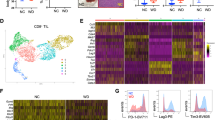
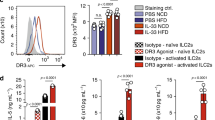
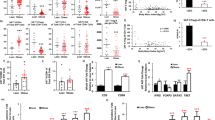
Abstract
Obesity and its associated metabolic syndromes represent a growing global challenge, yet mechanistic understanding of this pathology and current therapeutics are unsatisfactory. We discovered that CD4+ T lymphocytes, resident in visceral adipose tissue (VAT), control insulin resistance in mice with diet-induced obesity (DIO). Analyses of human tissue suggest that a similar process may also occur in humans. DIO VAT-associated T cells show severely biased T cell receptor Vα repertoires, suggesting antigen-specific expansion. CD4+ T lymphocyte control of glucose homeostasis is compromised in DIO progression, when VAT accumulates pathogenic interferon-γ (IFN-γ)-secreting T helper type 1 (TH1) cells, overwhelming static numbers of TH2 (CD4+GATA-binding protein-3 (GATA-3)+) and regulatory forkhead box P3 (Foxp3)+ T cells. CD4+ (but not CD8+) T cell transfer into lymphocyte-free Rag1-null DIO mice reversed weight gain and insulin resistance, predominantly through TH2 cells. In obese WT and ob/ob (leptin-deficient) mice, brief treatment with CD3-specific antibody or its F(ab′)2 fragment, reduces the predominance of TH1 cells over Foxp3+ cells, reversing insulin resistance for months, despite continuation of a high-fat diet. Our data suggest that the progression of obesity-associated metabolic abnormalities is under the pathophysiological control of CD4+ T cells. The eventual failure of this control, with expanding adiposity and pathogenic VAT T cells, can successfully be reversed by immunotherapy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Kahn, S.E., Hull, R.L. & Utzschneider, K.M. Mechanisms linking obesity to insulin resistance and type 2 diabetes. Nature 444, 840–846 (2006).
Déspres, J.P. & Lemieux, I. Abdominal obesity and metabolic syndrome. Nature 444, 881–887 (2006).
Dahabreh, I.J. Meta-analysis of rare events: an update and sensitivity analysis of cardiovascular events in randomized trials of rosiglitazone. Clin. Trials 5, 116–120 (2008).
Hanson, R.L., Imperatore, G., Bennett, P.H. & Knowler, W.C. Components of the 'metabolic syndrome' and incidence of type 2 diabetes. Diabetes 51, 3120–3127 (2002).
Hotamisligil, G.S. Inflammation and metabolic disorders. Nature 444, 860–867 (2006).
Kim, J.Y. et al. Obesity-associated improvements in metabolic profile through expansion of adipose tissue. J. Clin. Invest. 117, 2621–2637 (2007).
Furuhashi, M. et al. Adipocyte/macrophage fatty acid-binding proteins contribute to metabolic deterioration through actions in both macrophages and adipocytes in mice. J. Clin. Invest. 118, 2640–2650 (2008).
Kanda, H. et al. MCP-1 contributes to macrophage infiltration into adipose tissue, insulin resistance, and hepatic steatosis in obesity. J. Clin. Invest. 116, 1494–1505 (2006).
Yuan, M. et al. Reversal of obesity- and diet-induced insulin resistance with salicylates or targeted disruption of Ikkβ. Science 293, 1673–1677 (2001).
Weisberg, S.P. et al. CCR2 modulates inflammatory and metabolic effects of high-fat feeding. J. Clin. Invest. 116, 115–124 (2006).
Odegaard, J.I. et al. Macrophage-specific PPARγ controls alternative activation and improves insulin resistance. Nature 447, 1116–1120 (2007).
Tiemessen, M.M. et al. CD4+CD25+Foxp3+ regulatory T cells induce alternative activation of human monocytes/macrophages. Proc. Natl. Acad. Sci. USA 104, 19446–19451 (2007).
Martinez, F.O., Sica, A., Mantovani, A. & Locati, M. Macrophage activation and polarization. Front. Biosci. 13, 453–461 (2008).
Cintra, D.E. et al. Interleukin-10 is a protective factor against diet-induced insulin resistance in liver. J. Hepatol. 48, 628–637 (2008).
Lumeng, C.N., Bodzin, J.L. & Saltiel, A.R. Obesity induces a phenotypic switch in adipose tissue macrophage polarization. J. Clin. Invest. 117, 175–184 (2007).
Rausch, M.E., Weisberg, S., Vardhana, P. & Tortoriello, D.V. Obesity in C57BL/6J mice is characterized by adipose tissue hypoxia and cytotoxic T-cell infiltration. Int. J. Obes. (Lond.) 32, 451–463 (2008).
Kintscher, U. et al. T-lymphocyte infiltration in visceral adipose tissue. a primary event in adipose tissue inflammation and the development of obesity-mediated insulin resistance. Arterioscler. Thromb. Vasc. Biol. 28, 1304–1310 (2008).
Wu, H. et al. T-cell accumulation and regulated on activation, normal T cell expressed and secreted upregulation in adipose tissue in obesity. Circulation 115, 1029–1038 (2007).
Baker, F.J., Lee, M., Chien, Y.H. & Davis, M.M. Restricted islet-cell reactive T cell repertoire of early pancreatic islet infiltrates in NOD mice. Proc. Natl. Acad. Sci. USA 99, 9374–9379 (2002).
Turner, S.J., Doherty, P.C., McCluskey, J. & Rossjohn, J. Structural determinants of T-cell receptor bias in immunity. Nat. Rev. Immunol. 6, 883–894 (2006).
Oksenberg, J.R. et al. Limited heterogeneity of rearranged T-cell receptor V α transcripts in brains of multiple sclerosis patients. Nature 345, 344–346 (1990).
Jenkins, R.N., Nikaein, A., Zimmermann, A., Meek, K. & Lipsky, P.E. T cell receptor V β gene bias in rheumatoid arthritis. J. Clin. Invest. 92, 2688–2701 (1993).
Belghith, M. et al. TGF-β–dependent mechanisms mediate restoration of self-tolerance induced by antibodies to CD3 in overt autoimmune diabetes. Nat. Med. 9, 1202–1208 (2003).
Caspar-Bauguil, S. et al. Adipose tissues as an ancestral immune organ: site-specific change in obesity. FEBS Lett. 579, 3487–3492 (2005).
Szabo, S.J. et al. A novel transcription factor, T-bet, directs TH1 lineage commitment. Cell 100, 655–669 (2000).
Barnden, M.J., Allison, J., Heath, W.R. & Carbone, F.R. Defective TCR expression in transgenic mice constructed using cDNA-based α- and β-chain genes under the control of heterologous regulatory elements. Immunol. Cell Biol. 76, 34–40 (1998).
Padovan, E. et al. Expression of two T cell receptor α chains: dual receptor T cells. Science 262, 422–424 (1993).
Fontenot, J.D., Rasmussen, J.P., Gavin, M.A. & Rudensky, A.Y. A function for interleukin 2 in Foxp3-expressing regulatory T cells. Nat. Immunol. 6, 1142–1151 (2005).
Awasthi, A. et al. A dominant function for interleukin 27 in generating interleukin 10–producing anti-inflammatory T cells. Nat. Immunol. 8, 1380–1389 (2007).
Jankovic, D. et al. Single cell analysis reveals that IL-4 receptor/Stat6 signaling is not required for the in vivo or in vitro development of CD4+ lymphocytes with a TH2 cytokine profile. J. Immunol. 164, 3047–3055 (2000).
Shimoda, K. et al. Lack of IL-4-induced TH2 response and IgE class switching in mice with disrupted Stat6 gene. Nature 380, 630–633 (1996).
Kaplan, M.H., Schindler, U., Smiley, S.T. & Grusby, M.J. Stat6 is required for mediating responses to IL-4 and for development of TH2 cells. Immunity 4, 313–319 (1996).
Bisikirska, B., Colgan, J., Luban, J., Bluestone, J.A. & Herold, K.C. TCR stimulation with modified anti-CD3 mAb expands CD8+ T cell population and induces CD8+CD25+ Tregs. J. Clin. Invest. 115, 2904–2913 (2005).
Chatenoud, L. & Bluestone, J.A. CD3-specific antibodies: a portal to the treatment of autoimmunity. Nat. Rev. Immunol. 7, 622–632 (2007).
Alegre, M. et al. Acute toxicity of anti-CD3 monoclonal antibody in mice: a model for OKT3 first dose reactions. Transplant. Proc. 22, 1920–1921 (1990).
Alegre, M. et al. Hypothermia and hypoglycemia induced by anti-CD3 monoclonal antibody in mice: role of tumor necrosis factor. Eur. J. Immunol. 20, 707–710 (1990).
Claycombe, K., King, L.E. & Fraker, P.J. A role for leptin in sustaining lymphopoiesis and myelopoiesis. Proc. Natl. Acad. Sci. USA 105, 2017–2021 (2008).
Zeyda, M. & Stulnig, T.M. Adipose tissue macrophages. Immunol. Lett. 112, 61–67 (2007).
Rocha, V.Z. et al. Interferon-γ, a TH1 cytokine, regulates fat inflammation: a role for adaptive immunity in obesity. Circ. Res. 103, 467–476 (2008).
Korn, T. et al. Myelin-specific regulatory T cells accumulate in the CNS but fail to control autoimmune inflammation. Nat. Med. 13, 423–431 (2007).
Herold, K.C. et al. Anti-CD3 monoclonal antibody in new-onset type 1 diabetes mellitus. N. Engl. J. Med. 346, 1692–1698 (2002).
Steffens, S. & Mach, F. Adiponectin and adaptive immunity: linking the bridge from obesity to atherogenesis. Circ. Res. 102, 140–142 (2008).
Macia, L. et al. Impairment of dendritic cell functionality and steady-state number in obese mice. J. Immunol. 177, 5997–6006 (2006).
Kümpers, P. et al. Leptin is a coactivator of TGF-β in unilateral ureteral obstructive kidney disease. Am. J. Physiol. Renal Physiol. 293, F1355–F1362 (2007).
Perruche, S. et al. CD3-specific antibody–induced immune tolerance involves transforming growth factor-β from phagocytes digesting apoptotic T cells. Nat. Med. 14, 528–535 (2008).
Strissel, K.J. et al. Adipocyte death, adipose tissue remodeling, and obesity complications. Diabetes 56, 2910–2918 (2007).
Acknowledgements
These studies were supported by the Canadian Institutes of Health Research. Y.C. and G.P. were recipients of scholarships from the Canadian Institutes of Health Research and the Banting & Best Foundation. Antibodies to OT2 TCR heavy and light chain were a gift from D. Philpott (University of Toronto).
Author information
Authors and Affiliations
Contributions
S.W. and Y.C. conceived of the study and carried out the bulk of the experiments. G.P., D.T., H.T. and Y.M. performed a number of mouse experiments. R.D., Y.W. and J.Z. provided much of the TCR material, F.M. helped with adipocyte analysis, J.B. and D.D. provided metabolic studies, E.E. and D.W. generated the bulk of human data, S.W. and H.-M.D. wrote the manuscript, and H.-M.D. financed and supervised the project and the peer review process.
Corresponding author
Ethics declarations
Competing interests
H.M.D. holds shares in Afference Therapeutics, Inc., a start-up biotech corporation.
Supplementary information
Supplementary Text and Figures
Supplementary Figs. 1–7 (PDF 1088 kb)
Rights and permissions
About this article
Cite this article
Winer, S., Chan, Y., Paltser, G. et al. Normalization of obesity-associated insulin resistance through immunotherapy. Nat Med 15, 921–929 (2009). https://doi.org/10.1038/nm.2001
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.2001
This article is cited by
-
Macrophage and T cell networks in adipose tissue
Nature Reviews Endocrinology (2024)
-
Wnt10b knockdown regulates the relative balance of adipose tissue-resident T cells and inhibits white fat deposition
Molecular Biology Reports (2024)
-
Relevance and consequence of chronic inflammation for obesity development
Molecular and Cellular Pediatrics (2023)
-
Diesel Exhaust Particle (DEP)-induced glucose intolerance is driven by an intestinal innate immune response and NLRP3 activation in mice
Particle and Fibre Toxicology (2023)
-
Maternal helminth infection protects offspring from high-fat-diet-induced obesity through altered microbiota and SCFAs
Cellular & Molecular Immunology (2023)