Abstract
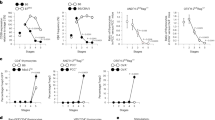
The pre–T cell receptor (TCR) is crucial for early T cell development and is proposed to function in a ligand-independent way. However, the molecular mechanism underlying the autonomous signals remains elusive. Here we show that the pre-TCR complex spontaneously formed oligomers. Specific charged residues in the extracellular domain of the pre-TCR α-chain mediated formation of the oligomers in vitro. Alteration of these residues eliminated the ability of the pre-TCR α-chain to support pre-TCR signaling in vivo. Dimerization but not raft localization of CD3ε was sufficient to simulate pre-TCR function and promote β-selection. These results suggest that the pre-TCR complex can deliver its signal autonomously through oligomerization of the pre-TCR α-chain mediated by charged residues.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Change history
09 December 2005
In the version of this article initially published online, the fifth subheading in the Results section was incorrect. The error has been corrected for the HTML and print versions of the article.
Notes
*Note: In the version of this article initially published online, the fifth subheading in the Results section was incorrect. It should read “Raft-targeted CD3ε fails to bypass β-selection.” The error has been corrected for the HTML and print versions of the article.
References
Fehling, H.J., Krotkova, A., Saint-Ruf, C. & von Boehmer, H. Crucial role of the pre-T-cell receptor α gene in development of αβ but not γδ T cells. Nature 375, 795–798 (1995).
Irving, B.A., Alt, F.W. & Killeen, N. Thymocyte development in the absence of pre-T cell receptor extracellular immunoglobulin domains. Science 280, 905–908 (1998).
Saint-Ruf, C. et al. Different initiation of pre-TCR and γδTCR signalling. Nature 406, 524–527 (2000).
Aifantis, I. et al. A critical role for the cytoplasmic tail of pTα in T lymphocyte development. Nat. Immunol. 3, 483–488 (2002).
Borowski, C., Li, X., Aifantis, I., Gounari, F. & von Boehmer, H. Pre-TCRα and TCRα are not interchangeable partners of TCRβ during T lymphocyte development. J. Exp. Med. 199, 607–615 (2004).
Haks, M.C. et al. Low activation threshold as a mechanism for ligand-independent signaling in pre-T cells. J. Immunol. 170, 2853–2861 (2003).
Huang, C.Y. & Kanagawa, O. Impact of early expression of TCRα chain on thymocyte development. Eur. J. Immunol. 34, 1532–1541 (2004).
Levelt, C.N. et al. Regulation of thymocyte development through CD3: functional dissociation between p56lck and CD3σ in early thymic selection. Immunity 3, 215–222 (1995).
Shinkai, Y. & Alt, F.W. CD3 ε-mediated signals rescue the development of CD4+CD8+ thymocytes in RAG-2−/− mice in the absence of TCRβ chain expression. Int. Immunol. 6, 995–1001 (1994).
Shinkai, Y., Ma, A., Cheng, H.L. & Alt, F.W. CD3ε and CD3ζ cytoplasmic domains can independently generate signals for T cell development and function. Immunity 2, 401–411 (1995).
Malissen, M. et al. Altered T cell development in mice with a targeted mutation of the CD3ε gene. EMBO J. 14, 4641–4653 (1995).
DeJarnette, J.B. et al. Specific requirement for CD3ε in T cell development. Proc. Natl. Acad. Sci. USA 95, 14909–14914 (1998).
Panigada, M. et al. Constitutive endocytosis and degradation of the pre-T cell receptor. J. Exp. Med. 195, 1585–1597 (2002).
Yoshimura, A., Longmore, G. & Lodish, H.F. Point mutation in the exoplasmic domain of the erythropoietin receptor resulting in hormone-independent activation and tumorigenicity. Nature 348, 647–649 (1990).
Saint-Ruf, C. et al. Analysis and expression of a cloned pre-T cell receptor gene. Science 266, 1208–1212 (1994).
Wang, J. et al. Atomic structure of an αβ T cell receptor (TCR) heterodimer in complex with an anti-TCR Fab fragment derived from a mitogenic antibody. EMBO J. 17, 10–26 (1998).
Boerth, N.J. et al. Recruitment of SLP-76 to the membrane and glycolipid-enriched membrane microdomains replaces the requirement for linker for activation of T cells in T cell receptor signaling. J. Exp. Med. 192, 1047–1058 (2000).
Yamasaki, S., Ishikawa, E., Kohno, M. & Saito, T. The quantity and duration of FcRγ signals determine mast cell degranulation and survival. Blood 103, 3093–3101 (2004).
Shinkai, Y. et al. Restoration of T cell development in RAG-2-deficient mice by functional TCR transgenes. Science 259, 822–825 (1993).
Anderson, S.J., Abraham, K.M., Nakayama, T., Singer, A. & Perlmutter, R.M. Inhibition of T-cell receptor β-chain gene rearrangement by overexpression of the non-receptor protein tyrosine kinase p56lck. EMBO J. 11, 4877–4886 (1992).
Leahy, D.J., Axel, R. & Hendrickson, W.A. Crystal structure of a soluble form of the human T cell coreceptor CD8 at 2.6 A resolution. Cell 68, 1145–1162 (1992).
Arcaro, A. et al. CD8β endows CD8 with efficient coreceptor function by coupling T cell receptor/CD3 to raft-associated CD8/p56lck complexes. J. Exp. Med. 194, 1485–1495 (2001).
Carleton, M. et al. Signals transduced by CD3ε, but not by surface pre-TCR complexes, are able to induce maturation of an early thymic lymphoma in vitro. J. Immunol. 163, 2576–2585 (1999).
Masuda, K. et al. Thymic anlage is colonized by progenitors restricted to T, NK, and dendritic cell lineages. J. Immunol. 174, 2525–2532 (2005).
Michie, A.M. & Zuniga-Pflucker, J.C. Regulation of thymocyte differentiation: pre-TCR signals and β-selection. Semin. Immunol. 14, 311–323 (2002).
Hayes, S.M., Shores, E.W. & Love, P.E. An architectural perspective on signaling by the pre-, αβ and γδ T cell receptors. Immunol. Rev. 191, 28–37 (2003).
Gibbons, D. et al. The biological activity of natural and mutant pTα alleles. J. Exp. Med. 194, 695–703 (2001).
Levelt, C.N., Mombaerts, P., Iglesias, A., Tonegawa, S. & Eichmann, K. Restoration of early thymocyte differentiation in T-cell receptor β-chain-deficient mutant mice by transmembrane signaling through CD3ε. Proc. Natl. Acad. Sci. USA 90, 11401–11405 (1993).
Gil, D., Schamel, W.W., Montoya, M., Sanchez-Madrid, F. & Alarcon, B. Recruitment of Nck by CD3ε reveals a ligand-induced conformational change essential for T cell receptor signaling and synapse formation. Cell 109, 901–912 (2002).
Howard, F.D. et al. The CD3ζ cytoplasmic domain mediates CD2-induced T cell activation. J. Exp. Med. 176, 139–145 (1992).
Carrasco, Y.R. et al. An endoplasmic reticulum retention function for the cytoplasmic tail of the human pre-T cell receptor (TCR) α chain: potential role in the regulation of cell surface pre-TCR expression levels. J. Exp. Med. 193, 1045–1058 (2001).
Carrasco, Y.R., Navarro, M.N. & Toribio, M.L. A role for the cytoplasmic tail of the pre-T cell receptor (TCR) α chain in promoting constitutive internalization and degradation of the pre-TCR. J. Biol. Chem. 278, 14507–14513 (2003).
Fehling, H.J. et al. Restoration of thymopoiesis in pTα−/− mice by anti-CD3ε antibody treatment or with transgenes encoding activated Lck or tailless pTα. Immunity 6, 703–714 (1997).
Trop, S., Steff, A.M., Denis, F., Wiest, D.L. & Hugo, P. The connecting peptide domain of pTα dictates weak association of the pre-T cell receptor with the TCRζ subunit. Eur. J. Immunol. 29, 2187–2196 (1999).
Trop, S., Rhodes, M., Wiest, D.L., Hugo, P. & Zuniga-Pflucker, J.C. Competitive displacement of pTα by TCR-α during TCR assembly prevents surface coexpression of pre-TCR and αβ TCR. J. Immunol. 165, 5566–5572 (2000).
Borowski, C. et al. On the brink of becoming a T cell. Curr. Opin. Immunol. 14, 200–206 (2002).
Muljo, S.A. & Schlissel, M.S. Pre-B and pre-T-cell receptors: conservation of strategies in regulating early lymphocyte development. Immunol. Rev. 175, 80–93 (2000).
Ohnishi, K. & Melchers, F. The nonimmunoglobulin portion of λ5 mediates cell-autonomous pre-B cell receptor signaling. Nat. Immunol. 4, 849–856 (2003).
Gauthier, L., Rossi, B., Roux, F., Termine, E. & Schiff, C. Galectin-1 is a stromal cell ligand of the pre-B cell receptor (BCR) implicated in synapse formation between pre-B and stromal cells and in pre-BCR triggering. Proc. Natl. Acad. Sci. USA 99, 13014–13019 (2002).
Bradl, H., Wittmann, J., Milius, D., Vettermann, C. & Jack, H.M. Interaction of murine precursor B cell receptor with stroma cells is controlled by the unique tail of λ5 and stroma cell-associated heparan sulfate. J. Immunol. 171, 2338–2348 (2003).
Yokosuka, T. et al. Predominant role of T cell receptor (TCR)-α chain in forming preimmune TCR repertoire revealed by clonal TCR reconstitution system. J. Exp. Med. 195, 991–1001 (2002).
Poirier, G., Lo, D., Reilly, C.R. & Kaye, J. Discrimination between thymic epithelial cells and peripheral antigen-presenting cells in the induction of immature T cell differentiation. Immunity 1, 385–391 (1994).
Chaffin, K.E. et al. Dissection of thymocyte signaling pathways by in vivo expression of pertussis toxin ADP-ribosyltransferase. EMBO J. 9, 3821–3829 (1990).
Kitamura, T. New experimental approaches in retrovirus-mediated expression screening. Int. J. Hematol. 67, 351–359 (1998).
Berman, H.M. et al. The Protein Data Bank. Nucleic Acids Res. 28, 235–242 (2000).
Needleman, S.B. & Wunsch, C.D. A general method applicable to the search for similarities in the amino acid sequence of two proteins. J. Mol. Biol. 48, 443–453 (1970).
Cornell, W.D. et al. A 2nd-generation force-field for the simulation of proteins, nucleic-acids, and small molecules. Abstr. Pap. Am. Chem. Soc. 204, 40-Comp (1992).
Pearlman, D.A. et al. Amber, a package of computer programs for Applying molecular mechanics, normal mode analysis, molecular dynamics and free-energy calculations to simulate the structural and energetic properties of molecules. Comput. Phys. Commun. 91, 1–41 (1995).
Tokunaga, M., Kitamura, K., Saito, K., Iwane, A.H. & Yanagida, T. Single molecule imaging of fluorophores and enzymatic reactions achieved by objective-type total internal reflection fluorescence microscopy. Biochem. Biophys. Res. Commun. 235, 47–53 (1997).
Funatsu, T., Harada, Y., Tokunaga, M., Saito, K. & Yanagida, T. Imaging of single fluorescent molecules and individual ATP turnovers by single myosin molecules in aqueous solution. Nature 374, 555–559 (1995).
Acknowledgements
We thank A. Yoshimura for cDNA; M. Unno and R. Shiina for technical help; H. Tomiyama for FRET analysis; N. Shinohara, S. Koyasu, A. Kosugi, H. Suzuki, K. Masuda, S. Sugawa, H. Fukuyama and O. Kanagawa for discussions; H. von Boehmer and S. Taki for critical reading of the manuscript; and H. Yamaguchi for secretarial assistance. Supported by a Grant-in-Aid for Priority Area Research (A) from the Ministry of Education, Culture, Sports, Science and Technology of Japan.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
pre-TCR has the tendency to form oligomers on the cell surface. (PDF 11 kb)
Supplementary Fig. 2
Generation of FRET in T cells co-expressing pTα–CFP and pTα–YFP. (PDF 45 kb)
Supplementary Fig. 3
Competitive BMT reconsitution assay of pTα-WT and pTα-R24A. (PDF 10 kb)
Supplementary Fig. 4
hCD8–CD3e chimera induced DN3 to DN4 cell transition in the absence of pTα. (PDF 29 kb)
Rights and permissions
About this article
Cite this article
Yamasaki, S., Ishikawa, E., Sakuma, M. et al. Mechanistic basis of pre–T cell receptor–mediated autonomous signaling critical for thymocyte development. Nat Immunol 7, 67–75 (2006). https://doi.org/10.1038/ni1290
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni1290
This article is cited by
-
Tuning charge density of chimeric antigen receptor optimizes tonic signaling and CAR-T cell fitness
Cell Research (2023)
-
Loss of Zfp335 triggers cGAS/STING-dependent apoptosis of post-β selection thymocytes
Nature Communications (2022)
-
The histone methyltransferase Setd2 is indispensable for V(D)J recombination
Nature Communications (2019)
-
Transcription factors and target genes of pre-TCR signaling
Cellular and Molecular Life Sciences (2015)
-
Pre-TCRα supports CD3-dependent reactivation and expansion of TCRα-deficient primary human T-cells
Molecular Therapy - Methods & Clinical Development (2014)