Abstract
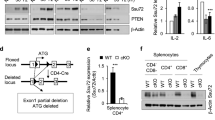
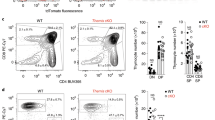
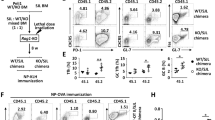
SH2D1A, which encodes signaling lymphocyte activation molecule (SLAM)–associated protein (SAP), is altered in patients with X-linked lymphoproliferative disease (XLP), a primary immunodeficiency. SAP-deficient mice infected with lymphocytic choriomeningitis virus had greatly increased numbers of CD8+ and CD4+ interferon-γ–producing spleen and liver cells compared to wild-type mice. The immune responses of SAP-deficient mice to infection with Leishmania major together with in vitro studies showed that activated SAP-deficient T cells had an impaired ability to differentiate into T helper 2 cells. The aberrant immune responses in SAP-deficient mice show that SAP controls several distinct key T cell signal transduction pathways, which explains in part the complexity of the XLP phenotypes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Sayos, J. et al. The X-linked lymphoproliferative-disease gene product SAP regulates signals induced through the co-receptor SLAM. Nature 395, 462–469 (1998).
Coffey, A. J. et al. Host response to EBV infection in X-linked lymphoproliferative disease results from mutations in an SH2-domain encoding gene. Nature Genet. 20, 129–135 (1998).
Nichols, K. E. et al. Inactivating mutations in an SH2 domain-encoding gene in X-linked lymphoproliferative syndrome. Proc. Natl Acad. Sci. USA 95, 13765–13770 (1998).
Wu, C. et al. Genomic organization and characterization of mouse SAP, the gene that is altered in X-linked lymphoproliferative disease. Immunogenetics 51, 805–815 (2000).
Parolini, S. et al. X-linked Lymphoproliferative Disease. 2B4 molecules displaying inhibitory rather than activating function are responsible for the inability of natural killer cells to kill epstein-barr virus-infected cells. J. Exp. Med. 192, 337–346 (2000).
Tangye, S. G. et al. Cutting edge: human 2B4, an activating NK cell receptor, recruits the protein tyrosine phosphatase SHP-2 and the adaptor signaling protein SAP. J. Immunol. 162, 6981–6985 (1999).
Benoit, L., Wang, X., Pabst, H.F., Dutz, J. & Tan, R. Defective natural killer cell activation in X-linked lymphoproliferative disease. J. Immunol. 165, 3549–3553 (2000).
Sayos, J. et al. Potential pathways for regulation of NK and T cell responses: differential X-linked lymphoproliferative syndrome gene product SAP interactions with SLAM and 2B4. Int. Immunol. 12, 1749–1757 (2000).
Sayos, J. et al. The cell surface receptors Ly-9 and CD84 recruit the XLP gene product SAP. Blood (in the press, 2001).
Purtilo, D. T., Grierson, H. L., Davis, J. R. & Okano, M. The X-linked lymphoproliferative disease: from autopsy toward cloning the gene 1975–1990. Pediatr. Pathol. 11, 685–710 (1991).
Seemayer, T. A.et al. X-linked lymphoproliferative disease: twenty-five years after the discovery. Pediatr. Res. 38, 471–478 (1995).
Maia, D. M. & Garwacki, C. P. X-linked lymphoproliferative disease: pathology and diagnosis. Pediatr. Dev. Pathol. 2, 72–77 (1999).
Morra, M. et al. X-linked lymphoproliferation disease: a progressive immunodeficiency. Annu. Rev. Immunol. 19, 657–682 (2001).
Zinkernagel, R. M. et al. T cell-mediated hepatitis in mice infected with lymphocytic choriomeningitis virus. Liver cell destruction by H-2 class I-restricted virus-specific cytotoxic T cells as a physiological correlate of the 51Cr-release assay? J. Exp. Med. 164, 1075–1092 (1986).
McIntyre, K. W. & Welsh, R. M. Accumulation of natural killer and cytotoxic T large granular lymphocytes in the liver during virus infection. J. Exp. Med. 164, 1667–1681 (1986).
Yoshimoto, T. & Paul, W. E. CD4pos, NK1.1pos T cells promptly produce interleukin 4 in response to in vivo challenge with anti-CD3. J. Exp. Med. 179, 1285–1295 (1994).
Yoshimoto, T., Bendelac, A., Watson, C., Hu-Li, J. & Paul, W. E. Role of NK1.1+ T cells in a TH2 response and in immunoglobulin E production. Science 270, 1845–1847 (1995).
Yoshimoto, T., Bendelac, A., Hu-Li, J. & Paul, W. E. Defective IgE production by SJL mice is linked to the absence of CD4+, NK1.1+ T cells that promptly produce interleukin 4. Proc. Natl Acad. Sci. USA 92, 11931–11934 (1995).
Himmelrich, H., Parra-Lopez, C., Tacchini-Cottier, F., Louis, J. A. & Launois, P. The IL-4 rapidly produced in BALB/c mice after infection with Leishmania major down-regulates IL-12 receptor β2-chain expression on CD4+ T cells resulting in a state of unresponsiveness to IL-12. J. Immunol. 161, 6156–6163 (1998).
Launois, P. et al. IL-4 rapidly produced by Vβ 4Vα 8 CD4+ T cells instructs Th2 development and susceptibility to Leishmania major in BALB/c mice. Immunity 6, 541–549 (1997).
Chatelain, R., Varkila, K. & Coffman, R. L. IL-4 induces a Th2 response in Leishmania major-infected mice. J. Immunol. 148, 1182–1187 (1992).
Satoskar, A. R. et al. NK cell-deficient mice develop a Th1-like response but fail to mount an efficient antigen-specific IgG2a antibody response. J. Immunol. 163, 5298–5302 (1999).
Cocks, B. G. et al. A novel receptor involved in T-cell activation. Nature 376, 260–263 (1995).
Tangye, S. G., Phillips, J. H., Lanier, L. L. & Nichols, K. E. Functional requirement for SAP in 2B4-mediated activation of human natural killer cells as revealed by the X-linked lymphoproliferative syndrome. J. Immunol. 165, 2932–2936 (2000).
Morra, M. et al. Missense mutations that affect non-phospho tyrosine interactions and stability of the SAP SH2-domain in X-linked proliferative disease patients. J. Exp. Med. (submitted, 2001).
Cousens, L. P. et al. Two roads diverged: interferon α/β- and interleukin 12-mediated pathways in promoting T cell interferon γ responses during viral infection. J. Exp. Med. 189, 1315–1328 (1999).
Murali-Krishna, K. et al. Counting antigen-specific CD8 T cells: a reevaluation of bystander activation during viral infection. Immunity 8, 177–187 (1998).
Pien, G. C. & Biron, C. A. Compartmental differences in NK cell responsiveness to IL-12 during lymphocytic choriomeningitis virus infection. J. Immunol. 164, 994–1001 (2000).
Satoskar, A. R. et al. Mice lacking NK cells develop an efficient TH1 response and control cutaneous Leishmania major infection. J. Immunol. 162, 6747–6754 (1999).
Acknowledgements
We thank L. Du and J. Burgess for their technical assistance in generating the SAP−/− mice; O. Leo, A. Bhan and S. Ju for their technical advice; J. Fingeroth, N. Houten and Y. Jong for a critical review of the manuscript and the NIAID Tetramer Facility, NIH AIDS Research and Reference Reagent Program and J. Altman for providing the LCMV tetramers. Supported by the Cancer Research Institute (C. W.), the Leukemia Society (D. H.) and grants from the NIH.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wu, C., Nguyen, K., Pien, G. et al. SAP controls T cell responses to virus and terminal differentiation of TH2 cells. Nat Immunol 2, 410–414 (2001). https://doi.org/10.1038/87713
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/87713
This article is cited by
-
Stepwise B-cell-dependent expansion of T helper clonotypes diversifies the T-cell response
Nature Communications (2016)
-
Identification of a new isoform of the murine Sh2d1a gene and its functional implications
Science China Life Sciences (2014)
-
SLAM family receptors and the SLAM-associated protein (SAP) modulate T cell functions
Seminars in Immunopathology (2010)
-
A module-based analytical strategy to identify novel disease-associated genes shows an inhibitory role for interleukin 7 Receptor in allergic inflammation
BMC Systems Biology (2009)
-
SLAM receptors and SAP influence lymphocyte interactions, development and function
Nature Reviews Immunology (2009)