Abstract
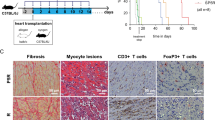
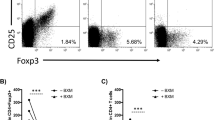
Transplant arteriosclerosis is the hallmark of chronic allograft dysfunction (CAD) affecting transplanted organs in the long term1,2. These fibroproliferative lesions lead to neointimal thickening of arteries in all transplanted allografts2. Luminal narrowing then leads to graft ischemia and organ demise. To date, there are no known tolerance induction strategies that prevent transplant arteriosclerosis3,4. Therefore, we designed this study to test the hypothesis that human regulatory T cells (Treg cells) expanded ex vivo can prevent transplant arteriosclerosis. Here we show the comparative capacity of Treg cells, sorted via two separate strategies, to prevent transplant arteriosclerosis in a clinically relevant chimeric humanized mouse system. We found that the in vivo development of transplant arteriosclerosis in human arteries was prevented by treatment of ex vivo–expanded human Treg cells. Additionally, we show that Treg cells sorted on the basis of low expression of CD127 provide a more potent therapy to conventional Treg cells. Our results demonstrate that human Treg cells can inhibit transplant arteriosclerosis by impairing effector function and graft infiltration. We anticipate our findings to serve as a foundation for the clinical development of therapeutics targeting transplant arteriosclerosis in both allograft transplantation and other immune-mediated causes of vasculopathy5.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Change history
26 May 2010
In the version of this article initially published online, in Figure 1a, underneath ‘Silencing’, the text reading “2 d: 200 U ml–1 IL-2 + beads” was incorrect. It should read “2 d: 200 U ml–1 IL-2 no beads.” The error has been corrected for the print, PDF and HTML versions of this article.
References
Pucci, A.M., Forbes, R.D. & Billingham, M.E. Pathologic features in long-term cardiac allografts. J. Heart Transplant. 9, 339–345 (1990).
Hillebrands, J.L. & Rozing, J. Chronic transplant dysfunction and transplant arteriosclerosis: new insights into underlying mechanisms. Expert Rev. Mol. Med. 33, 1–23 (2003).
Mitchell, R.N. & Lichtman, A.H. The link between IFN-γ and allograft arteriopathy: is the answer NO? J. Clin. Invest. 114, 762–764 (2004).
Taylor, D.O. et al. Registry of the International Society for Heart and Lung Transplantation: twenty-third official adult heart transplantation report–2006. J. Heart Lung Transplant. 25, 869–879 (2006).
Tullius, S.G. et al. Contribution of early acute rejection episodes to chronic rejection in a rat kidney retransplantation model. Kidney Int. 53, 465–472 (1998).
Nankivell, B.J. et al. The natural history of chronic allograft nephropathy. N. Engl. J. Med. 349, 2326–2333 (2003).
Kobashigawa, J.A. et al. Multicenter intravascular ultrasound validation study among heart transplant recipients: outcomes after five years. J. Am. Coll. Cardiol. 45, 1532–1537 (2005).
Tellides, G. et al. Interferon-γ elicits arteriosclerosis in the absence of leukocytes. Nature 403, 207–211 (2000).
Godfrey, W.R. et al. In vitro–expanded human CD4+CD25+ T-regulatory cells can markedly inhibit allogeneic dendritic cell–stimulated MLR cultures. Blood 104, 453–461 (2004).
Hoffmann, P., Eder, R., Kunz-Schughart, L.A., Andreesen, R. & Edinger, M. Large-scale in vitro expansion of polyclonal human CD4+CD25high regulatory T cells. Blood 104, 895–903 (2004).
Wood, K.J. & Sakaguchi, S. Regulatory T cells in transplantation tolerance. Nat. Rev. Immunol. 3, 199–210 (2003).
Wang, Y. et al. Interferon-γ induces human vascular smooth muscle cell proliferation and intimal expansion by phosphatidylinositol 3-kinase dependent mammalian target of rapamycin raptor complex 1 activation. Circ. Res. 101, 560–569 (2007).
Tellides, G. & Pober, J.S. Interferon-γ axis in graft arteriosclerosis. Circ. Res. 100, 622–632 (2007).
Cuffy, M.C. et al. Induction of indoleamine 2,3-dioxygenase in vascular smooth muscle cells by interferon-γ contributes to medial immunoprivilege. J. Immunol. 179, 5246–5254 (2007).
Oberle, N., Eberhardt, N., Falk, C.S., Krammer, P.H. & Suri-Payer, E. Rapid suppression of cytokine transcription in human CD4+CD25 T cells by CD4+Foxp3+ regulatory T cells: independence of IL-2 consumption, TGF-β and various inhibitors of TCR signaling. J. Immunol. 179, 3578–3587 (2007).
Lin, C.Y., Graca, L., Cobbold, S.P. & Waldmann, H. Dominant transplantation tolerance impairs CD8+ T cell function but not expansion. Nat. Immunol. 3, 1208–1213 (2002).
Sakaguchi, S., Sakaguchi, N., Asano, M., Itoh, M. & Toda, M. Immunologic self-tolerance maintained by activated T cells expressing IL-2 receptor α-chains (CD25). Breakdown of a single mechanism of self-tolerance causes various autoimmune diseases. J. Immunol. 155, 1151–1164 (1995).
Warnecke, G., Bushell, A., Nadig, S.N. & Wood, K.J. Regulation of transplant arteriosclerosis by CD25+CD4+ T cells generated to alloantigen in vivo. Transplantation 83, 1459–1465 (2007).
Liu, W. et al. CD127 expression inversely correlates with FoxP3 and suppressive function of human CD4+ Treg cells. J. Exp. Med. 203, 1701–1711 (2006).
Seddiki, N. et al. Expression of interleukin (IL)-2 and IL-7 receptors discriminates between human regulatory and activated T cells. J. Exp. Med. 203, 1693–1700 (2006).
Putnam, A.L. et al. Expansion of human regulatory T cells from patients with type 1 diabetes. Diabetes 58, 652–662 (2009).
Ermann, J. et al. Only the CD62L+ subpopulation of CD4+CD25+ regulatory T cells protects from lethal acute GVHD. Blood 105, 2220–2226 (2005).
Jutila, M.A., Watts, G., Walcheck, B. & Kansas, G.S. Characterization of a functionally important and evolutionarily well-conserved epitope mapped to the short consensus repeats of E-selectin and L-selectin. J. Exp. Med. 175, 1565–1573 (1992).
Koenen, H.J., Fasse, E. & Joosten, I. CD27/CFSE-based ex vivo selection of highly suppressive alloantigen-specific human regulatory T cells. J. Immunol. 174, 7573–7583 (2005).
Mussa, S., Choudhary, B.P. & Taggart, D.P. Radial artery conduits for coronary artery bypass grafting: current perspective. J. Thorac. Cardiovasc. Surg. 129, 250–253 (2005).
Koulack, J. et al. Development of a mouse aortic transplant model of chronic rejection. Microsurgery 16, 110–113 (1995).
Trzonkowski, P. et al. Homeostatic repopulation by CD28−CD8+ T cells in alemtuzumab-depleted kidney transplant recipients treated with reduced immunosuppression. Am. J. Transplant. 8, 338–347 (2008).
Acknowledgements
We thank the staff of the Biomedical Services Unit at the John Radcliffe Hospital for expert animal care, M. Barnardo and the staff at the Clinical Transplant Immunology Laboratory, The Oxford Transplant Centre for molecular human leukocyte antigen typing, M. Carvalho-Gaspar for advice on real-time PCR, F. Issa and R. Goto for assistance with some experiments, A. Bushell and N. Jones for valuable discussions and the cardiac surgical registrars and operating theater staff for assistance with the procurement of vessels. This work was supported by grants from The Wellcome Trust, the European Union Integrated Project, RISET, Medical Research Council UK and Garfield Weston Trust. S.N.N. is an International Society for Heart and Lung Transplantation Research Fellow; J.W. is a RISET investigator, G.W. is a Deutsche Forschungsgemeinschaft Fellow; W.Z. was supported by an unrestricted grant from Becton Dickinson; A.S. was supported by the Swedish Heart and Lung Foundation and the Swedish Research Council.
Author information
Authors and Affiliations
Contributions
S.N.N., J.W., G.W. and K.J.W. designed the experiments; S.N.N. and J.W. performed the experiments and analyzed the data; D.C.W., G.W., W.Z., S.L. and A.S. assisted with the experiments; D.P.T. provided expertise and advice along with human tissue; and S.N.N., J.W. and K.J.W. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–7 and Supplementary Table 1 (PDF 704 kb)
Rights and permissions
About this article
Cite this article
Nadig, S., Więckiewicz, J., Wu, D. et al. In vivo prevention of transplant arteriosclerosis by ex vivo–expanded human regulatory T cells. Nat Med 16, 809–813 (2010). https://doi.org/10.1038/nm.2154
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.2154
This article is cited by
-
Ex vivo-expanded human CD19+TIM-1+ regulatory B cells suppress immune responses in vivo and are dependent upon the TIM-1/STAT3 axis
Nature Communications (2022)
-
Immunological considerations and challenges for regenerative cellular therapies
Communications Biology (2021)
-
Therapeutically expanded human regulatory T-cells are super-suppressive due to HIF1A induced expression of CD73
Communications Biology (2021)
-
Intradermal injection of low dose human regulatory T cells inhibits skin inflammation in a humanized mouse model
Scientific Reports (2018)
-
Histone/protein deacetylase 11 targeting promotes Foxp3+ Treg function
Scientific Reports (2017)