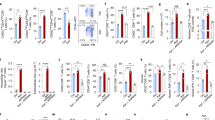
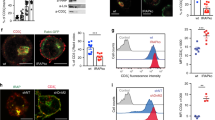
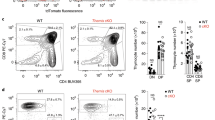
Abstract
T lymphocytes must regulate nutrient uptake to meet the metabolic demands of an immune response. Here we show that the intracellular supply of large neutral amino acids (LNAAs) in T cells was regulated by pathogens and the T cell antigen receptor (TCR). T cells responded to antigen by upregulating expression of many amino-acid transporters, but a single System L ('leucine-preferring system') transporter, Slc7a5, mediated uptake of LNAAs in activated T cells. Slc7a5-null T cells were unable to metabolically reprogram in response to antigen and did not undergo clonal expansion or effector differentiation. The metabolic catastrophe caused by loss of Slc7a5 reflected the requirement for sustained uptake of the LNAA leucine for activation of the serine-threonine kinase complex mTORC1 and for expression of the transcription factor c-Myc. Control of expression of the System L transporter by pathogens is thus a critical metabolic checkpoint for T cells.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Accession codes
Change history
25 June 2013
In the version of this article initially published, the legend for Figure 7b incorrectly included mutant cells. The correct legend should read "...OT-I lymph node T cells...." The error has been corrected in the HTML and PDF versions of the article.
References
Wang, R. & Green, D.R. Metabolic checkpoints in activated T cells. Nat. Immunol. 13, 907–915 (2012).
Carr, E.L. et al. Glutamine uptake and metabolism are coordinately regulated by ERK/MAPK during T lymphocyte activation. J. Immunol. 185, 1037–1044 (2010).
Beugnet, A., Tee, A.R., Taylor, P.M. & Proud, C.G. Regulation of targets of mTOR (mammalian target of rapamycin) signalling by intracellular amino acid availability. Biochem. J. 372, 555 (2003).
Schriever, S.C., Deutsch, M.J., Adamski, J., Roscher, A.A. & Ensenauer, R. Cellular signaling of amino acids towards mTORC1 activation in impaired human leucine catabolism. J. Nutr. Biochem. 10.1016/j.jnutbio.2012.04.018 (2012).
Zheng, Y. et al. Cells are metabolically anergic. J. Immunol. 183, 6095–6101 (2009).
Powell, J.D. & Delgoffe, G.M. The mammalian target of rapamycin: linking T cell differentiation, function, and metabolism. Immunity 33, 301–311 (2010).
Rao, R.R., Li, Q., Odunsi, K. & Shrikant, P.A. The mTOR kinase determines effector versus memory CD8+ T cell fate by regulating the expression of transcription factors T-bet and Eomesodermin. Immunity 32, 67–78 (2010).
Sinclair, L.V. et al. Phosphatidylinositol-3-OH kinase and nutrient-sensing mTOR pathways control T lymphocyte trafficking. Nat. Immunol. 9, 513–521 (2008).
Araki, K. et al. mTOR regulates memory CD8 T-cell differentiation. Nature 460, 108–112 (2009).
Pearce, E.L. et al. Enhancing CD8 T-cell memory by modulating fatty acid metabolism. Nature 460, 103–107 (2009).
Kopf, H., la Rosa de, G.M., Howard, O.M.Z. & Chen, X. Rapamycin inhibits differentiation of Th17 cells and promotes generation of FoxP3+ T regulatory cells. Int. Immunopharmacol. 7, 1819–1824 (2007).
Sauer, S. et al. T cell receptor signaling controls Foxp3 expression via PI3K, Akt, and mTOR. Proc. Natl. Acad. Sci. USA 105, 7797–7802 (2008).
Delgoffe, G.M. et al. The kinase mTOR regulates the differentiation of helper T cells through the selective activation of signaling by mTORC1 and mTORC2. Nat. Immunol. 12, 295–303 (2011).
Finlay, D.K. et al. PDK1/mTOR and Hypoxia-inducible factor 1 integrate metabolism and migration of CD8 T cells. J. Exp. Med. 209, 2441–2453 (2012).
Shi, L.Z. et al. HIF1 -dependent glycolytic pathway orchestrates a metabolic checkpoint for the differentiation of TH17 and Treg cells. J. Exp. Med. 208, 1367–1376 (2011).
Wang, R. et al. The transcription factor Myc controls metabolic reprogramming upon T lymphocyte activation. Immunity 35, 871–882 (2011).
Gottesdiener, K.M. et al. Isolation and structural characterization of the human 4F2 heavy-chain gene, an inducible gene involved in T-lymphocyte activation. Mol. Cell Biol. 8, 3809–3819 (1988).
Lindsten, T., June, C.H., Thompson, C.B. & Leiden, J.M. Regulation of 4F2 heavy-chain gene expression during normal human T-cell activation can be mediated by multiple distinct molecular mechanisms. Mol. Cell Biol. 8, 3820–3826 (1988).
Parmacek, M.S., Karpinski, B.A., Gottesdiener, K.M., Thompson, C.B. & Leiden, J.M. Structure, expression and regulation of the murine 4F2 heavy chain. Nucleic Acids Res. 17, 1915–1931 (1989).
Verrey, F.O. et al. CATs and HATs: the SLC7 family of amino acid transporters. Pflügers Archive European. J. Physiol. 447, 532–542 (2004).
Tsumura, H. et al. The targeted disruption of the CD98 gene results in embryonic lethality. Biochem. Biophys. Res. Commun. 308, 847–851 (2003).
Sato, Y., Heimeier, R.A., Li, C., Deng, C. & Shi, Y.-B. Extracellular domain of CD98hc is required for early murine development. Cell Biosci 1, 7 (2011).
Cantor, J. et al. CD98hc facilitates B cell proliferation and adaptive humoral immunity. Nat. Immunol. 10, 412–419 (2009).
Cantor, J., Slepak, M., Ege, N., Chang, J.T. & Ginsberg, M.H. Loss of T cell CD98 H chain specifically ablates T cell clonal expansion and protects from autoimmunity. J. Immunol. 187, 851–860 (2011).
Liu, Z. et al. Deletion of CD98 heavy chain in T cells results in cardiac allograft acceptance by increasing regulatory T cells. Transplantation 93, 1116–1124 (2012).
Usui, T. et al. Brasilicardin A, a natural immunosuppressant, targets amino acid transport System L. Chem. Biol. 13, 1153–1160 (2006).
Nicklin, P. et al. Bidirectional transport of amino acids regulates mTOR and autophagy. Cell 136, 521–534 (2009).
Navarro, M.N. et al. Protein kinase D2 has a restricted but critical role in T-cell antigen receptor signalling in mature T-cells. Biochem. J. 442, 649–659 (2012).
Heng, T.S.P. et al. The Immunological Genome Project: networks of gene expression in immune cells. Nat. Immunol. 9, 1091–1094 (2008).
Ogilvy, S. et al. Promoter elements of vav drive transgene expression in vivo throughout the hematopoietic compartment. Blood 94, 1855–1863 (1999).
Hann, S.R. & Eisenman, R.N. Proteins encoded by the human c-myc oncogene: differential expression in neoplastic cells. Mol. Cell Biol. 4, 2486–2497 (1984).
Hann, S.R., Abrams, H.D., Rohrschneider, L.R. & Eisenman, R.N. Proteins encoded by v-myc and c-myc oncogenes: identification and localization in acute leukemia virus transformants and bursal lymphoma cell lines. Cell 34, 789–798 (1983).
Delgoffe, G.M. et al. The mTOR kinase differentially regulates effector and regulatory T cell lineage commitment. Immunity 30, 832–844 (2009).
Bar-Peled, L., Schweitzer, L.D., Zoncu, R. & Sabatini, D.M. Ragulator is a GEF for the rag GTPases that signal amino acid levels to mTORC1. Cell 150, 1196–1208 (2012).
Han, J.M. et al. Leucyl-tRNA synthetase is an intracellular leucine sensor for the mTORC1-signaling pathway. Cell 149, 410–424 (2012).
Bonfils, G. et al. Leucyl-tRNA synthetase controls TORC1 via the EGO complex. Mol. Cell 46, 105–110 (2012).
Smith, K.A. & Cantrell, D.A. Interleukin 2 regulates its own receptors. Proc. Natl. Acad. Sci. USA 82, 864–868 (1985).
Kurts, C. et al. Constitutive class I-restricted exogenous presentation of self antigens in vivo. J. Exp. Med. 184, 923–930 (1996).
Pircher, H., Bürki, K., Lang, R., Hengartner, H. & Zinkernagel, R.M. Tolerance induction in double specific T-cell receptor transgenic mice varies with antigen. Nature 342, 559–561 (1989).
Holman, G.D. et al. Cell surface labeling of glucose transporter isoform GLUT4 by bis-mannose photolabel. Correlation with stimulation of glucose transport in rat adipose cells by insulin and phorbol ester. J. Biol. Chem. 265, 18172–18179 (1990).
Pearce, E.L. & Shen, H. Generation of CD8 T cell memory is regulated by IL-12. J. Immunol. 179, 2074–2081 (2007).
Acknowledgements
We thank G. Holman (University of Bath) for anti-Glut1; H. Shen (University of Pennsylvania) for L. monocytogenes; L. Chen for help in the initial characterization of amino-acid transporters expressed by CTLs; members of the Biological Services Unit; R. Clarke of the Flow Cytometry Facility; and members of the D.A.C. laboratory and S. Arthur for critical reading of the manuscript. Supported by the Wellcome Trust (065975/Z/01/A and 097418/Z/11Z to D.A.C., and WT094226 to P.M.T.) and the Intramural Research Program of the National Institute Child Health and Human Development of the US National Institutes of Health.
Author information
Authors and Affiliations
Contributions
L.V.S., all in vitro and most in vivo experiments; J.R., in vivo immunization with NP-OVA and infection with L. monocytogenes; E.E., genomic PCR analysis of Slc7a5fl/flCD4-Cre mice; Y.-B.S., generation of Slc7a5fl/fl mice; P.M.T., conceptual design; and D.A.C., conceptual design and manuscript authorship.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–2 (PDF 224 kb)
Rights and permissions
About this article
Cite this article
Sinclair, L., Rolf, J., Emslie, E. et al. Control of amino-acid transport by antigen receptors coordinates the metabolic reprogramming essential for T cell differentiation. Nat Immunol 14, 500–508 (2013). https://doi.org/10.1038/ni.2556
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni.2556
This article is cited by
-
Dysregulated cellular metabolism in atherosclerosis: mediators and therapeutic opportunities
Nature Metabolism (2024)
-
Immunosurveillance encounters cancer metabolism
EMBO Reports (2024)
-
Slc43a2+ T cell metastasis from spleen to brain in RGNNV infected teleost
Science China Life Sciences (2024)
-
Tumor metabolism rewiring in epithelial ovarian cancer
Journal of Ovarian Research (2023)
-
Amino acid metabolism in immune cells: essential regulators of the effector functions, and promising opportunities to enhance cancer immunotherapy
Journal of Hematology & Oncology (2023)