Abstract
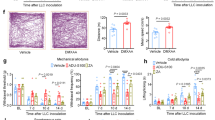
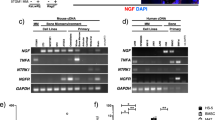
Pain is one of the most severe and debilitating symptoms associated with several forms of cancer1,2. Various types of carcinomas and sarcomas metastasize to skeletal bones and cause spontaneous bone pain and hyperalgesia, which is accompanied by bone degradation and remodeling of peripheral nerves2. Despite recent advances, the molecular mechanisms underlying the development and maintenance of cancer-evoked pain are not well understood2. Several types of non-hematopoietic tumors secrete hematopoietic colony-stimulating factors that act on myeloid cells3 and tumor cells4. Here we report that receptors and signaling mediators of granulocyte- and granulocyte-macrophage colony-stimulating factors (G-CSF and GM-CSF)3 are also functionally expressed on sensory nerves. GM-CSF sensitized nerves to mechanical stimuli in vitro and in vivo, potentiated CGRP release and caused sprouting of sensory nerve endings in the skin. Interruption of G-CSF and GM-CSF signaling in vivo led to reduced tumor growth and nerve remodeling, and abrogated bone cancer pain. The key significance of GM-CSF signaling in sensory neurons was revealed by an attenuation of tumor-evoked pain following a sensory nerve–specific knockdown of GM-CSF receptors. These results show that G-CSF and GM-CSF are important in tumor-nerve interactions and suggest that their receptors on primary afferent nerve fibers constitute potential therapeutic targets in cancer pain.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Portenoy, R.K. & Lesage, P. Management of cancer pain. Lancet 353, 1695–1700 (1999).
Honoré, P. & Mantyh, P.W. Bone cancer pain: from mechanism to model to therapy. Pain Med. 1, 303–309 (2000).
Hamilton, J.A. Colony-stimulating factors in inflammation and autoimmunity. Nat. Rev. Immunol. 8, 533–544 (2008).
Tachibana, M. & Murai, M. G-CSF production in human bladder cancer and its ability to promote autocrine growth: a review. Cytokines Cell. Mol. Ther. 4, 113–120 (1998).
Zhu, Z. et al. Nerve growth factor expression correlates with perineural invasion and pain in human pancreatic cancer. J. Clin. Oncol. 17, 2419–2428 (1999).
Wacnik, P.W. et al. Functional interactions between tumor and peripheral nerve: morphology, algogen identification, and behavioral characterization of a new murine model of cancer pain. J. Neurosci. 21, 9355–9366 (2001).
Schäbitz, W.R. et al. Neuroprotective effect of granulocyte colony-stimulating factor after focal cerebral ischemia. Stroke 34, 745–751 (2003).
Kasper, B. et al. Severe congenital neutropenia patients with point mutations in the granulocyte colony-stimulating factor (G-CSF) receptor mRNA express a normal G-CSF receptor protein. Blood 90, 2839–2841 (1997).
Lehtonen, A., Matikainen, S., Miettinen, M. & Julkunen, I. Granulocyte-macrophage colony-stimulating factor (GM-CSF)-induced STAT5 activation and target-gene expression during human monocyte/macrophage differentiation. J. Leukoc. Biol. 71, 511–519 (2002).
Monfardini, C. et al. Construction and binding kinetics of a soluble granulocyte-macrophage colony-stimulating factor receptor alpha-chain-Fc fusion protein. J. Biol. Chem. 273, 7657–7667 (1998).
de Groot, R.P., Coffer, P.J. & Koenderman, L. Regulation of proliferation, differentiation and survival by the IL-3/IL-5/GM-CSF receptor family. Cell. Signal. 10, 619–628 (1998).
Schneider, A. et al. The hematopoietic factor G-CSF is a neuronal ligand that counteracts programmed cell death and drives neurogenesis. J. Clin. Invest. 115, 2083–2098 (2005).
Giron-Michel, J. et al. Detection of a functional hybrid receptor γc/GM-CSFRβ in human hematopoietic CD34+ cells. J. Exp. Med. 197, 763–775 (2003).
Julius, D. & Basbaum, A.I. Molecular mechanisms of nociception. Nature 413, 203–210 (2001).
Honoré, E. The neuronal background K2P channels: focus on TREK1. Nat. Rev. Neurosci. 8, 251–261 (2007).
Cain, D.M. et al. Functional interactions between tumor and peripheral nerve: changes in excitability and morphology of primary afferent fibers in a murine model of cancer pain. J. Neurosci. 21, 9367–9376 (2001).
McIntyre, G.J. & Fanning, G.C. Design and cloning strategies for constructing shRNA expression vectors. BMC Biotechnol. 6, 1 (2006).
Glatzel, M. et al. Adenoviral and adeno-associated viral transfer of genes to the peripheral nervous system. Proc. Natl. Acad. Sci. USA 97, 442–447 (2000).
Lindsay, T.H. et al. Pancreatic cancer pain and its correlation with changes in tumor vasculature, macrophage infiltration, neuronal innervation, body weight and disease progression. Pain 119, 233–246 (2005).
Davar, G. Endothelin-1 and metastatic cancer pain. Pain Med. 2, 24–27 (2001).
Wacnik, P.W., Eikmeier, L.J., Simone, D.A., Wilcox, G.L. & Beitz, A.J. Nociceptive characteristics of tumor necrosis factor-α in naive and tumor-bearing mice. Neuroscience 132, 479–491 (2005).
Owen, J.L. et al. The expression of CCL2 by T lymphocytes of mammary tumor bearers: role of tumor-derived factors. Cell. Immunol. 235, 122–135 (2005).
Dedhar, S., Gaboury, L., Galloway, P. & Eaves, C. Human granulocyte-macrophage colony-stimulating factor is a growth factor active on a variety of cell types of nonhemopoietic origin. Proc. Natl. Acad. Sci. USA 85, 9253–9257 (1988).
Danis, V.A., Franic, G.M., Rathjen, D.A. & Brooks, P.M. Effects of granulocyte-macrophage colony-stimulating factor (GM-CSF), IL-2, interferon-γ (IFN-γ), tumour necrosis factor-α (TNF-α) and IL-6 on the production of immunoreactive IL-1 and TNF-α by human monocytes. Clin. Exp. Immunol. 85, 143–150 (1991).
Salh, B., Hoeflick, K., Kwan, W. & Pelech, S. Granulocyte-macrophage colony-stimulating factor and interleukin-3 potentiate interferon-γ-mediated endothelin production by human monocytes: role of protein kinase C. Immunology 95, 473–479 (1998).
Khasabova, I.A. et al. Chemical interactions between fibrosarcoma cancer cells and sensory neurons contribute to cancer pain. J. Neurosci. 27, 10289–10298 (2007).
Tigue, C.C. et al. Granulocyte-colony stimulating factor administration to healthy individuals and persons with chronic neutropenia or cancer: an overview of safety considerations from the Research on Adverse Drug Events and Reports project. Bone Marrow Transplant. 40, 185–192 (2007).
Waller, E.K. The role of sargramostim (rhGM-CSF) as immunotherapy. Oncologist 12 Suppl 2, 22–26 (2007).
Lindemann, A. et al. Hematologic effects of recombinant human granulocyte colony-stimulating factor in patients with malignancy. Blood 74, 2644–2651 (1989).
Vadhan-Raj, S. et al. Effects of recombinant human granulocyte-macrophage colony-stimulating factor in patients with myelodysplastic syndromes. N. Engl. J. Med. 317, 1545–1552 (1987).
Angst, M.S. et al. Cytokine profile in human skin in response to experimental inflammation, noxious stimulation, and administration of a COX-inhibitor: a microdialysis study. Pain 139, 15–27 (2008).
Carvalho, B., Clark, D.J. & Angst, M.S. Local and systemic release of cytokines, nerve growth factor, prostaglandin E2, and substance P in incisional wounds and serum following cesarean delivery. J. Pain 9, 650–657 (2008).
Cook, A.D., Braine, E.L., Campbell, I.K., Rich, M.J. & Hamilton, J.A. Blockade of collagen-induced arthritis post-onset by antibody to granulocyte-macrophage colony-stimulating factor (GM-CSF): requirement for GM-CSF in the effector phase of disease. Arthritis Res. 3, 293–298 (2001).
Hansen, G. et al. The structure of the GM-CSF receptor complex reveals a distinct mode of cytokine receptor activation. Cell 134, 496–507 (2008).
Acknowledgements
The authors thank H.-J. Wrede, C. Harding-Rose and D. Baumgartl-Ahlert for technical assistance and R. LeFaucheur for secretarial help. This work was supported by grants from the Association of International Cancer Research and the Landesstiftung Baden-Württemberg (Program RNAi) to R.K. and by US National Institutes of Health grants DA11471 and CA91007 to D.A.S.
Author information
Authors and Affiliations
Contributions
M. Schweizerhof performed the largest portion of the experiments and analyzed data; S.S., M.K. and C.N. performed several experiments and analyzed data; V.G., N.A., M. Schmelz, C.W.M., K.K.B. and S.B. performed some experiments and analyzed data; A.D. contributed knowledge of mouse models; D.A.S. contributed knowledge of mouse models and helped with some experiments and with the writing of the manuscript; R.K. designed and supervised the study, analyzed data and wrote the manuscript.
Corresponding author
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–3 and Supplementary Methods (PDF 460 kb)
Rights and permissions
About this article
Cite this article
Schweizerhof, M., Stösser, S., Kurejova, M. et al. Hematopoietic colony–stimulating factors mediate tumor-nerve interactions and bone cancer pain. Nat Med 15, 802–807 (2009). https://doi.org/10.1038/nm.1976
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.1976
This article is cited by
-
Neuropathic pain caused by miswiring and abnormal end organ targeting
Nature (2022)
-
Neuropathogenicity of non-viable Borrelia burgdorferi ex vivo
Scientific Reports (2022)
-
Nociceptive nerves regulate haematopoietic stem cell mobilization
Nature (2021)
-
The impact of Semaphorin 4C/Plexin-B2 signaling on fear memory via remodeling of neuronal and synaptic morphology
Molecular Psychiatry (2021)
-
Loss of POMC-mediated antinociception contributes to painful diabetic neuropathy
Nature Communications (2021)