Abstract
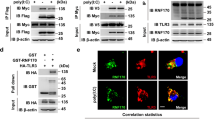
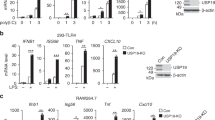
Activation of Toll-like receptors (TLRs) results in a proinflammatory response needed to combat infection. Thus, limiting TLR signaling is essential for preventing a protective response from causing injury to the host. Here we describe how a RING finger protein, Triad3A, acts as an E3 ubiquitin-protein ligase and enhances ubiquitination and proteolytic degradation of some TLRs. Triad3A overexpression promoted substantial degradation of TLR4 and TLR9 with a concomitant decrease in signaling, but did not affect TLR2 expression or signaling. Conversely, a reduction in endogenous Triad3A by small interfering RNA increased TLR expression and enhanced TLR activation. Thus, ubiquitination by Triad3A represents one pathway by which the intensity and duration of TLR signaling is controlled.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Chuang, T.H. & Ulevitch, R.J. Identification of hTLR10: a novel human Toll-like receptor preferentially expressed in immune cells. Biochim. Biophys. Acta 1518, 157–161 (2001).
Aderem, A. & Ulevitch, R.J. Toll-like receptors in the induction of the innate immune response. Nature 406, 782–787 (2000).
Takeda, K., Kaisho, T. & Akira, S. Toll-like receptors. Annu. Rev. Immunol. 21, 335–376 (2003).
Sabroe, I., Parker, L.C., Wilson, A.G., Whyte, M.K. & Dower, S.K. Toll-like receptors: their role in allergy and non-allergic inflammatory disease. Clin. Exp. Allergy 32, 984–989 (2002).
Zuany-Amorim, C., Hastewell, J. & Walker, C. Toll-like receptors as potential therapeutic targets for multiple diseases. Nat. Rev. Drug Discov. 1, 797–807 (2002).
Bochud, P.Y. & Calandra, T. Pathogenesis of sepsis: new concepts and implications for future treatment. BMJ 326, 262–266 (2003).
Sweet, M.J. et al. A novel pathway regulating lipopolysaccharide-induced shock by ST2/T1 via inhibition of Toll-like receptor 4 expression. J. Immunol. 166, 6633–6639 (2001).
Wald, D. et al. SIGIRR, a negative regulator of Toll-like receptor–interleukin 1 receptor signaling. Nat. Immunol. 4, 920–927 (2003).
Kobayashi, K. et al. IRAK-M is a negative regulator of Toll-like receptor signaling. Cell 110, 191–202 (2002).
Nakagawa, R. et al. SOCS-1 participates in negative regulation of LPS responses. Immunity. 17, 677–687 (2002).
Pickart, C.M. Mechanisms underlying ubiquitination. Annu. Rev. Biochem. 70, 503–533 (2001).
Glickman, M.H. & Ciechanover, A. The ubiquitin-proteasome proteolytic pathway: destruction for the sake of construction. Physiol Rev. 82, 373–428 (2002).
Strous, G.J. & Gent, J. Dimerization, ubiquitylation and endocytosis go together in growth hormone receptor function. FEBS Lett. 529, 102–109 (2002).
Dikic, I. & Giordano, S. Negative receptor signalling. Curr. Opin. Cell Biol. 15, 128–135 (2003).
Daun, J.M. & Fenton, M.J. Interleukin-1/Toll receptor family members: receptor structure and signal transduction pathways. J. Interferon Cytokine Res. 20, 843–855 (2000).
O'Neill, L.A., Fitzgerald, K.A. & Bowie, A.G. The Toll-IL-1 receptor adaptor family grows to five members. Trends Immunol. 24, 286–290 (2003).
Janssens, S. & Beyaert, R. Functional diversity and regulation of different interleukin-1 receptor-associated kinase (IRAK) family members. Mol. Cell 11, 293–302 (2003).
Barton, G.M. & Medzhitov, R. Linking Toll-like receptors to IFN-α/β expression. Nat. Immunol. 4, 432–433 (2003).
van der Reijden, B.A., Erpelinck-Verschueren, C.A., Lowenberg, B. & Jansen, J.H. TRIADs: a new class of proteins with a novel cysteine-rich signature. Protein Sci. 8, 1557–1561 (1999).
Chen, D., Li, X., Zhai, Z. & Shu, H.B. A novel zinc finger protein interacts with receptor-interacting protein (RIP) and inhibits tumor necrosis factor (TNF)- and IL1-induced NF-κB activation. J. Biol. Chem. 277, 15985–15991 (2002).
Shimura, H. et al. Ubiquitination of a new form of α-synuclein by parkin from human brain: implications for Parkinson's disease. Science 293, 263–269 (2001).
Chung, K.K. et al. Parkin ubiquitinates the α-synuclein-interacting protein, synphilin-1: implications for Lewy-body formation in Parkinson disease. Nat. Med. 7, 1144–1150 (2001).
Zhang, Y. et al. Parkin functions as an E2-dependent ubiquitin-protein ligase and promotes the degradation of the synaptic vesicle-associated protein, CDCrel-1. Proc. Natl. Acad. Sci. USA 97, 13354–13359 (2000).
Niwa, J. et al. Dorfin ubiquitylates mutant SOD1 and prevents mutant SOD1-mediated neurotoxicity. J. Biol. Chem. 277, 36793–36798 (2002).
Moynihan, T.P. et al. The ubiquitin-conjugating enzymes UbcH7 and UbcH8 interact with RING finger/IBR motif-containing domains of HHARI and H7-AP1. J. Biol. Chem. 274, 30963–30968 (1999).
Martinez-Noel, G., Niedenthal, R., Tamura, T. & Harbers, K. A family of structurally related RING finger proteins interacts specifically with the ubiquitin-conjugating enzyme UbcM4. FEBS Lett. 454, 257–261 (1999).
Lee, D.H. & Goldberg, A.L. Proteasome inhibitors: valuable new tools for cell biologists. Trends Cell Biol. 8, 397–403 (1998).
Gropper, R. et al. The ubiquitin-activating enzyme, E1, is required for stress-induced lysosomal degradation of cellular proteins. J. Biol. Chem. 266, 3602–3610 (1991).
Barrett, A.J. et al. L-trans-Epoxysuccinyl-leucylamido(4-guanidino)butane (E-64) and its analogues as inhibitors of cysteine proteinases including cathepsins B, H and L. Biochem. J. 201, 189–198 (1982).
Shimazu, R. et al. MD-2, a molecule that confers lipopolysaccharide responsiveness on Toll-like receptor 4. J. Exp. Med. 189, 1777–1782 (1999).
da Silva, C.J., Soldau, K., Christen, U., Tobias, P.S. & Ulevitch, R.J. Lipopolysaccharide is in close proximity to each of the proteins in its membrane receptor complex. transfer from CD14 to TLR4 and MD-2. J. Biol. Chem. 276, 21129–21135 (2001).
Pugin, J., Ulevitch, R.J. & Tobias, P.S. A critical role for monocytes and CD14 in endotoxin-induced endothelial cell activation. J. Exp. Med. 178, 2193–2200 (1993).
Sellati, T.J., Abrescia, L.D., Radolf, J.D. & Furie, M.B. Outer surface lipoproteins of Borrelia burgdorferi activate vascular endothelium in vitro. Infect. Immun. 64, 3180–3187 (1996).
Macfarlane, D.E. & Manzel, L. Antagonism of immunostimulatory CpG-oligodeoxynucleotides by quinacrine, chloroquine, and structurally related compounds. J. Immunol. 160, 1122–1131 (1998).
Hoebe, K. et al. Upregulation of costimulatory molecules induced by lipopolysaccharide and double-stranded RNA occurs by Trif-dependent and Trif-independent pathways. Nat. Immunol. 4, 1223–1229 (2003).
Kitada, T. et al. Mutations in the parkin gene cause autosomal recessive juvenile parkinsonism. Nature 392, 605–608 (1998).
Aguilera, M., Oliveros, M., Martinez-Padron, M., Barbas, J.A. & Ferrus, A. Ariadne-1: a vital Drosophila gene is required in development and defines a new conserved family of ring-finger proteins. Genetics 155, 1231–1244 (2000).
Sakata, E. et al. Parkin binds the Rpn10 subunit of 26S proteasomes through its ubiquitin-like domain. EMBO Rep. 4, 301–306 (2003).
Yamin, T.T. & Miller, D.K. The interleukin-1 receptor-associated kinase is degraded by proteasomes following its phosphorylation. J. Biol. Chem. 272, 21540–21547 (1997).
Li, L., Cousart, S., Hu, J. & McCall, C.E. Characterization of interleukin-1 receptor-associated kinase in normal and endotoxin-tolerant cells. J. Biol. Chem. 275, 23340–23345 (2000).
Bosisio, D. et al. Stimulation of toll-like receptor 4 expression in human mononuclear phagocytes by interferon-γ: a molecular basis for priming and synergism with bacterial lipopolysaccharide. Blood 99, 3427–3431 (2002).
Marsik, C. et al. Endotoxaemia modulates Toll-like receptors on leucocytes in humans. Br. J. Haematol. 121, 653–656 (2003).
Girnita, L., Girnita, A. & Larsson, O. Mdm2-dependent ubiquitination and degradation of the insulin-like growth factor 1 receptor. Proc. Natl. Acad. Sci. USA 100, 8247–8252 (2003).
Shenoy, S.K., McDonald, P.H., Kohout, T.A. & Lefkowitz, R.J. Regulation of receptor fate by ubiquitination of activated β2-adrenergic receptor and β-arrestin. Science 294, 1307–1313 (2001).
Cohen, B.D., Bariteau, J.T., Magenis, L.M. & Dias, J.A. Regulation of follitropin receptor cell surface residency by the ubiquitin-proteasome pathway. Endocrinology 144, 4393–4402 (2003).
Yamakami, M., Yoshimori, T. & Yokosawa, H. Tom 1, a VHS domain-containing protein, interacts with Tollip, ubiquitin, and clathrin. J. Biol. Chem. 278, 52865–52872 (2003).
Zhang, G. & Ghosh, S. Negative regulation of toll-like receptor-mediated signaling by Tollip. J. Biol. Chem. 277, 7059–7065 (2002).
Chuang, T.H., Lee, J., Kline, L., Mathison, J.C. & Ulevitch, R.J. Toll-like receptor 9 mediates CpG-DNA signaling. J. Leukoc. Biol. 71, 538–544 (2002).
Chuang, T.H. & Ulevitch, R.J. Cloning and characterization of a sub-family of human toll-like receptors: hTLR7, hTLR8 and hTLR9. Eur. Cytokine Netw. 11, 372–378 (2000).
Acknowledgements
We thank J.F. Lo and W.Y. Hu for providing reagents and for discussions, and J.C. Mathison for comments on the manuscript. Supported by National Institutes of Health (AI15136, GM28485 and AI54523 to R.J.U.).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Chuang, TH., Ulevitch, R. Triad3A, an E3 ubiquitin-protein ligase regulating Toll-like receptors. Nat Immunol 5, 495–502 (2004). https://doi.org/10.1038/ni1066
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni1066
This article is cited by
-
The RING finger protein family in health and disease
Signal Transduction and Targeted Therapy (2022)
-
RNF216 Alleviates Radiation-Induced Apoptosis and DNA Damage Through Regulating Ubiquitination-Mediated Degradation of p53 in Glioblastoma
Molecular Neurobiology (2022)
-
Molecular characterization and expression analysis of two RING-between-RING (RBR) ubiquitin ligase orthologues from the Asian seabass (Lates calcarifer)
Aquaculture International (2022)
-
Association of TLR 9 gene polymorphisms with remission in patients with rheumatoid arthritis receiving TNF-α inhibitors and development of machine learning models
Scientific Reports (2021)
-
Indole-3-carboxaldehyde regulates RSV-induced inflammatory response in RAW264.7 cells by moderate inhibition of the TLR7 signaling pathway
Journal of Natural Medicines (2021)