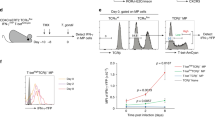
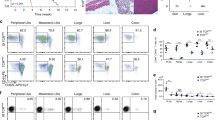
Abstract
T cells recognizing self proteins exist without causing autoimmunity in healthy individuals. These autoreactive T cells are kept in check by peripheral tolerance. Using a model for peripheral CD8+ T cell tolerance resulting from antigen presentation by resting dendritic cells in vivo, we show here that CD8+ T cell tolerance operates through T cell–intrinsic mechanisms such as deletion or functional inactivation. Peripheral CD8+ T cell tolerance depended on signaling via the costimulatory molecule PD-1, as an absence of PD-1 converted tolerance induction into priming. Blocking of the costimulatory molecule CTLA-4 resulted in impaired tolerance and enhanced the effect of the absence of PD-1, suggesting that PD-1 and CTLA-4 act synergistically. Thus PD-1 and CTLA-4 are crucial molecules for peripheral CD8+ T cell tolerance induced by resting dendritic cells.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Mondino, A., Khoruts, A. & Jenkins, M.K. The anatomy of T-cell activation and tolerance. Proc. Natl. Acad. Sci. USA 93, 2245–2252 (1996).
Derbinski, J., Schulte, A., Kyewski, B. & Klein, L. Promiscuous gene expression in medullary thymic epithelial cells mirrors the peripheral self. Nat. Immunol. 2, 1032–1039 (2001).
Kyewski, B., Derbinski, J., Gotter, J. & Klein, L. Promiscuous gene expression and central T-cell tolerance: more than meets the eye. Trends Immunol. 23, 364–371 (2002).
Bjorses, P., Aaltonen, J., Horelli-Kuitunen, N., Yaspo, M.L. & Peltonen, L. Gene defect behind APECED: a new clue to autoimmunity. Hum. Mol. Genet. 7, 1547–1553 (1998).
Anderson, M.S. et al. Projection of an immunological self shadow within the thymus by the aire protein. Science 298, 1395–1401 (2002).
Bouneaud, C., Kourilsky, P. & Bousso, P. Impact of negative selection on the T cell repertoire reactive to a self-peptide: a large fraction of T cell clones escapes clonal deletion. Immunity 13, 829–840 (2000).
Walker, L.S. & Abbas, A.K. The enemy within: keeping self-reactive T cells at bay in the periphery. Nat. Rev. Immunol. 2, 11–19 (2002).
Steinman, R.M. et al. Dendritic cell function in vivo during the steady state: a role in peripheral tolerance. Ann. NY Acad. Sci. 987, 15–25 (2003).
Steinman, R.M., Hawiger, D. & Nussenzweig, M.C. Tolerogenic dendritic cells. Annu. Rev. Immunol. 21, 685–711 (2003).
Sakaguchi, S. Naturally arising CD4+ regulatory T cells for immunologic self-tolerance and negative control of immune responses. Annu. Rev. Immunol. 22, 531–562 (2004).
Tivol, E.A. et al. Loss of CTLA-4 leads to massive lymphoproliferation and fatal multiorgan tissue destruction, revealing a critical negative regulatory role of CTLA-4. Immunity 3, 541–547 (1995).
Waterhouse, P. et al. Lymphoproliferative disorders with early lethality in mice deficient in Ctla-4. Science 270, 985–988 (1995).
Perez, V.L. et al. Induction of peripheral T cell tolerance in vivo requires CTLA-4 engagement. Immunity 6, 411–417 (1997).
Walunas, T.L. & Bluestone, J.A. CTLA-4 regulates tolerance induction and T cell differentiation in vivo. J. Immunol. 160, 3855–3860 (1998).
Lindsten, T. et al. Characterization of CTLA-4 structure and expression on human T cells. J. Immunol. 151, 3489–3499 (1993).
Brunner, M.C. et al. CTLA-4-mediated inhibition of early events of T cell proliferation. J. Immunol. 162, 5813–5820 (1999).
Ishida, Y., Agata, Y., Shibahara, K. & Honjo, T. Induced expression of PD-1, a novel member of the immunoglobulin gene superfamily, upon programmed cell death. EMBO J. 11, 3887–3895 (1992).
Agata, Y. et al. Expression of the PD-1 antigen on the surface of stimulated mouse T and B lymphocytes. Int. Immunol. 8, 765–772 (1996).
Nishimura, H., Nose, M., Hiai, H., Minato, N. & Honjo, T. Development of lupus-like autoimmune diseases by disruption of the PD-1 gene encoding an ITIM motif-carrying immunoreceptor. Immunity 11, 141–151 (1999).
Nishimura, H. et al. Autoimmune dilated cardiomyopathy in PD-1 receptor-deficient mice. Science 291, 319–322 (2001).
Freeman, G.J. et al. Engagement of the PD-1 immunoinhibitory receptor by a novel B7 family member leads to negative regulation of lymphocyte activation. J. Exp. Med. 192, 1027–1034 (2000).
Latchman, Y. et al. PD-L2 is a second ligand for PD-1 and inhibits T cell activation. Nat. Immunol. 2, 261–268 (2001).
Okazaki, T., Iwai, Y. & Honjo, T. New regulatory co-receptors: inducible co-stimulator and PD-1. Curr. Opin. Immunol. 14, 779–782 (2002).
Chen, L. Co-inhibitory molecules of the B7–CD28 family in the control of T-cell immunity. Nat. Rev. Immunol. 4, 336–347 (2004).
Probst, H.C., Lagnel, J., Kollias, G. & van den Broek, M. Inducible transgenic mice reveal resting dendritic cells as potent inducers of CD8+ T cell tolerance. Immunity 18, 713–720 (2003).
Moser, M. Dendritic cells in immunity and tolerance-do they display opposite functions? Immunity 19, 5–8 (2003).
Guermonprez, P., Valladeau, J., Zitvogel, L., Thery, C. & Amigorena, S. Antigen presentation and T cell stimulation by dendritic cells. Annu. Rev. Immunol. 20, 621–667 (2002).
Scheinecker, C., McHugh, R., Shevach, E.M. & Germain, R.N. Constitutive presentation of a natural tissue autoantigen exclusively by dendritic cells in the draining lymph node. J. Exp. Med. 196, 1079–1090 (2002).
Belz, G.T. et al. The CD8α+ dendritic cell is responsible for inducing peripheral self-tolerance to tissue-associated antigens. J. Exp. Med. 196, 1099–1104 (2002).
Lin, Y., Roberts, T.J., Sriram, V., Cho, S. & Brutkiewicz, R.R. Myeloid marker expression on antiviral CD8+ T cells following an acute virus infection. Eur. J. Immunol. 33, 2736–2743 (2003).
Probst, H.C. et al. Immunodominance of an antiviral cytotoxic T cell response is shaped by the kinetics of viral protein expression. J. Immunol. 171, 5415–5422 (2003).
Hou, W.S. & Van Parijs, L. A Bcl-2-dependent molecular timer regulates the lifespan and immunogenicity of dendritic cells. Nat. Immunol. 5, 583–589 (2004).
Ingulli, E., Mondino, A., Khoruts, A. & Jenkins, M.K. In vivo detection of dendritic cell antigen presentation to CD4+ T cells. J. Exp. Med. 185, 2133–2141 (1997).
Robinson, S.P., Langan-Fahey, S.M., Johnson, D.A. & Jordan, V.C. Metabolites, pharmacodynamics, and pharmacokinetics of tamoxifen in rats and mice compared to the breast cancer patient. Drug Metab. Dispos. 19, 36–43 (1991).
Krummel, M.F. & Allison, J.P. CD28 and CTLA-4 have opposing effects on the response of T cells to stimulation. J. Exp. Med. 182, 459–465 (1995).
Dhodapkar, M.V. & Steinman, R.M. Antigen-bearing immature dendritic cells induce peptide-specific CD8+ regulatory T cells in vivo in humans. Blood 100, 174–177 (2002).
Jonuleit, H., Schmitt, E., Schuler, G., Knop, J. & Enk, A.H. Induction of interleukin 10-producing, nonproliferating CD4+ T cells with regulatory properties by repetitive stimulation with allogeneic immature human dendritic cells. J. Exp. Med. 192, 1213–1222 (2000).
Mahnke, K., Qian, Y., Knop, J. & Enk, A.H. Induction of CD4+/CD25+ regulatory T cells by targeting of antigens to immature dendritic cells. Blood 101, 4862–4869 (2003).
Groux, H. et al. A transgenic model to analyze the immunoregulatory role of IL-10 secreted by antigen-presenting cells. J. Immunol. 162, 1723–1729 (1999).
Bonifaz, L. et al. Efficient targeting of protein antigen to the dendritic cell receptor DEC-205 in the steady state leads to antigen presentation on major histocompatibility complex class I products and peripheral CD8+ T cell tolerance. J. Exp. Med. 196, 1627–1638 (2002).
Hawiger, D. et al. Dendritic cells induce peripheral T cell unresponsiveness under steady state conditions in vivo. J. Exp. Med. 194, 769–779 (2001).
Misra, N., Bayry, J., Lacroix-Desmazes, S., Kazatchkine, M.D. & Kaveri, S.V. Cutting edge: human CD4+CD25+ T cells restrain the maturation and antigen-presenting function of dendritic cells. J. Immunol. 172, 4676–4680 (2004).
Albert, M.L. Death-defying immunity: do apoptotic cells influence antigen processing and presentation? Nat. Rev. Immunol. 4, 223–231 (2004).
Hernandez, J., Aung, S., Redmond, W.L. & Sherman, L.A. Phenotypic and functional analysis of CD8+ T cells undergoing peripheral deletion in response to cross-presentation of self-antigen. J. Exp. Med. 194, 707–717 (2001).
Hugues, S. et al. Tolerance to islet antigens and prevention from diabetes induced by limited apoptosis of pancreatic beta cells. Immunity 16, 169–181 (2002).
Kurts, C., Kosaka, H., Carbone, F.R., Miller, J.F. & Heath, W.R. Class I-restricted cross-presentation of exogenous self-antigens leads to deletion of autoreactive CD8+ T cells. J. Exp. Med. 186, 239–245 (1997).
Morgan, D.J., Kreuwel, H.T. & Sherman, L.A. Antigen concentration and precursor frequency determine the rate of CD8+ T cell tolerance to peripherally expressed antigens. J. Immunol. 163, 723–727 (1999).
Lo, D. et al. Peripheral tolerance to an islet cell-specific hemagglutinin transgene affects both CD4+ and CD8+ T cells. Eur. J. Immunol. 22, 1013–1022 (1992).
McCoy, K.D., Hermans, I.F., Fraser, J.H., Le Gros, G. & Ronchese, F. Cytotoxic T lymphocyte-associated antigen 4 (CTLA-4) can regulate dendritic cell-induced activation and cytotoxicity of CD8+ T cells independently of CD4+ T cell help. J. Exp. Med. 189, 1157–1162 (1999).
Nishimura, H., Honjo, T. & Minato, N. Facilitation of beta selection and modification of positive selection in the thymus of PD-1-deficient mice. J. Exp. Med. 191, 891–898 (2000).
Blank, C. et al. Absence of programmed death receptor 1 alters thymic development and enhances generation of CD4/CD8 double-negative TCR-transgenic T cells. J. Immunol. 171, 4574–4581 (2003).
Walunas, T.L. et al. CTLA-4 can function as a negative regulator of T cell activation. Immunity 1, 405–413 (1994).
Lehmann-Grube, F. Lymphocytic Choriomeningitis Virus. Virol. Monogr. 10, 1–173 (1971).
Gallimore, A. et al. Induction and exhaustion of lymphocytic choriomeningitis virus-specific cytotoxic T lymphocytes visualized using soluble tetrameric major histocompatibility complex class I-peptide complexes. J. Exp. Med. 187, 1383–1393 (1998).
Probst, H.C., Dumrese, T. & van den Broek, M.F. Cutting edge: competition for APC by CTLs of different specificities is not functionally important during induction of antiviral responses. J. Immunol. 168, 5387–5391 (2002).
Battegay, M. et al. Quantification of lymphocytic choriomeningitis virus with an immunological focus assay in 24- or 96-well plates. J. Virol. Methods 33, 191–198 (1991).
Acknowledgements
We thank K. Tschannen for technical assistance; M. Delic, J. Fehr and W. Kehrli for animal husbandry; A. Macpherson for reviewing this manuscript and for discussions; and R. Zinkernagel and H. Hengartner for discussions and support. Supported by the Swiss National Science Foundation, the Max Cloëtta Foundation Zurich, the European Community (QLG1-CT-1999-2002) and the Swiss Bundesamt für Bildung und Wissenschaft.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Probst, H., McCoy, K., Okazaki, T. et al. Resting dendritic cells induce peripheral CD8+ T cell tolerance through PD-1 and CTLA-4. Nat Immunol 6, 280–286 (2005). https://doi.org/10.1038/ni1165
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni1165
This article is cited by
-
Establishing a prognostic model based on immune-related genes and identification of BIRC5 as a potential biomarker for lung adenocarcinoma patients
BMC Cancer (2023)
-
TGF-β in correlation with tumor progression, immunosuppression and targeted therapy in colorectal cancer
Medical Oncology (2023)
-
Low expression of PRDM5 predicts poor prognosis of esophageal squamous cell carcinoma
BMC Cancer (2022)
-
PD-L1 signaling selectively regulates T cell lymphatic transendothelial migration
Nature Communications (2022)
-
CD27 expression on Treg cells limits immune responses against tumors
Journal of Molecular Medicine (2022)