1548 (2006).
Nakamura, S. et al. Possible association of heart failure status with synthetic balance between aldosterone and dehydroepiandrosterone in human heart. Circulation 110, 1787–1793 (2004).
Rousseau, M.F. et al. Beneficial neurohormonal profile of spironolactone in severe congestive heart failure: results from the RALES neurohormonal substudy. J. Am. Coll. Cardiol. 40, 1596–1601 (2002).
Huang, C.K., Zhan, L., Hannigan, M.O., Ai, Y. & Leto, T.L. P47(phox)-deficient NADPH oxidase defect in neutrophils of diabetic mouse strains, C57BL/6J-m db/db and db/+. J. Leukoc. Biol. 67, 210–215 (2000).
Swaminathan, P.D. et al. Oxidized CaMKII causes cardiac sinus node dysfunction in mice. J. Clin. Invest. 121, 3277–3288 (2011).
Kusch, M., Farman, N. & Edelman, I.S. Binding of aldosterone to cytoplasmic and nuclear receptors of the urinary bladder epithelium of Bufo marinus. Am. J. Physiol. 235, C82–C89 (1978).
Iwashima, F. et al. Aldosterone induces superoxide generation via Rac1 activation in endothelial cells. Endocrinology 149, 1009–1014 (2008).
Zimmerman, M.C. et al. Requirement for Rac1-dependent NADPH oxidase in the cardiovascular and dipsogenic actions of angiotensin II in the brain. Circ. Res. 95, 532–539 (2004).
Weber, K.T. Aldosterone in congestive heart failure. N. Engl. J. Med. 345, 1689–1697 (2001).
Swedberg, K., Eneroth, P., Kjekshus, J. & Wilhelmsen, L. Hormones regulating cardiovascular function in patients with severe congestive heart failure and their relation to mortality. CONSENSUS Trial Study Group. Circulation 82, 1730–1736 (1990).
Matsumura, S. et al. Targeted deletion or pharmacological inhibition of MMP-2 prevents cardiac rupture after myocardial infarction in mice. J. Clin. Invest. 115, 599–609 (2005).
Heymans, S. et al. Inhibition of plasminogen activators or matrix metalloproteinases prevents cardiac rupture but impairs therapeutic angiogenesis and causes cardiac failure. Nat. Med. 5, 1135–1142 (1999).
Singh, M.V. et al. Ca2+/calmodulin-dependent kinase II triggers cell membrane injury by inducing complement factor B gene expression in the mouse heart. J. Clin. Invest. 119, 986–996 (2009).
van den Borne, S.W. et al. Increased matrix metalloproteinase-8 and -9 activity in patients with infarct rupture after myocardial infarction. Cardiovasc. Pathol. 18, 37–43 (2009).
Bradley, P.P., Priebat, D.A., Christensen, R.D. & Rothstein, G. Measurement of cutaneous inflammation: estimation of neutrophil content with an enzyme marker. J. Invest. Dermatol. 78, 206–209 (1982).
Nahrendorf, M. et al. Activatable magnetic resonance imaging agent reports myeloperoxidase activity in healing infarcts and noninvasively detects the antiinflammatory effects of atorvastatin on ischemia-reperfusion injury. Circulation 117, 1153–1160 (2008).
Kim, Y. et al. The MEF2D transcription factor mediates stress-dependent cardiac remodeling in mice. J. Clin. Invest. 118, 124–132 (2008).
Wu, C.Y., Hsieh, H.L., Sun, C.C. & Yang, C.M. IL-1β induces MMP-9 expression via a Ca(2)-dependent CaMKII/JNK/c-JUN cascade in rat brain astrocytes. Glia 57, 1775–1789 (2009).
Munaut, C. et al. Murine matrix metalloproteinase 9 gene. 5′-upstream region contains cis-acting elements for expression in osteoclasts and migrating keratinocytes in transgenic mice. J. Biol. Chem. 274, 5588–5596 (1999).
Backs, J., Song, K., Bezprozvannaya, S., Chang, S. & Olson, E.N. CaM kinase II selectively signals to histone deacetylase 4 during cardiomyocyte hypertrophy. J. Clin. Invest. 116, 1853–1864 (2006).
Naya, F.J., Wu, C., Richardson, J.A., Overbeek, P. & Olson, E.N. Transcriptional activity of MEF2 during mouse embryogenesis monitored with a MEF2-dependent transgene. Development 126, 2045–2052 (1999).
Kolodziejczyk, S.M. et al. MEF2 is upregulated during cardiac hypertrophy and is required for normal post-natal growth of the myocardium. Curr. Biol. 9, 1203–1206 (1999).
Chang, S. et al. Histone deacetylase 7 maintains vascular integrity by repressing matrix metalloproteinase 10. Cell 126, 321–334 (2006).
Hiratsuka, S. et al. MMP9 induction by vascular endothelial growth factor receptor-1 is involved in lung-specific metastasis. Cancer Cell 2, 289–300 (2002).
Timmins, J.M. et al. Calcium/calmodulin-dependent protein kinase II links ER stress with Fas and mitochondrial apoptosis pathways. J. Clin. Invest. 119, 2925–2941 (2009).
Zhao, B.Q. et al. Role of matrix metalloproteinases in delayed cortical responses after stroke. Nat. Med. 12, 441–445 (2006).
Khoo, M.S. et al. Death, cardiac dysfunction, and arrhythmias are increased by calmodulin kinase II in calcineurin cardiomyopathy. Circulation 114, 1352–1359 (2006).
Backs, J. et al. The delta isoform of CaM kinase II is required for pathological cardiac hypertrophy and remodeling after pressure overload. Proc. Natl. Acad. Sci. USA 106, 2342–2347 (2009).
Doerries, C. et al. Critical role of the NAD(P)H oxidase subunit p47phox for left ventricular remodeling/dysfunction and survival after myocardial infarction. Circ. Res. 100, 894–903 (2007).
Kinugawa, S. et al. Treatment with dimethylthiourea prevents left ventricular remodeling and failure after experimental myocardial infarction in mice: role of oxidative stress. Circ. Res. 87, 392–398 (2000).
Yusuf, S., Dagenais, G., Pogue, J., Bosch, J. & Sleight, P. Vitamin E supplementation and cardiovascular events in high-risk patients. The Heart Outcomes Prevention Evaluation Study Investigators. N. Engl. J. Med. 342, 154–160 (2000).
Rapola, J.M. et al. Randomised trial of alpha-tocopherol and beta-carotene supplements on incidence of major coronary events in men with previous myocardial infarction. Lancet 349, 1715–1720 (1997).
Heart Protection Study Collaborative Group. MRC/BHF Heart Protection Study of antioxidant vitamin supplementation in 20,536 high-risk individuals: a randomised placebo-controlled trial. Lancet 360, 23–33 (2002).
Jauslin, M.L., Meier, T., Smith, R.A. & Murphy, M.P. Mitochondria-targeted antioxidants protect Friedreich Ataxia fibroblasts from endogenous oxidative stress more effectively than untargeted antioxidants. FASEB J. 17, 1972–1974 (2003).
Chen, C.H. et al. Activation of aldehyde dehydrogenase-2 reduces ischemic damage to the heart. Science 321, 1493–1495 (2008).
Tomaselli, G.F. & Barth, A.S. Sudden cardio arrest: oxidative stress irritates the heart. Nat. Med. 16, 648–649 (2010).
Maack, C. et al. Oxygen free radical release in human failing myocardium is associated with increased activity of rac1-GTPase and represents a target for statin treatment. Circulation 108, 1567–1574 (2003).
Dargie, H.J. Effect of carvedilol on outcome after myocardial infarction in patients with left-ventricular dysfunction: the CAPRICORN randomised trial. Lancet 357, 1385–1390 (2001).
Dickstein, K. & Kjekshus, J. & OPTIMAAL Steering Committee of the OPTIMAAL Study Group. Effects of losartan and captopril on mortality and morbidity in high-risk patients after acute myocardial infarction: the OPTIMAAL randomised trial. Optimal Trial in Myocardial Infarction with Angiotensin Antagonist Losartan. Lancet 360, 752–760 (2002).
Christensen, M.D. et al. Oxidized calmodulin kinase II regulates conduction following myocardial infarction: a computational analysis. PLOS Comput. Biol. 5, e1000583 (2009).
Wehrens, X.H. & Doevendans, P.A. Cardiac rupture complicating myocardial infarction. Int. J. Cardiol. 95, 285–292 (2004).
Pouleur, A.C. et al. Pathogenesis of sudden unexpected death in a clinical trial of patients with myocardial infarction and left ventricular dysfunction, heart failure or both. Circulation 122, 597–602 (2010).
Fonarow, G.C., Lukas, M.A., Robertson, M., Colucci, W.S. & Dargie, H.J. Effects of carvedilol early after myocardial infarction: analysis of the first 30 days in Carvedilol Post-Infarct Survival Control in Left Ventricular Dysfunction (CAPRICORN). Am. Heart J. 154, 637–644 (2007).
Ambrosioni, E., Borghi, C. & Magnani, B. The effect of the angiotensin-converting-enzyme inhibitor zofenopril on mortality and morbidity after anterior myocardial infarction. The Survival of Myocardial Infarction Long-Term Evaluation (SMILE) Study Investigators. N. Engl. J. Med. 332, 80–85 (1995).
Beygui, F. et al. Rationale for an early aldosterone blockade in acute myocardial infarction and design of the ALBATROSS trial. Am. Heart J. 160, 642–648 (2010).
Weiss, R.M., Ohashi, M., Miller, J.D., Young, S.G. & Heistad, D.D. Calcific aortic valve stenosis in old hypercholesterolemic mice. Circulation 114, 2065–2069 (2006).
Thiel, W.H. et al. Proarrhythmic defects in Timothy syndrome require calmodulin kinase II. Circulation 118, 2225–2234 (2008).
Mohler, P.J. et al. Defining the cellular phenotype of “ankyrin-B syndrome” variants: human ANK2 variants associated with clinical phenotypes display a spectrum of activities in cardiomyocytes. Circulation 115, 432–441 (2007).
Li, H. et al. Calmodulin kinase II is required for Ang II-mediated vascular smooth muscle hypertrophy. Am. J. Physiol. Heart Circ. Physiol. 298, H688–H698 (2010).
Brot, N., Weissbach, L., Werth, J. & Weissbach, H. Enzymatic reduction of protein-bound methionine sulfoxide. Proc. Natl. Acad. Sci. USA 78, 2155–2158 (1981).
Owusu-Ansah, E., Yavari, A. & Banerjee, U. A protocol for in vivo detection of reactive oxygen species. Protocol Exchange doi:10.1038/nprot.2008.23 (2008).
Acknowledgements
We are grateful for discussions with K. Campbell, W. Nauseef and F. Abboud (University of Iowa). We acknowledge the technical contributions of D. Farley and M. Scheel (University of Iowa). We thank N. Sinclair, P. Yarolem and J. Schwarting (University of Iowa) for their technical expertise in generating transgenic mice. J. Robbins (University of Cincinnati) provided the αMHC complementary DNA (cDNA) for creating the transgenic mice. E. Olson (University of Texas Southwestern) provided mice harboring the MEF2-lacZ reporter gene. MsrA−/− mice were provided by the late E. Stadtman of the US National Institutes of Health. Transgenic mice were engineered at the University of Iowa Transgenic Animal Facility, and viral constructs were generated at the University of Iowa Gene Vector Transfer Core, which are both funded by the US National Institutes of Health. We acknowledge support by the US National Institutes of Health (1F30HL-095325 to B.J.H., RR-017369 to R.M.W., P30 CA086862 and R01CA133114 to D.R.S., R01HL083422 to P.J.M., and R01HL70250, R01HL079031 and R01HL096652 to M.E.A.) and by a grant (08CVD01) from the Fondation Leducq as part of the 'Alliance for CaMKII Signaling in Heart'. S.H. received a Vidi grant from the Netherlands Organization for Scientific Research (91796338) and research grants from the Netherlands Heart Foundation (NHS 2007B036 and 2008B011), Research Foundation–Flanders (FWO 1183211N, 1167610N, G074009N), European Union, FP7-HEALTH-2010, MEDIA, Large scale integrating project.
Author information
Authors and Affiliations
Contributions
B.J.H. designed experiments, analyzed data and wrote the manuscript. M.A.J. designed experiments and assisted with the tissue and image analyses. M.V.S. assisted with the MPO activity assay, analyzed gene array data and assisted with qRT-PCR design and analysis. E.D.L. assisted with animal studies, immunoblotting, experimental design and data analysis. P.D.S. assisted with immunoblotting, experimental design and data analysis. O.M.K. assisted in cell culture isolation. W.K. performed mouse surgeries and analyzed data. C.A. performed immunostaining studies. J.Y. performed mouse studies and assisted with mouse models. X.G. assisted with subcloning work. K.Z. performed echocardiographic experiments and analyzed data. I.M.G. assisted in developing adenoviral constructs and edited the manuscript. R.M.W. designed echocardiographic studies, analyzed data and edited the manuscript. D.R.S. assisted with the MsrA transgenic mouse design and development of the MsrA assay, analyzed data and edited the manuscript. C.D.S. developed the MsrA transgenic mice, analyzed data and edited the manuscript. W.M.B., S.H. and P.J.M. designed experiments, analyzed data and edited the manuscript. M.E.A. designed experiments, analyzed data, co-wrote the manuscript and supervised the project.
Corresponding author
Ethics declarations
Competing interests
M.E.A. has intellectual property claiming to treat myocardial infarction by CaMKII inhibition and is a co-founder of Allosteros Therapeutics, a biotech company aiming to develop enzyme-based therapies.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–8 and Supplementary Methods (PDF 3566 kb)
Rights and permissions
About this article
Cite this article
He, B., Joiner, Ml., Singh, M. et al. Oxidation of CaMKII determines the cardiotoxic effects of aldosterone. Nat Med 17, 1610–1618 (2011). https://doi.org/10.1038/nm.2506
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.2506
This article is cited by
-
Role of CaMKII in diabetes induced vascular injury and its interaction with anti-diabetes therapy
Reviews in Endocrine and Metabolic Disorders (2024)
-
JOSD2 mediates isoprenaline-induced heart failure by deubiquitinating CaMKIIδ in cardiomyocytes
Cellular and Molecular Life Sciences (2024)
-
Effect of Colchicine in reducing MMP-9, NOX2, and TGF- β1 after myocardial infarction
BMC Cardiovascular Disorders (2023)
-
Mineralocorticoid receptor antagonists in diabetic kidney disease — mechanistic and therapeutic effects
Nature Reviews Nephrology (2022)
-
Bisphenol A induces coronary endothelial cell necroptosis by activating RIP3/CamKII dependent pathway
Scientific Reports (2020)