Abstract
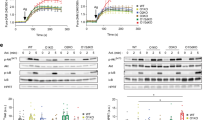
CRACM1 (also called Orai1) constitutes the pore subunit of store-operated calcium release–activated calcium channels. A point mutation in the gene encoding CRACM1 is associated with severe combined immunodeficiency disease in humans. Here we generated CRACM1-deficient mice in which β-galactosidase activity 'reported' CRACM1 expression. CRACM1-deficient mice were smaller in size. Mast cells derived from CRACM1-deficient mice showed grossly defective degranulation and cytokine secretion, and the allergic reactions elicited in vivo were inhibited in CRACM1-deficient mice. We detected robust CRACM1 expression in skeletal muscles and some regions of the brain, heart and kidney but not in the lymphoid regions of thymus and spleen. In contrast, we found CRACM2 expression to be much higher in mouse T cells. In agreement with those findings, the store-operated calcium influx and development and proliferation of CRACM1-deficient T cells was unaffected. Thus, CRACM1 is crucial in mouse mast cell effector function, but mouse T cell calcium release–activated calcium channels are functional in the absence of CRACM1.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Hoth, M. & Penner, R. Depletion of intracellular calcium stores activates a calcium current in mast cells. Nature 355, 353–356 (1992).
Parekh, A.B. & Putney, J.W., Jr. Store-operated calcium channels. Physiol. Rev. 85, 757–810 (2005).
Vig, M. et al. CRACM1 is a plasma membrane protein essential for store-operated Ca2+ entry. Science 312, 1220–1223 (2006).
Feske, S. et al. A mutation in Orai1 causes immune deficiency by abrogating CRAC channel function. Nature 441, 179–185 (2006).
Zhang, S.L. et al. Genome-wide RNAi screen of Ca2+ influx identifies genes that regulate Ca2+ release-activated Ca2+ channel activity. Proc. Natl. Acad. Sci. USA 103, 9357–9362 (2006).
Vig, M. et al. CRACM1 multimers form the ion-selective pore of the CRAC channel. Curr. Biol. 16, 2073–2079 (2006).
Yeromin, A.V. et al. Molecular identification of the CRAC channel by altered ion selectivity in a mutant of Orai. Nature 443, 226–229 (2006).
Prakriya, M. et al. Orai1 is an essential pore subunit of the CRAC channel. Nature 443, 230–233 (2006).
Turner, H. & Kinet, J.P. Signalling through the high-affinity IgE receptor Fc epsilonRI. Nature 402, B24–B30 (1999).
Parravicini, V. et al. Fyn kinase initiates complementary signals required for IgE-dependent mast cell degranulation. Nat. Immunol. 3, 741–748 (2002).
Nadler, M.J. & Kinet, J.P. Uncovering new complexities in mast cell signaling. Nat. Immunol. 3, 707–708 (2002).
Kraft, S. & Kinet, J.P. New developments in FcεRI regulation, function and inhibition. Nat. Rev. Immunol. 7, 365–378 (2007).
Chang, W.C. & Parekh, A.B. Close functional coupling between Ca2+ release-activated Ca2+ channels, arachidonic acid release, and leukotriene C4 secretion. J. Biol. Chem. 279, 29994–29999 (2004).
Chang, W.C., Nelson, C. & Parekh, A.B. Ca2+ influx through CRAC channels activates cytosolic phospholipase A2, leukotriene C4 secretion, and expression of c-fos through ERK-dependent and -independent pathways in mast cells. FASEB J. 20, 2381–2383 (2006).
Feske, S. et al. Severe combined immunodeficiency due to defective binding of the nuclear factor of activated T cells in T lymphocytes of two male siblings. Eur. J. Immunol. 26, 2119–2126 (1996).
Skarnes, W.C. Gene trapping methods for the identification and functional analysis of cell surface proteins in mice. Methods Enzymol. 328, 592–615 (2000).
Stryke, D. et al. BayGenomics: a resource of insertional mutations in mouse embryonic stem cells. Nucleic Acids Res. 31, 278–281 (2003).
Kawakami, T. & Galli, S.J. Regulation of mast-cell and basophil function and survival by IgE. Nat. Rev. Immunol. 2, 773–786 (2002).
Morgan, A.J. & Jacob, R. Ionomycin enhances Ca2+ influx by stimulating store-regulated cation entry and not by a direct action at the plasma membrane. Biochem. J. 300, 665–672 (1994).
Klein, M. et al. Specific and redundant roles for NFAT transcription factors in the expression of mast cell-derived cytokines. J. Immunol. 177, 6667–6674 (2006).
Kalesnikoff, J. et al. SHIP negatively regulates IgE + antigen-induced IL-6 production in mast cells by inhibiting NF-κB activity. J. Immunol. 168, 4737–4746 (2002).
Wershil, B.K., Mekori, Y.A., Murakami, T. & Galli, S.J. 125I-fibrin deposition in IgE-dependent immediate hypersensitivity reactions in mouse skin. Demonstration of the role of mast cells using genetically mast cell-deficient mice locally reconstituted with cultured mast cells. J. Immunol. 139, 2605–2614 (1987).
Fleming, T.J. et al. Negative regulation of FcεRI-mediated degranulation by CD81. J. Exp. Med. 186, 1307–1314 (1997).
DeHaven, W.I., Smyth, J.T., Boyles, R.R. & Putney, J.W., Jr. Calcium inhibition and calcium potentiation of Orai1, Orai2, and Orai3 calcium release-activated calcium channels. J. Biol. Chem. 282, 17548–17556 (2007).
Blank, U. & Rivera, J. The ins and outs of IgE-dependent mast-cell exocytosis. Trends Immunol. 25, 266–273 (2004).
Penner, R. & Neher, E. Secretory responses of rat peritoneal mast cells to high intracellular calcium. FEBS Lett. 226, 307–313 (1988).
Penner, R., Matthews, G. & Neher, E. Regulation of calcium influx by second messengers in rat mast cells. Nature 334, 499–504 (1988).
Cooper, D.M., Yoshimura, M., Zhang, Y., Chiono, M. & Mahey, R. Capacitative Ca2+ entry regulates Ca2+-sensitive adenylyl cyclases. Biochem. J. 297, 437–440 (1994).
Fagan, K.A., Mons, N. & Cooper, D.M. Dependence of the Ca2+-inhibitable adenylyl cyclase of C6–2B glioma cells on capacitative Ca2+ entry. J. Biol. Chem. 273, 9297–9305 (1998).
Fagan, K.A., Mahey, R. & Cooper, D.M. Functional co-localization of transfected Ca2+-stimulable adenylyl cyclases with capacitative Ca2+ entry sites. J. Biol. Chem. 271, 12438–12444 (1996).
Rettig, J. & Neher, E. Emerging roles of presynaptic proteins in Ca++-triggered exocytosis. Science 298, 781–785 (2002).
Chapman, E.R. Synaptotagmin: a Ca2+ sensor that triggers exocytosis? Nat. Rev. Mol. Cell Biol. 3, 498–508 (2002).
Serfling, E. et al. The role of NF-AT transcription factors in T cell activation and differentiation. Biochim. Biophys. Acta 1498, 1–18 (2000).
Macian, F. NFAT proteins: key regulators of T-cell development and function. Nat. Rev. Immunol. 5, 472–484 (2005).
Lewis, R.S. Calcium oscillations in T-cells: mechanisms and consequences for gene expression. Biochem. Soc. Trans. 31, 925–929 (2003).
Gonzalez-Espinosa, C. et al. Preferential signaling and induction of allergy-promoting lymphokines upon weak stimulation of the high affinity IgE receptor on mast cells. J. Exp. Med. 197, 1453–1465 (2003).
Gwack, Y. et al. Biochemical and functional characterization of Orai proteins. J. Biol. Chem. 282, 16232–16243 (2007).
Lis, A. et al. CRACM1, CRACM2, and CRACM3 are store-operated Ca2+ channels with distinct functional properties. Curr. Biol. 17, 794–800 (2007).
Martens, S., Kozlov, M.M. & McMahon, H.T. How synaptotagmin promotes membrane fusion. Science 316, 1205–1208 (2007).
Bird, G.S. & Putney, J.W., Jr. Capacitative calcium entry supports calcium oscillations in human embryonic kidney cells. J. Physiol. (Lond.) 562, 697–706 (2005).
Acknowledgements
We thank A. Dani for discussions and help with X-gal staining and analysis; M.K. Huberson for help with genotyping; B. Koller for help with the generation and analysis of in Cracm1−/− mice; and C. Bortner (National Institute of Environmental Health Sciences–National Institutes of Health, Department of Health and Human Services) for help with flow cytometry. Supported by the National Institutes of Health (GM 053950 to J.-P.K.), the Irvington Institute (M.V.) and the intramural program of the National Institutes of Health, National Institute of Environmental Health Sciences.
Author information
Authors and Affiliations
Contributions
M.V. designed and executed the study and wrote the paper; W.I.D. and G.S.B. did the Ca2+ imaging and electrophysiology experiments; J.M.B. did the retroviral reconstitutions; H.W., P.E.R. and A.B.H. helped with genotyping, real-time PCR and T cell cytokine analysis, respectively; and M.-H.J., J.W.P. and J.-P.K. provided overall advice on the project.
Corresponding author
Ethics declarations
Competing interests
M.V. and J.-P.K. are consultants for Synta Pharmaceuticals.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–5, Table 1 and Methods (PDF 149 kb)
Rights and permissions
About this article
Cite this article
Vig, M., DeHaven, W., Bird, G. et al. Defective mast cell effector functions in mice lacking the CRACM1 pore subunit of store-operated calcium release–activated calcium channels. Nat Immunol 9, 89–96 (2008). https://doi.org/10.1038/ni1550
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni1550
This article is cited by
-
Physiological functions of calcium signaling via Orai1 in cancer
The Journal of Physiological Sciences (2023)
-
Direct control of store-operated calcium channels by ultrafast laser
Cell Research (2021)
-
Calcium signalling in T cells
Nature Reviews Immunology (2019)
-
The Pyrazole Derivative BTP2 Attenuates IgG Immune Complex-induced Inflammation
Inflammation (2018)
-
RETRACTED ARTICLE: Inhibition of airway inflammation in a cockroach allergen model of asthma by agonists of miRNA-33b
Scientific Reports (2017)