Abstract
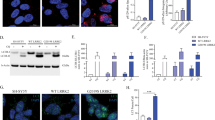
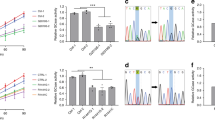
Leucine-rich repeat kinase-2 (LRRK2) mutations are a common cause of Parkinson's disease. Here we identify inhibitors of LRRK2 kinase that are protective in in vitro and in vivo models of LRRK2-induced neurodegeneration. These results establish that LRRK2-induced degeneration of neurons in vivo is kinase dependent and that LRRK2 kinase inhibition provides a potential new neuroprotective paradigm for the treatment of Parkinson's disease.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Gasser, T. Expert Rev. Mol. Med. 11, e22 (2009).
Greggio, E. et al. Neurobiol. Dis. 23, 329–341 (2006).
Smith, W.W. et al. Nat. Neurosci. 9, 1231–1233 (2006).
West, A.B. et al. Hum. Mol. Genet. 16, 223–232 (2007).
Whaley, N.R., Uitti, R.J., Dickson, D.W., Farrer, M.J. & Wszolek, Z.K. J. Neural Transm. Suppl. 70, 221–229 (2006).
Mata, I.F., Wedemeyer, W.J., Farrer, M.J., Taylor, J.P. & Gallo, K.A. Trends Neurosci. 29, 286–293 (2006).
Chin, P.C. et al. J. Neurochem. 90, 595–608 (2004).
Imai, Y. et al. EMBO J. 27, 2432–2443 (2008).
Smith, W.W. et al. Proc. Natl. Acad. Sci. USA 102, 18676–18681 (2005).
Leclerc, S. et al. J. Biol. Chem. 276, 251–260 (2001).
Wang, W. et al. Neuropharmacology 52, 1678–1684 (2007).
Anand, V.S. et al. FEBS J. 276, 466–478 (2009).
Covy, J.P. & Giasson, B.I. Biochem. Biophys. Res. Commun. 378, 473–477 (2009).
Nichols, R.J. et al. Biochem. J. 424, 47–60 (2009).
Reichling, L.J. & Riddle, S.M. Biochem. Biophys. Res. Commun. 384, 255–258 (2009).
Acknowledgements
We thank C. Burris and L. Lotta for packaging helper virus–free amplicons. C. Cook, K. Kehoe, J. Dunmore and C. Eckman provided technical support for some of the in vivo HSV studies. We also thank G. and K. Caldwell and S. Hamamichi for helpful discussions. This work was supported by grants from the US National Institutes of Health, P50NS38377, R01ES014470 (K.A.M.-Z.), R01-AG023593 (W.J.B.), R00-NS058111 (A.B.W.), NS36420 (H.J.F.) and Army Medical Research and Materiel Command, DAMD17-02-1-0695 (H.J.F.), the Mayo Foundation and the Michael J. Fox Foundation.
Author information
Authors and Affiliations
Contributions
B.D.L., J.V., L.P., A.B.W., V.L.D. and T.M.D. designed the experiments. B.D.L., J.V., H.S.K., Y.I.L. and J.-H.S. generated data. K.A.M.-Z., W.J.B. and H.J.F. generated HSV-encoded LRRK2s. B.D.L. and J.V. analyzed data. B.D.L., V.L.D. and T.M.D. wrote the manuscript. All authors discussed the results and commented on the manuscript.
Corresponding authors
Ethics declarations
Competing interests
T.M.D. is a paid consultant to Merck KGAA. The terms of this arrangement are being managed by the Johns Hopkins University in accordance with its conflict of interest policies.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–6, Supplementary Tables 1–3 and Supplementary Methods (PDF 1295 kb)
Rights and permissions
About this article
Cite this article
Lee, B., Shin, JH., VanKampen, J. et al. Inhibitors of leucine-rich repeat kinase-2 protect against models of Parkinson's disease. Nat Med 16, 998–1000 (2010). https://doi.org/10.1038/nm.2199
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.2199
This article is cited by
-
Inflammation and immune dysfunction in Parkinson disease
Nature Reviews Immunology (2022)
-
The role of tyrosine hydroxylase–dopamine pathway in Parkinson’s disease pathogenesis
Cellular and Molecular Life Sciences (2022)
-
Preclinical modeling of chronic inhibition of the Parkinson’s disease associated kinase LRRK2 reveals altered function of the endolysosomal system in vivo
Molecular Neurodegeneration (2021)
-
LncRNAs as putative biomarkers and therapeutic targets for Parkinson’s disease
Neurological Sciences (2021)
-
In Vitro CRISPR/Cas9-Directed Gene Editing to Model LRRK2 G2019S Parkinson’s Disease in Common Marmosets
Scientific Reports (2020)