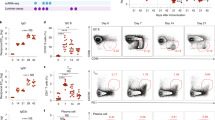
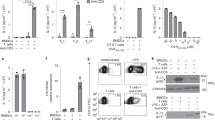
Abstract
Aluminum-based adjuvants (aluminum salts or alum) are widely used in human vaccination, although their mechanisms of action are poorly understood. Here we report that, in mice, alum causes cell death and the subsequent release of host cell DNA, which acts as a potent endogenous immunostimulatory signal mediating alum adjuvant activity. Furthermore, we propose that host DNA signaling differentially regulates IgE and IgG1 production after alum-adjuvanted immunization. We suggest that, on the one hand, host DNA induces primary B cell responses, including IgG1 production, through interferon response factor 3 (Irf3)-independent mechanisms. On the other hand, we suggest that host DNA also stimulates 'canonical' T helper type 2 (TH2) responses, associated with IgE isotype switching and peripheral effector responses, through Irf3-dependent mechanisms. The finding that host DNA released from dying cells acts as a damage-associated molecular pattern that mediates alum adjuvant activity may increase our understanding of the mechanisms of action of current vaccines and help in the design of new adjuvants.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Glenny, A.T., Pope, C.G., Waddington, H. & Wallace, U. Immunological Notes: XVII–XXIV. J. Pathol. Bacteriol. 29, 31–40 (1926).
Mannhalter, J.W., Neychev, H.O., Zlabinger, G.J., Ahmad, R. & Eibl, M.M. Modulation of the human immune response by the non-toxic and non-pyrogenic adjuvant aluminium hydroxide: effect on antigen uptake and antigen presentation. Clin. Exp. Immunol. 61, 143–151 (1985).
Li, H., Nookala, S. & Re, F. Aluminum hydroxide adjuvants activate Caspase-1 and induce IL-1β and IL-18 release. J. Immunol. 178, 5271–5276 (2007).
Hornung, V. et al. Silica crystals and aluminum salts activate the NALP3 inflammasome through phagosomal destabilization. Nat. Immunol. 9, 847–856 (2008).
Eisenbarth, S.C., Colegio, O.R., O'Connor, W., Sutterwala, F.S. & Flavell, R.A. Crucial role for the Nalp3 inflammasome in the immunostimulatory properties of aluminium adjuvants. Nature 453, 1122–1126 (2008).
Franchi, L. & Nunez, G. The Nlrp3 inflammasome is critical for aluminium hydroxide-mediated IL-1β; secretion but dispensable for adjuvant activity. Eur. J. Immunol. 38, 2085–2089 (2008).
Kool, M. et al. Cutting Edge: alum adjuvant stimulates inflammatory dendritic cells through activation of the NALP3 inflammasome. J. Immunol. 181, 3755–3759 (2008).
Li, H., Willingham, S.B., Ting, J.P.Y. & Re, F. Cutting Edge: Inflammasome activation by alum and alum's adjuvant effect are mediated by NLRP3. J. Immunol. 181, 17–21 (2008).
Spreafico, R., Ricciardi-Castagnoli, P. & Mortellaro, A. The controversial relationship between NLRP3, alum, danger signals and the next-generation adjuvants. Eur. J. Immunol. 40, 638–642 (2010).
McKee, A.S. et al. Alum induces innate immune responses through macrophage and mast cell sensors, but these sensors are not required for alum to act as an adjuvant for specific immunity. J. Immunol. 183, 4403–4414 (2009).
Kool, M. et al. Alum adjuvant boosts adaptive immunity by inducing uric acid and activating inflammatory dendritic cells. J. Exp. Med. 205, 869–882 (2008).
Goto, N. et al. Local tissue irritating effects and adjuvant activities of calcium phosphate and aluminium hydroxide with different physical properties. Vaccine 15, 1364–1371 (1997).
Kono, H. & Rock, K.L. How dying cells alert the immune system to danger. Nat. Rev. Immunol. 8, 279–289 (2008).
Chen, G.Y. & Nunez, G. Sterile inflammation: sensing and reacting to damage. Nat. Rev. Immunol. 10, 826–837 (2010).
Ishii, K.J. et al. Genomic DNA released by dying cells induces the maturation of APCs. J. Immunol. 167, 2602–2607 (2001).
Takeshita, F. & Ishii, K.J. Intracellular DNA sensors in immunity. Curr. Opin. Immunol. 20, 383–388 (2008).
Takeuchi, O. & Akira, S. Pattern recognition receptors and inflammation. Cell 140, 805–820 (2010).
Lande, R. et al. Plasmacytoid dendritic cells sense self-DNA coupled with antimicrobial peptide. Nature 449, 564–569 (2007).
Imaeda, A.B. et al. Acetaminophen-induced hepatotoxicity in mice is dependent on Tlr9 and the Nalp3 inflammasome. J. Clin. Invest. 119, 305–314 (2009).
Takaoka, A. et al. DAI (DLM-1/ZBP1) is a cytosolic DNA sensor and an activator of innate immune response. Nature 448, 501–505 (2007).
Muruve, D.A. et al. The inflammasome recognizes cytosolic microbial and host DNA and triggers an innate immune response. Nature 452, 103–107 (2008).
Chiu, Y.-H., MacMillan, J.B. & Chen, Z.J. RNA Polymerase III detects cytosolic DNA and induces type I Interferons through the RIG-I pathway. Cell 138, 576–591 (2009).
Fernandes-Alnemri, T., Yu, J.-W., Datta, P., Wu, J. & Alnemri, E.S. AIM2 activates the inflammasome and cell death in response to cytoplasmic DNA. Nature 458, 509–513 (2009).
Hornung, V. et al. AIM2 recognizes cytosolic dsDNA and forms a caspase-1-activating inflammasome with ASC. Nature 458, 514–518 (2009).
Ishikawa, H., Ma, Z. & Barber, G.N. STING regulates intracellular DNA-mediated, type I interferon-dependent innate immunity. Nature 461, 788–792 (2009).
Yanai, H. et al. HMGB proteins function as universal sentinels for nucleic-acid-mediated innate immune responses. Nature 462, 99–103 (2009).
Yang, P. et al. The cytosolic nucleic acid sensor LRRFIP1 mediates the production of type I interferon via a β-catenin–dependent pathway. Nat. Immunol. 11, 487–494 (2010).
Ishii, K.J. et al. A Toll-like receptor-independent antiviral response induced by double-stranded B-form DNA. Nat. Immunol. 7, 40–48 (2006).
Bonnard, M. et al. Deficiency of T2K leads to apoptotic liver degeneration and impaired NF-κB-dependent gene transcription. EMBO J. 19, 4976–4985 (2000).
Ishii, K.J. et al. TANK-binding kinase-1 delineates innate and adaptive immune responses to DNA vaccines. Nature 451, 725–729 (2008).
Marichal, T. et al. Interferon response factor 3 is essential for house dust mite-induced airway allergy. J. Allergy Clin. Immunol. 126, 836–844 (2010).
Reinhardt, R.L., Liang, H.-E. & Locksley, R.M. Cytokine-secreting follicular T cells shape the antibody repertoire. Nat. Immunol. 10, 385–393 (2009).
Naik, S.H. et al. Intrasplenic steady-state dendritic cell precursors that are distinct from monocytes. Nat. Immunol. 7, 663–671 (2006).
Shortman, K. & Naik, S.H. Steady-state and inflammatory dendritic-cell development. Nat. Rev. Immunol. 7, 19–30 (2007).
Khader, S.A. et al. Interleukin 12p40 is required for dendritic cell migration and T cell priming after Mycobacterium tuberculosis infection. J. Exp. Med. 203, 1805–1815 (2006).
Robinson, R.T. et al. Yersinia pestis evades TLR4-dependent induction of IL-12(p40)2 by dendritic cells and subsequent cell migration. J. Immunol. 181, 5560–5567 (2008).
Yasutomo, K. et al. Mutation of DNASE1 in people with systemic lupus erythematosus. Nat. Genet. 28, 313–314 (2001).
Yoshida, H., Okabe, Y., Kawane, K., Fukuyama, H. & Nagata, S. Lethal anemia caused by interferon-beta produced in mouse embryos carrying undigested DNA. Nat. Immunol. 6, 49–56 (2005).
Kawane, K. et al. Chronic polyarthritis caused by mammalian DNA that escapes from degradation in macrophages. Nature 443, 998–1002 (2006).
Pulendran, B. & Ahmed, R. Translating Innate Immunity into Immunological Memory: Implications for Vaccine Development. Cell 124, 849–863 (2006).
Coffman, R.L., Sher, A. & Seder, R.A. Vaccine adjuvants: putting innate immunity to work. Immunity 33, 492–503 (2010).
Fazilleau, N., McHeyzer-Williams, L.J., Rosen, H. & McHeyzer-Williams, M.G. The function of follicular helper T cells is regulated by the strength of T cell antigen receptor binding. Nat. Immunol. 10, 375–384 (2009).
Fazilleau, N., Mark, L., McHeyzer-Williams, L.J. & McHeyzer-Williams, M.G. Follicular helper T cells: lineage and location. Immunity 30, 324–335 (2009).
Nakano, H. et al. Blood-derived inflammatory dendritic cells in lymph nodes stimulate acute T helper type 1 immune responses. Nat. Immunol. 10, 394–402 (2009).
Mohr, E. et al. Dendritic cells and monocyte/macrophages that create the IL-6/APRIL-rich lymph node microenvironments where plasmablasts mature. J. Immunol. 182, 2113–2123 (2009).
Hemmi, H. et al. A Toll-like receptor recognizes bacterial DNA. Nature 408, 740–745 (2000).
Hemmi, H. et al. The roles of two IκB kinase–related kinases in lipopolysaccharide and double stranded RNA signaling and viral infection. J. Exp. Med. 199, 1641–1650 (2004).
Sato, M. et al. Distinct and essential roles of transcription factors IRF-3 and IRF-7 in response to viruses for IFN-α/β gene induction. Immunity 13, 539–548 (2000).
Kimura, T. et al. Essential and non-redundant roles of p48 (ISGF3 gamma) and IRF-1 in both type I and type II interferon responses, as revealed by gene targeting studies. Genes Cells 1, 115–124 (1996).
Mariathasan, S. et al. Differential activation of the inflammasome by caspase-1 adaptors ASC and Ipaf. Nature 430, 213–218 (2004).
Mariathasan, S. et al. Cryopyrin activates the inflammasome in response to toxins and ATP. Nature 440, 228–232 (2006).
Bedoret, D. et al. Lung interstitial macrophages alter dendritic cell functions to prevent airway allergy in mice. J. Clin. Invest. 119, 3723–3738 (2009).
Nigg, A.P. et al. Dendritic cell-derived IL-12p40 homodimer contributes to susceptibility in cutaneous leishmaniasis in BALB/c mice. J. Immunol. 178, 7251–7258 (2007).
Acknowledgements
The authors thank T. Taniguchi (University of Tokyo) and RIKEN BioResource Center for providing Irf3−/− and Irf9−/− mice, and V. Dixit (Genentech) for providing Nlrp3−/− and Casp1−/− mice. We also thank S. Ormenese and R. Stephaan of the Cell Imaging and Flow Cytometry Technological Platform of the Groupe Interdisciplinaire de Génoprotéomique Appliquée for help with fluorescent-activated cell sorting (FACS) analyses; M. Lebrun for help with confocal microscopy; P. Drion and G. Gaudray for animal management; F. Andris, S. Goriely and O. Leo for helpful discussions; and V. Conrath, L. Duwez, R. Fares, C. François, F. Olivier, J. Parisi, F. Perin and I. Sbai for excellent technical and secretarial assistance.
T.M., D.B., C.M. and C.S. are research fellows, and C.J.D. is a postdoctoral fellow of the Fonds National de la Recherche Scientifique (FRS-FNRS; Belgium). This work was supported by grants of the FRS-FNRS, the Belgian Fonds de la Recherche Scientifique Médicale and the Belgian Programme on Interuniversity Attraction Poles (IUAP; FEDIMMUNE, Belgian Science Policy). This work was also partly supported by the Knowledge Cluster Initiative (K.J.I.); a Grant-in-Aid for Scientific Research (K.J.I. and C.C.) of the Japanese Ministry of Education, Culture, Sports, Science and Technology (MEXT); and by the Core Research Evolutionary Science and Technology (CREST) program at the Japan Science and Technology Agency (K.J.I.).
Author information
Authors and Affiliations
Contributions
T.M., K.J.I., F.B. and C.J.D. designed the experiments; C.C., K.J.I., F.B. and C.J.D. supervised the project; T.M. and D.B. made the initial observation; T.M. did most of the experiments and compiled the data; T.M., K.O. and K.K. did the experiments involving Tbk1/Tnf double-knockout mice, Zbp1−/−, Ifnar2−/− and Mavs−/− mice; C.M. and C.S. did the FACS analyses; S.A. provided the Tbk1/Tnf double-knockout mice and Zbp1−/− mice; P.L., S.A., K.J.I. and F.B. secured funding; K.J.I. and F.B. provided feedback on the manuscript; and C.J.D. wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–17 and Supplementary Methods (PDF 3117 kb)
Rights and permissions
About this article
Cite this article
Marichal, T., Ohata, K., Bedoret, D. et al. DNA released from dying host cells mediates aluminum adjuvant activity. Nat Med 17, 996–1002 (2011). https://doi.org/10.1038/nm.2403
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.2403
This article is cited by
-
Vaccine adjuvants: mechanisms and platforms
Signal Transduction and Targeted Therapy (2023)
-
Advances on the early cellular events occurring upon exposure of human macrophages to aluminum oxyhydroxide adjuvant
Scientific Reports (2023)
-
Dynamics of redox signaling in aging via autophagy, inflammation, and senescence
Biogerontology (2023)
-
Effective adjuvantation of nanograms of influenza vaccine and induction of cross-protective immunity by physical radiofrequency adjuvant
Scientific Reports (2022)
-
Emerging concepts in the science of vaccine adjuvants
Nature Reviews Drug Discovery (2021)