Abstract
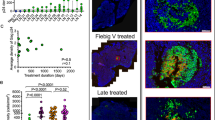
To explore the mechanism by which herpes simplex virus (HSV)-2 infection is related to HIV-1 acquisition, we conducted in situ analysis of the cellular infiltrate from sequential biopsies of HSV-2 lesions from patients on and off antiviral therapy. CD4+ and CD8+ T cells and a mixed population of plasmacytoid and myeloid dendritic cells (DCs), including cells expressing the C-type lectin receptor DC-SIGN, persisted at sites of HSV-2 reactivation for months after healing, even with daily antiviral therapy. The CD4+ T cells that persisted reacted to HSV-2 antigen, were enriched for expression of the chemokine receptor CCR5, and were contiguous to DCs expressing the interleukin-3 receptor CD123 or DC-SIGN. Ex vivo infection with a CCR5-tropic strain of HIV-1 revealed greater concentrations of integrated HIV-1 DNA in cells derived from healed genital lesion biopsies than in cells from control skin biopsies. The persistence and enrichment of HIV receptor–positive inflammatory cells in the genitalia help explain the inability of anti–HSV-2 therapy to reduce HIV acquisition.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Freeman, E.E. et al. Herpes simplex virus 2 infection increases HIV acquisition in men and women: systematic review and meta-analysis of longitudinal studies. AIDS 20, 73–83 (2006).
Wald, A. & Link, K. Risk of human immunodeficiency virus (HIV) infection in herpes simplex virus type-2 (HSV-2) seropositive persons: a meta-analysis. J. Infect. Dis. 185, 45–52 (2002).
Koelle, D.M. et al. Antigenic specificities of human CD4+ T-cell clones recovered from recurrent genital herpes simplex virus type 2 lesions. J. Virol. 68, 2803–2810 (1994).
Wald, A. et al. Frequent genital HSV-2 shedding in immunocompetent women. J. Clin. Invest. 99, 1092–1097 (1997).
Gupta, R. et al. Valacyclovir and acyclovir for suppression of shedding of herpes simplex virus in the genital tract. J. Infect. Dis. 190, 1374–1381 (2004).
Corey, L. et al. Once-daily valacyclovir to reduce the risk of transmission of genital herpes. N. Engl. J. Med. 350, 11–20 (2004).
Watson-Jones, D. et al. Effect of herpes simplex suppression on incidence of HIV among women in Tanzania. N. Engl. J. Med. 358, 1560–1571 (2008).
Celum, C. et al. Effect of aciclovir on HIV-1 acquisition in herpes simplex virus 2 seropositive women and men who have sex with men: a randomised, double-blind, placebo-controlled trial. Lancet 371, 2109–2119 (2008).
Zhu, J. et al. Virus-specific CD8+ T cells accumulate near sensory nerve endings in genital skin during subclinical HSV-2 reactivation. J. Exp. Med. 204, 595–603 (2007).
Barcy, S., Huang, M.L., Corey, L. & Koelle, D.M. Longitudinal analysis of herpes simplex virus-specific CD4+ cell clonotypes in infected tissues and blood. J. Infect. Dis. 191, 2012–2021 (2005).
Mark, K.E. et al. Rapidly cleared episodes of herpes simplex virus reactivation in immunocompetent adults. J. Infect. Dis. 198, 1141–1149 (2008).
Magaret, A.S., Wald, A., Huang, M.L., Selke, S. & Corey, L. Optimizing PCR positivity criterion for detection of herpes simplex virus DNA on skin and mucosa. J. Clin. Microbiol. 45, 1618–1620 (2007).
Koelle, D.M., Abbo, H., Peck, A., Ziegweid, K. & Corey, L. Direct recovery of herpes simplex virus (HSV)-specific T lymphocyte clones from recurrent genital HSV-2 lesions. J. Infect. Dis. 169, 956–961 (1994).
Segerer, S., Mac, K.M., Regele, H., Kerjaschki, D. & Schlondorff, D. Expression of the C–C chemokine receptor 5 in human kidney diseases. Kidney Int. 56, 52–64 (1999).
Wakim, L.M., Waithman, J., van Rooijen, N., Heath, W.R. & Carbone, F.R. Dendritic cell-induced memory T cell activation in nonlymphoid tissues. Science 319, 198–202 (2008).
Donaghy, H. et al. A role for plasmacytoid dendritic cells in the immune control of human recurrent herpes simplex. J. Virol. 83, 1952–1961 (2009).
Pope, M. et al. Conjugates of dendritic cells and memory T lymphocytes from skin facilitate productive infection with HIV-1. Cell 78, 389–398 (1994).
Masten, B.J. et al. Characterization of myeloid and plasmacytoid dendritic cells in human lung. J. Immunol. 177, 7784–7793 (2006).
Dzionek, A. et al. BDCA-2, BDCA-3, and BDCA-4: three markers for distinct subsets of dendritic cells in human peripheral blood. J. Immunol. 165, 6037–6046 (2000).
Granelli-Piperno, A., Shimeliovich, I., Pack, M., Trumpfheller, C. & Steinman, R.M. HIV-1 selectively infects a subset of nonmaturing BDCA1-positive dendritic cells in human blood. J. Immunol. 176, 991–998 (2006).
Ochoa, M.T., Loncaric, A., Krutzik, S.R., Becker, T.C. & Modlin, R.L. “Dermal dendritic cells” comprise two distinct populations: CD1+ dendritic cells and CD209+ macrophages. J. Invest. Dermatol. 128, 2225–2231 (2008).
O'Doherty, U., Swiggard, W.J. & Malim, M.H. Human immunodeficiency virus type 1 spinoculation enhances infection through virus binding. J. Virol. 74, 10074–10080 (2000).
Brussel, A. & Sonigo, P. Analysis of early human immunodeficiency virus type 1 DNA synthesis by use of a new sensitive assay for quantifying integrated provirus. J. Virol. 77, 10119–10124 (2003).
Posavad, C.M., Koelle, D.M. & Corey, L. Tipping the scales of herpes simplex virus reactivation: the important responses are local. Nat. Med. 4, 381–382 (1998).
Johnston, C. et al. Impact of HIV infection and Kaposi sarcoma on human herpesvirus-8 mucosal replication and dissemination in Uganda. PLoS One 4, e4222 (2009).
Corey, L., Adams, H.G., Brown, Z.A. & Holmes, K.K. Genital herpes simplex virus infections: clinical manifestations, course, and complications. Ann. Intern. Med. 98, 958–972 (1983).
Rebbapragada, A. et al. Negative mucosal synergy between Herpes simplex type 2 and HIV in the female genital tract. AIDS 21, 589–598 (2007).
Estes, J.D. et al. Premature induction of an immunosuppressive regulatory T cell response during acute simian immunodeficiency virus infection. J. Infect. Dis. 193, 703–712 (2006).
Weiler, A.M. et al. Genital ulcers facilitate rapid viral entry and dissemination following intravaginal inoculation with cell-associated simian immunodeficiency virus SIVmac239. J. Virol. 82, 4154–4158 (2008).
Wald, A., Zeh, J., Selke, S., Ashley, R.L. & Corey, L. Virologic characteristics of subclinical and symptomatic genital herpes infections. N. Engl. J. Med. 333, 770–775 (1995).
Benedetti, J., Corey, L. & Ashley, R. Recurrence rates in genital herpes after symptomatic first-episode infection. Ann. Intern. Med. 121, 847–854 (1994).
Castro, K.G. et al. Transmission of HIV in Belle Glade, Florida: lessons for other communities in the United States. Science 239, 193–197 (1988).
Hook, E.W. III et al. Herpes simplex virus infection as a risk factor for human immunodeficiency virus infection in heterosexuals. J. Infect. Dis. 165, 251–255 (1992).
Bailey, R.C. et al. Male circumcision for HIV prevention in young men in Kisumu, Kenya: a randomised controlled trial. Lancet 369, 643–656 (2007).
Auvert, B. et al. Randomized, controlled intervention trial of male circumcision for reduction of HIV infection risk: the ANRS 1265 Trial. PLoS Med. 2, e298 (2005).
Gray, R.H. et al. Male circumcision for HIV prevention in men in Rakai, Uganda: a randomised trial. Lancet 369, 657–666 (2007).
Koelle, D.M. et al. Clearance of HSV-2 from recurrent genital lesions correlates with infiltration of HSV-specific cytotoxic T lymphocytes. J. Clin. Invest. 101, 1500–1508 (1998).
Koelle, D.M., Huang, J., Hensel, M.T. & McClurkan, C.L. Innate immune responses to herpes simplex virus type 2 influence skin homing molecule expression by memory CD4+ lymphocytes. J. Virol. 80, 2863–2872 (2006).
Hladik, F. et al. Initial events in establishing vaginal entry and infection by human immunodeficiency virus type-1. Immunity 26, 257–270 (2007).
Diggle, P., Heagerty, P., Liang, K. & Zeger, S. Analysis of Longitudinal Data (Oxford University Press, Oxford, UK, 2002).
Acknowledgements
We thank M. Mack (University Hospital Regensburg, Germany) for the antibody to CCR5 and C. McClurkan, L. Ballweber and H. Xie for technical assistance. We also thank our dedicated study participants. This work was supported by the National Institutes of Health (R37AI042528, P01AI030731, AI50132 and HD51455) and the Tietze Foundation.
Author information
Authors and Affiliations
Contributions
J.Z. designed the study, developed the technologies, conducted the experiments, prepared the figures and wrote the draft. A. Woodward and A.K. performed the staining and quantification. T.P. developed the first data suggesting DCs are part of the inflammatory milieu in the healing biopsy stage. F.H. conducted the ex vivo organ culture studies. C.J. and M.R. enrolled subjects and conducted the biopsies. A. Wald supervised the clinic. D.M.K. designed and analyzed experiments on HSV-2 specificity of skin-derived T cells and HIV co-receptor flow cytometry. A.M. conducted the statistical analyses. L.C. and A. Wald formulated the hypothesis and designed the study. L.C. provided funding for the study and led the writing of the paper. All authors contributed to critical revisions of the paper.
Corresponding author
Rights and permissions
About this article
Cite this article
Zhu, J., Hladik, F., Woodward, A. et al. Persistence of HIV-1 receptor–positive cells after HSV-2 reactivation is a potential mechanism for increased HIV-1 acquisition. Nat Med 15, 886–892 (2009). https://doi.org/10.1038/nm.2006
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.2006
This article is cited by
-
Modeling human HSV infection via a vascularized immune-competent skin-on-chip platform
Nature Communications (2022)
-
Polymerase chain reaction for the diagnosis of herpesvirus infections in dermatology
Wiener klinische Wochenschrift (2020)
-
T cells and the skin: from protective immunity to inflammatory skin disorders
Nature Reviews Immunology (2019)
-
The immunological anatomy of the skin
Nature Reviews Immunology (2019)
-
Injection and Heterosexual Risk Behaviors for HIV Infection Among Non-gay Identifying Men Who Have Sex with Men and Women
AIDS and Behavior (2019)