Abstract
An imbalance in the lineages of immunosuppressive regulatory T cells (Treg cells) and the inflammatory TH17 subset of helper T cells leads to the development of autoimmune and/or inflammatory disease. Here we found that TAZ, a coactivator of TEAD transcription factors of Hippo signaling, was expressed under TH17 cell–inducing conditions and was required for TH17 differentiation and TH17 cell–mediated inflammatory diseases. TAZ was a critical co-activator of the TH17-defining transcription factor RORγt. In addition, TAZ attenuated Treg cell development by decreasing acetylation of the Treg cell master regulator Foxp3 mediated by the histone acetyltransferase Tip60, which targeted Foxp3 for proteasomal degradation. In contrast, under Treg cell–skewing conditions, TEAD1 expression and sequestration of TAZ from the transcription factors RORγt and Foxp3 promoted Treg cell differentiation. Furthermore, deficiency in TAZ or overexpression of TEAD1 induced Treg cell differentiation, whereas expression of a transgene encoding TAZ or activation of TAZ directed TH17 cell differentiation. Our results demonstrate a pivotal role for TAZ in regulating the differentiation of Treg cells and TH17 cells.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
Change history
20 July 2017
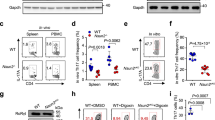
In the version of this article initially published, the description of Figure 1d,e in the first subsection of Results was incorrect; "...(Lck-Cre): Tazfl/flLck-Cre mice) immunized with KLH exhibited a larger TH17 population and fewer Treg cells than that of their Tazfl/fl littermates..." should read: "...(Lck-Cre)): Tazfl/flLck-Cre mice immunized with KLH exhibited a smaller TH17 population and more Treg cells than that of their Tazfl/fl littermates...". Also, the second sentence of the panel legend for Figure 1a incorrectly identified the numbers in the plots on the top row as "percent TH7 cells..."; this should read "percent TH17 cells...". These errors have been corrected in the PDF and HTML versions of this article.
14 February 2018
In the version of this article initially published, the institution name for affiliation 3 (Maryland Anderson Cancer Center) was incorrect. The correct institution is MD Anderson Cancer Center. The error has been corrected in the HTML and PDF versions of the article.
18 October 2017
Nat. Immunol. 18, 800–812 (2017); published online 15 May 2017; corrected after print 20 July 2017 In the version of this article initially published, the description of Figure 1d,e in the first subsection of Results was incorrect; “...(Lck-Cre): Tazfl/flLck-Cre mice) immunized with KLH exhibited a larger TH17 population and fewer Treg cells than that of their Tazfl/fl littermates.
References
Korn, T., Bettelli, E., Oukka, M. & Kuchroo, V.K. IL-17 and Th17 cells. Annu. Rev. Immunol. 27, 485–517 (2009).
Littman, D.R. & Rudensky, A.Y. Th17 and regulatory T cells in mediating and restraining inflammation. Cell 140, 845–858 (2010).
Bettelli, E. et al. Reciprocal developmental pathways for the generation of pathogenic effector TH17 and regulatory T cells. Nature 441, 235–238 (2006).
Zhou, L. et al. TGF-β-induced Foxp3 inhibits TH17 cell differentiation by antagonizing RORgammat function. Nature 453, 236–240 (2008).
Xu, L., Kitani, A., Fuss, I. & Strober, W. Cutting edge: regulatory T cells induce CD4+CD25−Foxp3− T cells or are self-induced to become Th17 cells in the absence of exogenous TGF-β. J. Immunol. 178, 6725–6729 (2007).
Yang, X.O. et al. Molecular antagonism and plasticity of regulatory and inflammatory T cell programs. Immunity 29, 44–56 (2008).
Zhou, X. et al. Instability of the transcription factor Foxp3 leads to the generation of pathogenic memory T cells in vivo. Nat. Immunol. 10, 1000–1007 (2009).
Zhou, L., Chong, M.M. & Littman, D.R. Plasticity of CD4+ T cell lineage differentiation. Immunity 30, 646–655 (2009).
Hong, J.H. et al. TAZ, a transcriptional modulator of mesenchymal stem cell differentiation. Science 309, 1074–1078 (2005).
Kanai, F. et al. TAZ: a novel transcriptional co-activator regulated by interactions with 14-3-3 and PDZ domain proteins. EMBO J. 19, 6778–6791 (2000).
Hong, W. & Guan, K.L. The YAP and TAZ transcription co-activators: key downstream effectors of the mammalian Hippo pathway. Semin. Cell Dev. Biol. 23, 785–793 (2012).
Zanconato, F., Cordenonsi, M. & Piccolo, S. YAP/TAZ at the roots of cancer. Cancer Cell 29, 783–803 (2016).
Varelas, X. et al. TAZ controls Smad nucleocytoplasmic shuttling and regulates human embryonic stem-cell self-renewal. Nat. Cell Biol. 10, 837–848 (2008).
Varelas, X. et al. The Crumbs complex couples cell density sensing to Hippo-dependent control of the TGF-β-SMAD pathway. Dev. Cell 19, 831–844 (2010).
Yu, F.X., Zhao, B. & Guan, K.L. Hippo Pathway in organ size control, tissue homeostasis, and cancer. Cell 163, 811–828 (2015).
Pan, D. The hippo signaling pathway in development and cancer. Dev. Cell 19, 491–505 (2010).
Harvey, K.F., Zhang, X. & Thomas, D.M. The Hippo pathway and human cancer. Nat. Rev. Cancer 13, 246–257 (2013).
Johnson, R. & Halder, G. The two faces of Hippo: targeting the Hippo pathway for regenerative medicine and cancer treatment. Nat. Rev. Drug Discov. 13, 63–79 (2014).
Halder, G., Dupont, S. & Piccolo, S. Transduction of mechanical and cytoskeletal cues by YAP and TAZ. Nat. Rev. Mol. Cell Biol. 13, 591–600 (2012).
Avruch, J. et al. Protein kinases of the Hippo pathway: regulation and substrates. Semin. Cell Dev. Biol. 23, 770–784 (2012).
Nehme, N.T . et al. MST1 mutations in autosomal recessive primary immunodeficiency characterized by defective naive T-cell survival. Blood 119, 3458–3468 (2012).
Abdollahpour, H. et al. The phenotype of human STK4 deficiency. Blood 119, 3450–3457 (2012).
Zhou, D. et al. The Nore1B/Mst1 complex restrains antigen receptor-induced proliferation of naïve T cells. Proc. Natl. Acad. Sci. USA 105, 20321–20326 (2008).
Katagiri, K., Imamura, M. & Kinashi, T. Spatiotemporal regulation of the kinase Mst1 by binding protein RAPL is critical for lymphocyte polarity and adhesion. Nat. Immunol. 7, 919–928 (2006).
Katagiri, K., Maeda, A., Shimonaka, M. & Kinashi, T. RAPL, a Rap1-binding molecule that mediates Rap1-induced adhesion through spatial regulation of LFA-1. Nat. Immunol. 4, 741–748 (2003).
Katagiri, K. et al. Crucial functions of the Rap1 effector molecule RAPL in lymphocyte and dendritic cell trafficking. Nat. Immunol. 5, 1045–1051 (2004).
Katagiri, K. et al. Deficiency of Rap1-binding protein RAPL causes lymphoproliferative disorders through mislocalization of p27kip1. Immunity 34, 24–38 (2011).
Li, J. et al. Mammalian sterile 20-like kinase 1 (Mst1) enhances the stability of Forkhead box P3 (Foxp3) and the function of regulatory T Cells by modulating Foxp3 acetylation. J. Biol. Chem. 290, 30762–30770 (2015).
Dong, Y. et al. A cell-intrinsic role for Mst1 in regulating thymocyte egress. J. Immunol. 183, 3865–3872 (2009).
Du, X. et al. Mst1/Mst2 regulate development and function of regulatory T cells through modulation of Foxo1/Foxo3 stability in autoimmune disease. J. Immunol. 192, 1525–1535 (2014).
Katagiri, K. et al. Mst1 controls lymphocyte trafficking and interstitial motility within lymph nodes. EMBO J. 28, 1319–1331 (2009).
Ueda, Y. et al. Mst1 regulates integrin-dependent thymocyte trafficking and antigen recognition in the thymus. Nat. Commun. 3, 1098 (2012).
Nishikimi, A. et al. Rab13 acts downstream of the kinase Mst1 to deliver the integrin LFA-1 to the cell surface for lymphocyte trafficking. Sci. Signal. 7, ra72 (2014).
Tang, F. et al. The kinases NDR1/2 act downstream of the Hippo homolog MST1 to mediate both egress of thymocytes from the thymus and lymphocyte motility. Sci. Signal. 8, ra100 (2015).
Moroishi, T. et al. The Hippo pathway knases LATS1/2 suppress cancer immunity. Cell 167, 1525–1539 (2016).
Liu, B. et al. Toll receptor-mediated Hippo signaling controls innate immunity in Drosophila. Cell 164, 406–419 (2016).
Geng, J. et al. Kinases Mst1 and Mst2 positively regulate phagocytic induction of reactive oxygen species and bactericidal activity. Nat. Immunol. 16, 1142–1152 (2015).
Mou, F. et al. The Mst1 and Mst2 kinases control activation of rho family GTPases and thymic egress of mature thymocytes. J. Exp. Med. 209, 741–759 (2012).
Jiao, S. et al. The kinase MST4 limits inflammatory responses through direct phosphorylation of the adaptor TRAF6. Nat. Immunol. 16, 246–257 (2015).
Raab, M. et al. T cell receptor “inside-out” pathway via signaling module SKAP1-RapL regulates T cell motility and interactions in lymph nodes. Immunity 32, 541–556 (2010).
Li, W. et al. STK4 regulates TLR pathways and protects against chronic inflammation-related hepatocellular carcinoma. J. Clin. Invest. 125, 4239–4254 (2015).
Guo, X. et al. Single tumor-initiating cells evade immune clearance by recruiting type II macrophages. Genes Dev. 31, 247–259 (2017).
van Loosdregt, J. & Coffer, P.J. Post-translational modification networks regulating FOXP3 function. Trends Immunol. 35, 368–378 (2014).
Lei, Q.Y. et al. TAZ promotes cell proliferation and epithelial-mesenchymal transition and is inhibited by the hippo pathway. Mol. Cell. Biol. 28, 2426–2436 (2008).
Zhang, H. et al. TEAD transcription factors mediate the function of TAZ in cell growth and epithelial-mesenchymal transition. J. Biol. Chem. 284, 13355–13362 (2009).
Azzolin, L. et al. YAP/TAZ incorporation in the β-catenin destruction complex orchestrates the Wnt response. Cell 158, 157–170 (2014).
Zhang, F., Meng, G. & Strober, W. Interactions among the transcription factors Runx1, RORγt and Foxp3 regulate the differentiation of interleukin 17-producing T cells. Nat. Immunol. 9, 1297–1306 (2008).
Lazarevic, V. et al. T-bet represses T(H)17 differentiation by preventing Runx1-mediated activation of the gene encoding RORγt. Nat. Immunol. 12, 96–104 (2011).
Lang, R. et al. SOCS3 regulates the plasticity of gp130 signaling. Nat. Immunol. 4, 546–550 (2003).
Croker, B.A. et al. SOCS3 negatively regulates IL-6 signaling in vivo. Nat. Immunol. 4, 540–545 (2003).
Bendall, S.C. et al. Single-cell mass cytometry of differential immune and drug responses across a human hematopoietic continuum. Science 332, 687–696 (2011).
Amir, A.D. et al. viSNE enables visualization of high dimensional single-cell data and reveals phenotypic heterogeneity of leukemia. Nat. Biotechnol. 31, 545–552 (2013).
Wu, H. et al. The Ets transcription factor GABP is a component of the hippo pathway essential for growth and antioxidant defense. Cell Rep. 3, 1663–1677 (2013).
Acknowledgements
We thank J. Avruch for comments on the manuscript. Supported by the National Basic Research Program (973) of China (2015CB910502 to L.C.), the National Natural Science Foundation of China (81422018 to L.C.; 31625010 and U1505224 to D.Z.; U1405225 and 81372617 to L.C.; J1310027 to D.Z.; 81472229 to L.H.; and 31600698 to J. Geng), the 111 Projects (B12001 and B06016), China's 1000 Young Talents Program (D.Z., and L.C.), the Fundamental Research Funds for the Central Universities of China-Xiamen University (20720160071 to D.Z. and 20720160054 to L.H.) and Major disease research projects of Xiamen (3502Z20149029 to L.C.). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
J. Geng, S.Y., H.Z., X.S., P.W., X.X., L.H., J. Gao, Y.S. and J.P. performed experimental biological research; X.L. provided human blood samples; C.X. performed mass-spectrometry analysis; R.L.J. provided mutant mice; D.Z. and L.C. conceived of the project, with input from R.L.J., N.X., L.L. and J.H., co-wrote the paper; and all authors edited the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
Supplementary Figure 1 Characterization of mice with conditional knockout of MST1/2 or TAZ in T cells.
(a) Flow cytometric analysis (top panel) and quantification (lower panel) of thymocytes from 8-week-old Mst1f/fMst2f/f mice or Mst1f/fMst2f/f Ox40-Cre mice, stained with anti-CD4 and anti-CD8 antibodies. (b-d) Flow cytometric analysis (b) and quantification of CD3+CD4+ or CD3+CD8+ T cell (c), the naïve T cell (CD62LhighCD44low) and the effector T cell (CD62LlowCD44high) (d) populations in the spleen or lymph node from Mst1f/fMst2f/f mice and Mst1f/fMst2f/f Ox40-Cre mice, with the indicated antibodies. (e and f) Photograph of 10-month-old Mst1-/- KO mice with Sjögren’s syndrome (e, left panel). Representative H&E-stained eye sections with associated lymphocytic cell infiltration of the lacrimal gland (e, right panel). Relative mRNA levels of Taz, Ifng, Il17a, Il17f and Il23r genes in the lacrimal gland by Real-time quantitative PCR (f). Scale bars, 500μm. The data are representative of three independent experiments with similar results (a-f). The data represent mean ± s.d. (n=3). ns, not significant (P > 0.05); *P < 0.05, **P < 0.01 and ***P < 0.001 compared with control, Student’s t-test.
Supplementary Figure 2 Characterization of T cells from Tazf/fLck-Cre mice.
(a) Flow cytometric analysis (top panel) and quantification (lower panel) of thymocytes from 8-week-old Tazf/f mice or Tazf/f Lck-Cre mice, stained with anti-CD4 and anti-CD8 antibodies. (b-d) Flow cytometric analysis (b) and quantification of B220+ B cells, CD3+CD4+ or CD3+CD8+ T cell (c), the naïve T cell (CD62LhighCD44low) and the effector T cell (CD62LlowCD44high) (d) populations in the spleen or lymph node of Tazf/f mice and Tazf/f Lck-Cre mice, with the indicated antibodies. (e-f) Flow cytometry analysis of the activation markers CD69, CD25, CD62L, and CD44 on cell surfaces (e) and enzyme-linked immunosorbent assay of IL-2 or IFNγ production (f) in culture medium of CD4+ T cells isolated from 8-week-old Tazf/f mice or Tazf/f Lck-Cre mice, stimulated with anti-CD3 or anti-CD3 plus anti-CD28 antibodies for 24 hours. Data were assessed with Student’s t-test and are represented as mean ± s.d. ns, not significant. (g) CFSE proliferation assay of CD4+ T cells isolated from 8-week-old Tazf/f mice or Tazf/f Lck-Cre mice, stimulated with anti-CD3 plus anti-CD28 antibodies for 48 hours. (h) CFSE proliferation assay of Th17 or Treg cells differentiated from Tazf/f mice or Tazf/f Lck-Cre naïve T cells, stimulated with anti-CD3 plus anti-CD28 antibodies for indicated time. (i and j) Quantification of intracellular or ELISA assay of IL-17F expression (i) or Real-time quantitative PCR analysis of the expression levels of indicated genes in CD4+ naïve T cells from Tazf/f mice or Tazf/f Lck-Cre mice differentiated under TH17. (k and l) H&E (k) and anti-CD3 (l) staining for lymphocyte infiltration in the spinal cord of Tazf/f mice and Tazf/f Lck-Cre mice subjected to EAE by MOG immunization. Scale bars, 100μM. (m and n) Proliferation rate (m) and IL-2 production (n) of responder CD4+ naïve T cells cocultured with CD4+CD25+ Treg cells from Tazf/f mice or Tazf/f Lck-Cre mice and wild-type irradiated spleen cells and stimulated with anti-CD3 antibodies. The data are representative of three independent experiments with similar results. The data represent the mean ± s.d. (n=3). ns, not significant (P > 0.05); *P < 0.05, **P < 0.01 and ***P < 0.001 compared with control, Student’s t-test.
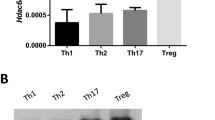
Supplementary Figure 3 Smad3 and STAT3 synergistically upregulate Taz expression.
Identification of Smad3 and STAT3 by mass spectrometry in streptavidin-precipitation assays using biotinylated Taz gene 1-kb promoter (Taz p1000) DNA fragments in lymphoid tissue lysates of WT mouse. The control DNA was amplified from PGL3-Basic vectors using the same primer set. The DNA gel in the bottom shows the input of biotinylated DNA fragments. M, molecular size markers. The data are representative of three independent experiments with similar results.
Supplementary Figure 4 TAZ directly binds to RORγt and enhances its transcriptional activity.
(a) Identification and the list (the right panel) of TEAD3, TEAD4, RORγt, Foxp3 and Tip60 by mass spectrometry in a Flag-tagged TAZ-precipitation assay in lymphoid tissue lysates of WT or Taz Tg mice. *, migration of TAZ; IP, immunoprecipitation; IB, immunoblot analysis; M, molecular size markers. (b, c) Immunoassays of 293T cells expressing various combinations (above lanes) of GFP-tagged RORγt and full-length (FL) or the truncated fragment of Flag-TAZ as indicated; immunoprecipitation with anti-Flag and analysis by immunoblot with the indicated antibodies; below, immunoblot analysis of total cell lysates (TCL) without immunoprecipitation. (d) Immunoassays (as in b, c) of 293T cells expressing various combinations (above lanes) of GFP-tagged TAZ and full-length (FL) or the truncated fragment of Flag-RORγt as indicated. (e) Super-resolution immunofluorescence imaging (SIM) of HeLa cells transfected with nuclear localization sequence (NLS)/Flag-tagged TAZ (red), full-length, DBD, Hinge or LBD fragment of RORγt (green). Scale bars, 20 μm. The data are representative of three independent experiments with similar results (a-e), or with ~50 cells (e)
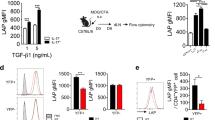
Supplementary Figure 5 TAZ blocks acetylation of Foxp3 and promotes its degradation.
(a) Real-time quantitative PCR (RT-qPCR) analysis of the expression levels of Foxp3 in CD4+ naïve T cells cultured with anti-CD3/CD28 antibodies in the presence of TGF-β (2.5 ng/ml) or TGF-β plus IL-6 (30 ng/ml) for 1 and 2 days. Data were assessed with Student’s t-test and are represented as mean ± s.d. ns, not significant. (b) Immunoassays of 293T cells expressing various combinations (above lanes) of HA-tagged Tip60, and full-length (FL) or the truncated fragment of Flag-TAZ as indicated; immunoprecipitation with anti-Flag and analysis by immunoblot with the indicated antibodies; below, immunoblot analysis of total cell lysates (TCL) without immunoprecipitation. (c) Immunoassays (as in b) of 293T cells expressing various combinations (above lanes) of GFP-tagged TAZ, full-length (FL) or the truncated fragment of Flag-Tip60 as indicated. (d) Immunoassays (as in b) of 293T cells expressing various combinations (above lanes) of GFP-tagged Foxp3, and full-length (FL) or the truncated fragment of Flag-Tip60 as indicated. (e) Immunoassays (as in b) of 293T cells expressing various combinations (above lanes) of Myc-tagged Tip60, Flag-Foxp3 and increasing doses of HA-tagged TAZ. (f) SIM of HeLa cells transfected with NLS/Flag-tagged TAZ (purple), HA-tagged Tip60 (red) or Flag-Foxp3 (green); Scale bars, 20 μm. The data are representative of three independent experiments with similar results (a-f), or with ~50 cells (f).
Supplementary Figure 6 TAZ activates RORγt by blocking the inhibitory effect of Foxp3 on RORγt.
(a) Immunoassays of 293T cells expressing various combinations (above lanes) of GFP-tagged Foxp3, and full-length (FL) or the truncated fragment of Flag-TAZ as indicated. immunoprecipitation with anti-Flag and analysis by immunoblot with the indicated antibodies; below, immunoblot analysis of total cell lysates (TCL) without immunoprecipitation. (b) Immunoblot analysis (as in a) of Foxp3 and various forms of TAZ in total lysates (bottom) and anti-Flag immunoprecipitates (top) from 293T cells expressing GFP-tagged Foxp3 and Flag-tagged TB-WW or the TB fragment of TAZ. (c) Immunoassays (as in a) of 293T cells expressing various combinations (above lanes) of GFP-tagged TAZ and full-length (FL) or the truncated fragment of Flag-Foxp3 as indicated. (d) Immunoassays (as in a) of 293T cells expressing various combinations (above lanes) of GFP-tagged RORγt, mCherry-tagged Foxp3WT or Foxp3LL/AA and Flag-tagged TAZ. (e) SIM of HeLa cells transfected with NLS/Flag-tagged TAZ (purple), mCherry-tagged Foxp3 (red) and GFP- RORγt (green); Scale bars, 20 μm. (f) A proposed working model showing dimerized/polymerized TAZ required for the assembly of the Foxp3-TAZ-RORγt complex. The data are representative of three independent experiments with similar results (a-e), or with ~50 cells (e).
Supplementary Figure 7 TEAD sequesters TAZ from the TH17 master regulator RORγt to promote the development of Treg cells.
(a) Immunoassays of 293T cells expressing various combinations (above lanes) of Flag-tagged RORγt, Foxp3 or TEAD1 and HA-tagged TAZ; immunoprecipitation with anti-Flag and analysis by immunoblot with the indicated antibodies; below, immunoblot analysis of total cell lysates (TCL) without immunoprecipitation. (b) Immunoassays (as in a) of 293T cells expressing various combinations (above lanes) of Flag-tagged TAZWT or TAZS51A, GFP-tagged TEAD1 or increasing doses of GFP-tagged RORγt. (c) SIM of HeLa cells cotransfected with NLS/Flag-tagged TAZWT or TAZS51A (purple), HA-tagged TEAD1 (red) or GFP-tagged Foxp3 (green); outlined areas are enlarged 4× in the corner insets. Scale bars, 20 μm. (d) Immunoassays (as in a) of 293T cells expressing various combinations (above lanes) of Flag-tagged TAZ, HA-tagged Tip60 or increasing doses of HA-tagged TEAD1. (e) Immunoassay (as in a) of 293T cells expressing various combinations (above lanes) of HA-tagged Tip60, HA-tagged p300, Flag-tagged Foxp3 and/or GFP-tagged TAZ or HA-tagged TEAD1, treated with DMSO or the histone deacetylase inhibitors Trichostatin A and Nicotinamide (TSA&NAM). immunoprecipitation with anti-Flag and analysis by immunoblot with anti-K-Ace (α-K-Ace), anti-Flag (α-Flag), anti-HA (α-HA) or anti-GFP (α-GFP); below, immunoblot analysis of total cell lysates (TCL) without immunoprecipitation.
(f) Immunoblot analysis of TEAD1 in CD4+ naive T cells infected with a retrovirus expressing shRNA for knocking down TEAD1 or control shRNA, differentiated under Treg -polarizing conditions. The data are representative of three independent experiments with similar results (a-f), or with ~50 cells (c).
Supplementary Figure 8 A proposed working model showing TAZ-mediated regulation of TH17 or Treg cell differentiation.
TAZ potentiates TH17 differentiation through direct transcriptional activation of RORγt and promoting Foxp3 degradation by reducing Tip60-mediated acetylation of Foxp3, while under Treg-skewing conditions, upregulated TEAD1 sequesters TAZ from RORγt, Tip60 and Foxp3, thereby negatively regulating TAZ-mediated TH17 differentiation but promoting Treg differentiation.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–8 and Supplementary Tables 1 and 2
Supplementary Dataset 1
Full length gels and blots
Rights and permissions
About this article
Cite this article
Geng, J., Yu, S., Zhao, H. et al. The transcriptional coactivator TAZ regulates reciprocal differentiation of TH17 cells and Treg cells. Nat Immunol 18, 800–812 (2017). https://doi.org/10.1038/ni.3748
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni.3748
This article is cited by
-
TAZ deficiency exacerbates psoriatic pathogenesis by increasing the histamine-releasing factor
Cell & Bioscience (2024)
-
Expected and unexpected effects after systemic inhibition of Hippo transcriptional output in cancer
Nature Communications (2024)
-
Distinct palmitoylation of Foxp3 regulates the function of regulatory T cells via palmitoyltransferases
Cellular & Molecular Immunology (2024)
-
β-Indole-3-acetic acid attenuated collagen-induced arthritis through reducing the ubiquitination of Foxp3 via the AhR-TAZ-Tip60 pathway
Immunologic Research (2024)
-
The Role of H3K27me3-Mediated Th17 Differentiation in Ankylosing Spondylitis
Inflammation (2024)