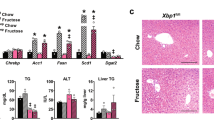
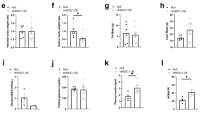
Abstract
To date, the only known role of the spliced form of X-box–binding protein-1 (XBP-1s) in metabolic processes has been its ability to act as a transcription factor that regulates the expression of genes that increase the endoplasmic reticulum (ER) folding capacity, thereby improving insulin sensitivity. Here we show that XBP-1s interacts with the Forkhead box O1 (FoxO1) transcription factor and directs it toward proteasome-mediated degradation. Given this new insight, we tested modest hepatic overexpression of XBP-1s in vivo in mouse models of insulin deficiency or insulin resistance and found it improved serum glucose concentrations, even without improving insulin signaling or ER folding capacity. The notion that XBP-1s can act independently of its role in the ER stress response is further supported by our finding that in the severely insulin resistant ob/ob mouse strain a DNA-binding–defective mutant of XBP-1s, which does not have the ability to increase ER folding capacity, is still capable of reducing serum glucose concentrations and increasing glucose tolerance. Our results thus provide the first evidence to our knowledge that XBP-1s, through its interaction with FoxO1, can bypass hepatic insulin resistance independent of its effects on ER folding capacity, suggesting a new therapeutic approach for the treatment of type 2 diabetes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
James, P.T., Rigby, N. & Leach, R. The obesity epidemic, metabolic syndrome and future prevention strategies. Eur. J. Cardiovasc. Prev. Rehabil. 11, 3–8 (2004).
Kitamura, Y. & Accili, D. New insights into the integrated physiology of insulin action. Rev. Endocr. Metab. Disord. 5, 143–149 (2004).
de Luca, C. & Olefsky, J.M. Stressed out about obesity and insulin resistance. Nat. Med. 12, 41–42 discussion 42 (2006).
Biddinger, S.B. & Kahn, C.R. From mice to men: insights into the insulin resistance syndromes. Annu. Rev. Physiol. 68, 123–158 (2006).
Saltiel, A.R. New perspectives into the molecular pathogenesis and treatment of type 2 diabetes. Cell 104, 517–529 (2001).
Schröder, M. & Kaufman, R.J. The mammalian unfolded protein response. Annu. Rev. Biochem. 74, 739–789 (2005).
Marciniak, S.J. & Ron, D. Endoplasmic reticulum stress signaling in disease. Physiol. Rev. 86, 1133–1149 (2006).
Bernales, S., Papa, F.R. & Walter, P. Intracellular signaling by the unfolded protein response. Annu. Rev. Cell Dev. Biol. 22, 487–508 (2006).
Ron, D. & Walter, P. Signal integration in the endoplasmic reticulum unfolded protein response. Nat. Rev. Mol. Cell Biol. 8, 519–529 (2007).
Zhang, K. & Kaufman, R.J. From endoplasmic-reticulum stress to the inflammatory response. Nature 454, 455–462 (2008).
Cox, J.S., Shamu, C.E. & Walter, P. Transcriptional induction of genes encoding endoplasmic reticulum resident proteins requires a transmembrane protein kinase. Cell 73, 1197–1206 (1993).
Mori, K., Ma, W., Gething, M.J. & Sambrook, J. A transmembrane protein with a cdc2+/CDC28-related kinase activity is required for signaling from the ER to the nucleus. Cell 74, 743–756 (1993).
Yoshida, H., Matsui, T., Yamamoto, A., Okada, T. & Mori, K. XBP1 mRNA is induced by ATF6 and spliced by IRE1 in response to ER stress to produce a highly active transcription factor. Cell 107, 881–891 (2001).
Lee, K. et al. IRE1-mediated unconventional mRNA splicing and S2P-mediated ATF6 cleavage merge to regulate XBP1 in signaling the unfolded protein response. Genes Dev. 16, 452–466 (2002).
Calfon, M. et al. IRE1 couples endoplasmic reticulum load to secretory capacity by processing the XBP-1 mRNA. Nature 415, 92–96 (2002).
Lee, A.H., Iwakoshi, N.N. & Glimcher, L.H. XBP-1 regulates a subset of endoplasmic reticulum resident chaperone genes in the unfolded protein response. Mol. Cell. Biol. 23, 7448–7459 (2003).
Sriburi, R., Jackowski, S., Mori, K. & Brewer, J.W. XBP1: a link between the unfolded protein response, lipid biosynthesis, and biogenesis of the endoplasmic reticulum. J. Cell Biol. 167, 35–41 (2004).
Ozcan, U. et al. Loss of the tuberous sclerosis complex tumor suppressors triggers the unfolded protein response to regulate insulin signaling and apoptosis. Mol. Cell 29, 541–551 (2008).
Ozcan, U. et al. Chemical chaperones reduce ER stress and restore glucose homeostasis in a mouse model of type 2 diabetes. Science 313, 1137–1140 (2006).
Ozcan, U. et al. Endoplasmic reticulum stress links obesity, insulin action, and type 2 diabetes. Science 306, 457–461 (2004).
Ozcan, L. et al. Endoplasmic reticulum stress plays a central role in development of leptin resistance. Cell Metab. 9, 35–51 (2009).
Park, S.W. et al. The regulatory subunits of PI3K, p85α and p85β, interact with XBP-1 and increase its nuclear translocation. Nat. Med. 16, 429–437 (2010).
Gross, D.N., van den Heuvel, A.P. & Birnbaum, M.J. The role of FoxO in the regulation of metabolism. Oncogene 27, 2320–2336 (2008).
Accili, D. & Arden, K.C. FoxOs at the crossroads of cellular metabolism, differentiation, and transformation. Cell 117, 421–426 (2004).
Hedrick, S.M. The cunning little vixen: Foxo and the cycle of life and death. Nat. Immunol. 10, 1057–1063 (2009).
Puigserver, P. et al. Insulin-regulated hepatic gluconeogenesis through FOXO1–PGC-1α interaction. Nature 423, 550–555 (2003).
Matsumoto, M. & Accili, D. The tangled path to glucose production. Nat. Med. 12, 33–34 (2006).
Daitoku, H. et al. Silent information regulator 2 potentiates Foxo1-mediated transcription through its deacetylase activity. Proc. Natl. Acad. Sci. USA 101, 10042–10047 (2004).
Motta, M.C. et al. Mammalian SIRT1 represses forkhead transcription factors. Cell 116, 551–563 (2004).
Frescas, D., Valenti, L. & Accili, D. Nuclear trapping of the forkhead transcription factor FoxO1 via Sirt-dependent deacetylation promotes expression of glucogenetic genes. J. Biol. Chem. 280, 20589–20595 (2005).
Yamagata, K. et al. Arginine methylation of FOXO transcription factors inhibits their phosphorylation by Akt. Mol. Cell 32, 221–231 (2008).
Huang, H. et al. Skp2 inhibits FOXO1 in tumor suppression through ubiquitin-mediated degradation. Proc. Natl. Acad. Sci. USA 102, 1649–1654 (2005).
Matsuzaki, H., Daitoku, H., Hatta, M., Tanaka, K. & Fukamizu, A. Insulin-induced phosphorylation of FKHR (Foxo1) targets to proteasomal degradation. Proc. Natl. Acad. Sci. USA 100, 11285–11290 (2003).
Aoki, M., Jiang, H. & Vogt, P.K. Proteasomal degradation of the FoxO1 transcriptional regulator in cells transformed by the P3k and Akt oncoproteins. Proc. Natl. Acad. Sci. USA 101, 13613–13617 (2004).
Nakae, J., Kitamura, T., Silver, D.L. & Accili, D. The forkhead transcription factor Foxo1 (Fkhr) confers insulin sensitivity onto glucose-6-phosphatase expression. J. Clin. Invest. 108, 1359–1367 (2001).
Dong, X.C. et al. Inactivation of hepatic Foxo1 by insulin signaling is required for adaptive nutrient homeostasis and endocrine growth regulation. Cell Metab. 8, 65–76 (2008).
Michael, M.D. et al. Loss of insulin signaling in hepatocytes leads to severe insulin resistance and progressive hepatic dysfunction. Mol. Cell 6, 87–97 (2000).
Matsumoto, M., Pocai, A., Rossetti, L., Depinho, R.A. & Accili, D. Impaired regulation of hepatic glucose production in mice lacking the forkhead transcription factor Foxo1 in liver. Cell Metab. 6, 208–216 (2007).
Ozawa, K. et al. The endoplasmic reticulum chaperone improves insulin resistance in type 2 diabetes. Diabetes 54, 657–663 (2005).
Henis-Korenblit, S. et al. Insulin/IGF-1 signaling mutants reprogram ER stress response regulators to promote longevity. Proc. Natl. Acad. Sci. USA 107, 9730–9735 (2010).
Fan, W. et al. FoxO1 regulates Tlr4 inflammatory pathway signalling in macrophages. EMBO J (2010).
Kenyon, C.J. The genetics of ageing. Nature 464, 504–512 (2010).
Russell, S.J. & Kahn, C.R. Endocrine regulation of ageing. Nat. Rev. Mol. Cell Biol. 8, 681–691 (2007).
Blüher, M., Kahn, B.B. & Kahn, C.R. Extended longevity in mice lacking the insulin receptor in adipose tissue. Science 299, 572–574 (2003).
Taguchi, A., Wartschow, L.M. & White, M.F. Brain IRS2 signaling coordinates life span and nutrient homeostasis. Science 317, 369–372 (2007).
Altomonte, J. et al. Inhibition of Foxo1 function is associated with improved fasting glycemia in diabetic mice. Am. J. Physiol. Endocrinol. Metab. 285, E718–E728 (2003).
Acknowledgements
We thank members of the Ozcan laboratory for their help during the execution of the experiments. We thank L. Glimcher (Harvard School of Public Health) for providing us with the Xbp1flox/flox mouse strain. We are grateful to D. Accili (Columbia University) for providing us with various FoxO1 adenoviruses and to R. Kahn (Joslin Diabetes Center) for his permission to use LIRKO mice in our studies. We thank J. Majzoub (Harvard Medical School) for providing us with the mammalian two-hybrid system and R. King (Harvard Medical School) for providing us with a ubiquitin-expressing plasmid. We thank M. Birnbaum (University of Pennsylvania) for providing us with Akt1/2 double-knockout cells. This study was supported by junior faculty start-up funds provided to U.O. by Children's Hospital Boston, an RO1 grant (R01DK081009) provided to U.O. by the US National Institutes of Health, and the Timothy Murphy funds provided to the Division of Endocrinology, Children's Hospital Boston.
Author information
Authors and Affiliations
Contributions
Y.Z. came up with the hypothesis, designed and performed the experiments, analyzed the data and wrote the manuscript. Justin Lee, C.M.R., C.S., S.W.P., J.C., Jaemin Lee and S.J.F. performed the experiments. M.F.W. provided liver specific IRS1/2 double-knockout mice. S.B.B. provided LIRKO mice and performed experiments. U.O. came up with the hypothesis, designed and performed the experiments, analyzed the data and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–7 and Supplementary Methods (PDF 392 kb)
Rights and permissions
About this article
Cite this article
Zhou, Y., Lee, J., Reno, C. et al. Regulation of glucose homeostasis through a XBP-1–FoxO1 interaction. Nat Med 17, 356–365 (2011). https://doi.org/10.1038/nm.2293
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.2293
This article is cited by
-
Body mass index stratified meta-analysis of genome-wide association studies of polycystic ovary syndrome in women of European ancestry
BMC Genomics (2024)
-
Transcriptional regulation of autophagy and its implications in human disease
Cell Death & Differentiation (2023)
-
Chronic Cerebral Hypoperfusion-Induced Disturbed Proteostasis of Mitochondria and MAM Is Reflected in the CSF of Rats by Proteomic Analysis
Molecular Neurobiology (2023)
-
The activation of spliced X-box binding protein 1 by isorhynchophylline therapy improves diabetic encephalopathy
Cell Biology and Toxicology (2023)
-
Pharmacologic IRE1/XBP1s activation promotes systemic adaptive remodeling in obesity
Nature Communications (2022)