Abstract
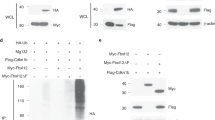
RhoH is a hematopoietic-specific, GTPase-deficient member of the Rho GTPase family with unknown physiological function. Here we demonstrate that Rhoh−/− mice have impaired T cell receptor (TCR)–mediated thymocyte selection and maturation, resulting in T cell deficiency. RhoH deficiency resulted in defective CD3ζ phosphorylation, impaired translocation of the signaling molecule Zap70 to the immunological synapse and reduced activation of Zap70-mediated signaling in thymic and peripheral T cells. Proteomic analyses demonstrated that RhoH is a component of TCR signaling and is required for recruitment of Zap70 to the TCR through interaction with RhoH noncanonical immunoreceptor tyrosine-based activation motifs (ITAMs). In vivo reconstitution studies also demonstrated that RhoH function depends on phosphorylation of the RhoH ITAMs. These findings suggest that RhoH is a critical regulator of thymocyte development and TCR signaling by mediating recruitment and activation of Zap70.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Rothenberg, E.V. & Taghon, T. Molecular genetics of T cell development. Annu. Rev. Immunol. 23, 601–649 (2005).
Mombaerts, P. et al. Mutations in T-cell antigen receptor genes α and β block thymocyte development at different stages. Nature 360, 225–231 (1992).
Fehling, H.J., Krotkova, A., Saint-Ruf, C. & von Boehmer, H. Crucial role of the pre-T-cell receptor α gene in development of αβ but not γδ T cells. Nature 375, 795–798 (1995).
Robey, E. & Fowlkes, B.J. Selective events in T cell development. Annu. Rev. Immunol. 12, 675–705 (1994).
Cantrell, D.A. GTPases and T cell activation. Immunol. Rev. 192, 122–130 (2003).
Dallery, E. et al. TTF, a gene encoding a novel small G protein, fuses to the lymphoma- associated LAZ3 gene by t(3;4) chromosomal translocation. Oncogene 10, 2171–2178 (1995).
Pasqualucci, L. et al. Hypermutation of multiple proto-oncogenes in B-cell diffuse large-cell lymphomas. Nature 412, 341–346 (2001).
Li, X. et al. The hematopoiesis-specific GTP-binding protein RhoH is GTPase deficient and modulates activities of other Rho GTPases by an inhibitory function. Mol. Cell. Biol. 22, 1158–1171 (2002).
Gu, Y., Jasti, A.C., Jansen, M. & Siefring, J.E. RhoH, a hematopoietic-specific Rho GTPase, regulates proliferation, survival, migration, and engraftment of hematopoietic progenitor cells. Blood 105, 1467–1475 (2005).
Cherry, L.K., Li, X., Schwab, P., Lim, B. & Klickstein, L.B. RhoH is required to maintain the integrin LFA-1 in a nonadhesive state on lymphocytes. Nat. Immunol. 5, 961–967 (2004).
Lahousse, S. et al. Structural features of hematopoiesis-specific RhoH/ARHH gene: high diversity of 5′-UTR in different hematopoietic lineages suggests a complex post-transcriptional regulation. Gene 343, 55–68 (2004).
Littman, D.R., Davis, C.B., Killeen, N. & Xu, H. Signal transduction during T cell development. Adv. Exp. Med. Biol. 365, 63–71 (1994).
Gauen, L.K. et al. Interactions of p59fyn and Zap70 with T-cell receptor activation motifs: defining the nature of a signalling motif. Mol. Cell. Biol. 14, 3729–3741 (1994).
Kane, L.P., Lin, J. & Weiss, A. Signal transduction by the TCR for antigen. Curr. Opin. Immunol. 12, 242–249 (2000).
Rincon, M. MAP-kinase signaling pathways in T cells. Curr. Opin. Immunol. 13, 339–345 (2001).
Negishi, I. et al. Essential role for Zap70 in both positive and negative selection of thymocytes. Nature 376, 435–438 (1995).
Elder, M.E. et al. Human severe combined immunodeficiency due to a defect in Zap70, a T cell tyrosine kinase. Science 264, 1596–1599 (1994).
Arpaia, E., Shahar, M., Dadi, H., Cohen, A. & Roifman, C.M. Defective T cell receptor signaling and CD8+ thymic selection in humans lacking Zap70 kinase. Cell 76, 947–958 (1994).
Boncristiano, M. et al. Defective recruitment and activation of Zap70 in common variable immunodeficiency patients with T cell defects. Eur. J. Immunol. 30, 2632–2638 (2000).
Shortman, K. Cellular aspects of early T-cell development. Curr. Opin. Immunol. 4, 140–146 (1992).
Godfrey, D.I., Kennedy, J., Suda, T. & Zlotnik, A. A developmental pathway involving four phenotypically and functionally distinct subsets of CD3–CD4-CD8- triple-negative adult mouse thymocytes defined by CD44 and CD25 expression. J. Immunol. 150, 4244–4252 (1993).
Fowlkes, B.J., Edison, L., Mathieson, B.J. & Chused, T.M. Early T lymphocytes. Differentiation in vivo of adult intrathymic precursor cells. J. Exp. Med. 162, 802–822 (1985).
Punt, J.A., Osborne, B.A., Takahama, Y., Sharrow, S.O. & Singer, A. Negative selection of CD4+CD8+ thymocytes by T cell receptor-induced apoptosis requires a costimulatory signal that can be provided by CD28. J. Exp. Med. 179, 709–713 (1994).
Yamashita, I., Nagata, T., Tada, T. & Nakayama, T. CD69 cell surface expression identifies developing thymocytes which audition for T cell antigen receptor-mediated positive selection. Int. Immunol. 5, 1139–1150 (1993).
Swat, W., Dessing, M., von Boehmer, H. & Kisielow, P. CD69 expression during selection and maturation of CD4+8+ thymocytes. Eur. J. Immunol. 23, 739–746 (1993).
Pircher, H. et al. Molecular analysis of the antigen receptor of virus-specific cytotoxic T cells and identification of a new Vα family. Eur. J. Immunol. 17, 1843–1846 (1987).
Shinkai, Y. et al. RAG-2-deficient mice lack mature lymphocytes owing to inability to initiate V(D)J rearrangement. Cell 68, 855–867 (1992).
Sloan-Lancaster, J., Shaw, A.S., Rothbard, J.B. & Allen, P.M. Partial T cell signaling: altered phospho-ζ and lack of Zap70 recruitment in APL-induced T cell anergy. Cell 79, 913–922 (1994).
van Oers, N.S., Killeen, N. & Weiss, A. Zap70 is constitutively associated with tyrosine-phosphorylated TCRζ in murine thymocytes and lymph node T cells. Immunity 1, 675–685 (1994).
Reth, M. Antigen receptor tail clue. Nature 338, 383–384 (1989).
Cambier, J.C. Antigen and Fc receptor signaling. The awesome power of the immunoreceptor tyrosine-based activation motif (ITAM). J. Immunol. 155, 3281–3285 (1995).
Waksman, G., Kumaran, S. & Lubman, O. SH2 domains: role, structure and implications for molecular medicine. Expert Rev. Mol. Med. 6, 1–18 (2004).
Lee, K.H. et al. T cell receptor signaling precedes immunological synapse formation. Science 295, 1539–1542 (2002).
Yokosuka, T. et al. Newly generated T cell receptor microclusters initiate and sustain T cell activation by recruitment of Zap70 and SLP-76. Nat. Immunol. 6, 1253–1262 (2005).
Pelosi, M. et al. Tyrosine 319 in the interdomain B of Zap70 is a binding site for the Src homology 2 domain of Lck. J. Biol. Chem. 274, 14229–14237 (1999).
Zhang, W., Sloan-Lancaster, J., Kitchen, J., Trible, R.P. & Samelson, L.E. LAT: the Zap70 tyrosine kinase substrate that links T cell receptor to cellular activation. Cell 92, 83–92 (1998).
Li, B. et al. Role of the guanosine triphosphatase Rac2 in T helper 1 cell differentiation. Science 288, 2219–2222 (2000).
Gu, Y. et al. Hematopoietic cell regulation by Rac1 and Rac2 guanosine triphosphatases. Science 302, 445–449 (2003).
Walmsley, M.J. et al. Critical roles for Rac1 and Rac2 GTPases in B cell development and signaling. Science 302, 459–462 (2003).
Azzam, H.S. et al. CD5 expression is developmentally regulated by T cell receptor (TCR) signals and TCR avidity. J. Exp. Med. 188, 2301–2311 (1998).
Molina, T.J. et al. Profound block in thymocyte development in mice lacking p56lck. Nature 357, 161–164 (1992).
Fujikawa, K. et al. Vav1/2/3-null mice define an essential role for Vav family proteins in lymphocyte development and activation but a differential requirement in MAPK signaling in T and B cells. J. Exp. Med. 198, 1595–1608 (2003).
Kersh, E.N., Shaw, A.S. & Allen, P.M. Fidelity of T cell activation through multistep T cell receptor ζ phosphorylation. Science 281, 572–575 (1998).
Chan, A.C. & Shaw, A.S. Regulation of antigen receptor signal transduction by protein tyrosine kinases. Curr. Opin. Immunol. 8, 394–401 (1996).
Nusser, N. et al. Serine phosphorylation differentially affects RhoA binding to effectors: implications to NGF-induced neurite outgrowth. Cell. Signal. 18, 704–714 (2006).
Filippi, M.D. et al. Localization of Rac2 via the C terminus and aspartic acid 150 specifies superoxide generation, actin polarity and chemotaxis in neutrophils. Nat. Immunol. 5, 744–751 (2004).
Henning, S.W., Galandrini, R., Hall, A. & Cantrell, D.A. The GTPase Rho has a critical regulatory role in thymus development. EMBO J. 16, 2397–2407 (1997).
Delaguillaumie, A., Lagaudriere-Gesbert, C., Popoff, M.R. & Conjeaud, H. Rho GTPases link cytoskeletal rearrangements and activation processes induced via the tetraspanin CD82 in T lymphocytes. J. Cell Sci. 115, 433–443 (2002).
Hanenberg, H. et al. Optimization of fibronectin-assisted retroviral gene transfer into human CD34+ hematopoietic cells. Hum. Gene Ther. 8, 2193–2206 (1997).
Wahlers, A. et al. Influence of multiplicity of infection and protein stability on retroviral vector-mediated gene expression in hematopoietic cells. Gene Ther. 8, 477–486 (2001).
Wakabayashi, Y. et al. Bcl11b is required for differentiation and survival of αβ T lymphocytes. Nat. Immunol. 4, 533–539 (2003).
Movassagh, M. et al. Retrovirus-mediated gene transfer into T cells: 95% transduction efficiency without further in vitro selection. Hum. Gene Ther. 11, 1189–1200 (2000).
Kumaraguru, U., Rouse, R.J., Nair, S.K., Bruce, B.D. & Rouse, B.T. Involvement of an ATP-dependent peptide chaperone in cross-presentation after DNA immunization. J. Immunol. 165, 750–759 (2000).
Acknowledgements
We thank D. Schumann and Y. Yuan (Genome Research Institute, University of Cincinnati, Cincinnati, Ohio) for assistance with mass spectrometry analysis; A.S. Shaw (Washington University School of Medicine, St. Louis. Missouri) for the pGEM3Z-Zap70, pGEM3Z-Zap70ΔN-SH2 and pGEM3Z-Zap70ΔC-SH2 constructs; C.T. Baldari (University of Siena, Siena, Italy) for the constitutively active Lck construct; P. Marrack (National Jewish Medical and Research Center, Denver, Colorado) for breeding pairs of p14 lymphocytic choriomeningitis virus TCR–transgenic mice; S. Li, J. Bailey and V. Summer (Cincinnati Children's Hospital, Cincinnati, Ohio) for technical assistance; and Y. Zheng for discussions and critical comments on the manuscript. Supported by the National Cancer Institute (KO1 CA107110 to Y.G.) and the US National Institutes of Health (RO1 DK62757 to D.A.W. and RO1 CA113969).
Author information
Authors and Affiliations
Contributions
Y.G. designed and did experiments and wrote the draft of the paper; H.-D.C. did experiments; J.E.S. and A.C.J. assisted in experiments; D.A.H. provided advice and directed some experiments; and D.A.W. provided oversight for all experiments, designed experiments and edited the draft paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
Gene-targeted deletion of the murine Rhoh allele. (PDF 756 kb)
Supplementary Fig. 2
Viability and proliferation of WT and Rhoh−/− thymocytes. (PDF 337 kb)
Supplementary Fig. 3
Thymic cellularity and distribution of CD4+CD8+ thymocyte subsets in p14tg/+ Rhoh−/− mice. (PDF 163 kb)
Supplementary Fig. 4
Impaired phosophorylation and recruitment of Zap70 to the immunological synapse in Rhoh−/− peripheral T cells. (PDF 968 kb)
Supplementary Table 1
T and B lymphocyte populations in Rhoh−/− mice. (PDF 84 kb)
Supplementary Table 2
Engraftment and T cell reconstitution of the RhoH-transduced Rhoh−/− BM cells in Rag2−/− recipient mice. (PDF 87 kb)
Rights and permissions
About this article
Cite this article
Gu, Y., Chae, HD., Siefring, J. et al. RhoH GTPase recruits and activates Zap70 required for T cell receptor signaling and thymocyte development. Nat Immunol 7, 1182–1190 (2006). https://doi.org/10.1038/ni1396
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni1396
This article is cited by
-
Molecular basis and current insights of atypical Rho small GTPase in cancer
Molecular Biology Reports (2024)
-
The neuroimmune axis of Alzheimer’s disease
Genome Medicine (2023)
-
T cell receptor signaling for γδT cell development
Inflammation and Regeneration (2019)
-
Genome-wide association study of Mycobacterium avium subspecies Paratuberculosis infection in Chinese Holstein
BMC Genomics (2018)
-
An RNAi screen of Rho signalling networks identifies RhoH as a regulator of Rac1 in prostate cancer cell migration
BMC Biology (2018)