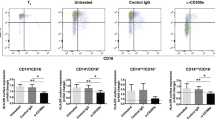
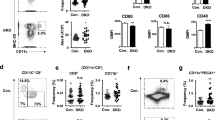
Abstract
The molecular mechanisms involved in the full activation of innate immunity achieved through Toll-like receptors (TLRs) remain to be fully elucidated. In addition to their classical antigen-presenting function, major histocompatibility complex (MHC) class II molecules might mediate reverse signaling. Here we report that deficiency in MHC class II attenuated the TLR-triggered production of proinflammatory cytokines and type I interferon in macrophages and dendritic cells, which protected mice from endotoxin shock. Intracellular MHC class II molecules interacted with the tyrosine kinase Btk via the costimulatory molecule CD40 and maintained Btk activation, but cell surface MHC class II molecules did not. Then, Btk interacted with the adaptor molecules MyD88 and TRIF and thereby promoted TLR signaling. Therefore, intracellular MHC class II molecules can act as adaptors, promoting full activation of TLR-triggered innate immune responses.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Accession codes
Accessions
Mouse Genome Informatics
NCBI Reference Sequence
References
Takeda, K., Kaisho, T. & Akira, S. Toll-like receptors. Annu. Rev. Immunol. 21, 335–376 (2003).
Barton, G.M. & Medzhitov, R. Toll-like receptor signaling pathways. Science 300, 1524–1525 (2003).
O'Neill, L.A. & Bowie, A.G. The family of five: TIR-domain-containing adaptors in Toll-like receptor signalling. Nat. Rev. Immunol. 7, 353–364 (2007).
Kawai, T. & Akira, S. TLR signaling. Semin. Immunol. 19, 24–32 (2007).
Marshak-Rothstein, A. & Rifkin, I.R. Immunologically active autoantigens: the role of toll-like receptors in the development of chronic inflammatory disease. Annu. Rev. Immunol. 25, 419–441 (2007).
Liew, F.Y., Xu, D., Brint, E.K. & O'Neill, L.A. Negative regulation of Toll-like receptor-mediated immune responses. Nat. Rev. Immunol. 5, 446–458 (2005).
Huang, Q. et al. Differential regulation of interleukin 1 receptor and Toll-like receptor signaling by MEKK3. Nat. Immunol. 5, 98–103 (2003).
Liu, X. et al. CaMKII promotes TLR-triggered proinflammatory cytokine and type I interferon production by directly binding and activating TAK1 and IRF3 in macrophages. Blood 112, 4961–4970 (2008).
Schafer, P.H., Pierce, S.K. & Jardetzky, T.S. The structure of MHC class II: a role for dimer of dimers. Semin. Immunol. 7, 389–398 (1995).
McDevitt, H.O. Discovering the role of the major histocompatibility complex in the immune response. Annu. Rev. Immunol. 18, 1–17 (2000).
Al-Daccak, R., Mooney, N. & Charron, D. MHC class II signaling in antigen-presenting cells. Curr. Opin. Immunol. 16, 108–113 (2004).
Mourad, W., Geha, R.S. & Chatila, T. Engagement of major histocompatibility complex class II molecules induces sustained, lymphocyte function-associated molecule 1-dependent cell adhesion. J. Exp. Med. 172, 1513–1516 (1990).
Spertini, F., Chatila, T. & Geha, R.S. Signals delivered via MHC class II molecules synergize with signals delivered via TCR/CD3 to cause proliferation and cytokine gene expression in T cells. J. Immunol. 149, 65–70 (1992).
Hauschildt, S., Bessler, W.G. & Scheipers, P. Engagement of major histocompatibility complex class II molecules leads to nitrite production in bone marrow derived macrophages. Eur. J. Immunol. 23, 2988–2992 (1993).
Trede, N.S., Geha, R.S. & Chatila, T. Transcriptional activation of IL-1β and tumor necrosis factor α genes by MHC class II ligands. J. Immunol. 146, 2310–2315 (1991).
Cambier, J.C. & Lehmann, K.R. Ia-mediated signal transduction leads to proliferation of primed B lymphocytes. J. Exp. Med. 170, 877–886 (1989).
Drenou, B. et al. Caspase-independent pathway of MHC class II antigen-mediated apoptosis of human B lymphocytes. J. Immunol. 163, 4115–4124 (1999).
Bishop, G. Requirements of class II-mediated B cell differentiation for class II cross-linking and cyclic AMP. J. Immunol. 147, 1107–1114 (1991).
Cambier, J.C. et al. Ia binding ligand and cAMP stimulate translocation of PKC in B lymphocytes. Nature 327, 629–632 (1987).
Mooney, N.A., Grillot-Courvalin, C., Hivroz, C., Ju, L.Y. & Charron, D. Early Biochemical events after MHC class II-mediated signaling on human B lymphocytes. J. Immunol. 145, 2070–2076 (1990).
Lane, P.J.L., McConnell, F.M., Schieven, G.L., Clark, E.A. & Ledbetter, J.A. The role of class II molecules in human B cell activation: association with phosphatidyl inositol turnover, protein tyrosine phosphorylation, and proliferation. J. Immunol. 144, 3684–3692 (1990).
Neumann, J., Eis-Hübinger, A.M. & Koch, N. Herpes simplex virus type 1 targets the MHC class II processing pathway for immune evasion. J. Immunol. 171, 3075–3083 (2003).
Lapaque, N. et al. Salmonella regulates polyubiquitination and surface expression of MHC class II antigens. Proc. Natl. Acad. Sci. USA 106, 14052–14057 (2009).
Piani, A. et al. Expression of MHC class II molecules contributes to lipopolysaccharide responsiveness. Eur. J. Immunol. 30, 3140–3146 (2000).
Beharka, A.A., Armstrong, J.W. & Chapes, S.K. Macrophage cell lines derived from major histocompatibility complex II-negative mice. In Vitro Cell. Dev. Biol. Anim. 34, 499–507 (1998).
de Weers, M. et al. B-cell antigen receptor stimulation activates the human Bruton's tyrosine kinase, which is deficient in X-linked agammaglobulinemia. J. Biol. Chem. 269, 23857–23860 (1994).
Wahl, M.I. et al. Phosphorylation of two regulatory tyrosine residues in the activation of Bruton's tyrosine kinase via alternative receptors. Proc. Natl. Acad. Sci. USA 94, 11526–11533 (1997).
Brunner, C., Avots, A., Kreth, H.W., Serfling, E. & Schuster, V. Bruton's tyrosine kinase is activated upon CD40 stimulation in human B lymphocytes. Immunobiology 206, 432–440 (2002).
Jefferies, C.A. et al. Bruton's tyrosine kinase is a Toll/interleukin-1 receptor domain-binding protein that participates in nuclear factor κB activation by Toll-like receptor 4. J. Biol. Chem. 278, 26258–26264 (2003).
Gray, P. et al. MyD88 adapter-like (Mal) is phosphorylated by Bruton's tyrosine kinase during TLR2 and TLR4 signal transduction. J. Biol. Chem. 281, 10489–10495 (2006).
Mansell, A. et al. Suppressor of cytokine signaling 1 negatively regulates Toll-like receptor signaling by mediating Mal degradation. Nat. Immunol. 7, 148–155 (2006).
Horwood, N.J. et al. Bruton's tyrosine kinase is required for lipopolysaccharide-induced tumor necrosis factor-α production. J. Exp. Med. 197, 1603–1611 (2003).
Mukhopadhyay, S. et al. Macrophage effector functions controlled by Bruton's tyrosine kinase are more crucial than the cytokine balance of T cell responses for microfilarial clearance. J. Immunol. 168, 2914–2921 (2002).
Bonnefoy, J.Y. et al. The low-affinity receptor for IgE (CD23) on B lymphocytes is spatially associated with HLA-DR antigens. J. Exp. Med. 167, 57–72 (1988).
Bradbury, L.E. et al. The CD19/CD21 signal transducing complex of human B lymphocytes includes the target of antiproliferative antibody-1 and Leu-13 molecules. J. Immunol. 149, 2841–2850 (1992).
Léveillé, C., AL-Daccak, R. & Mourad, W. CD20 is physically and functionally coupled to MHC class II and CD40 on human B cell lines. Eur. J. Immunol. 29, 65–74 (1999).
Léveillé, C., Chandad, F., Al-Daccak, R. & Mourad, W. CD40 associates with the MHC class II molecules on human B cells. Eur. J. Immunol. 29, 3516–3526 (1999).
Harton, J.A., Van Hagen, A.E. & Bishop, G.A. The cytoplasmic and transmembrane domains of MHC class II β chains deliver distinct signals required for MHC class II-mediated B cell activation. Immunity 3, 349–358 (1995).
Harton, J.A. & Bishop, G.A. Length and sequence requirements of the cytoplasmic domain of the Aβ molecule for class II-mediated B cell signaling. J. Immunol. 151, 5282–5289 (1993).
Frei, R. et al. MHC class II molecules enhance Toll-like receptor mediated innate immune responses. PLoS ONE 5, e8808 (2010).
Appel, S.H., Wellhausen, S.R., Montgomery, R., DeWeese, R.C. & Polk, H.C. Jr. Experimental and clinical significance of endotoxin-dependent HLA-DR expression on monocytes. J. Surg. Res. 47, 39–44 (1989).
Astiz, M., Saha, D., Lustbader, D., Lin, R. & Rackow, E. Monocyte response to bacterial toxins, expression of cell surface receptors, and release of anti-inflammatory cytokines during sepsis. J. Lab. Clin. Med. 128, 594–600 (1996).
Hershman, M.J., Cheadle, W.G., Wellhausen, S.R., Davidson, P.F. & Polk, H.C. Jr. Monocyte HLA-DR antigen expression characterizes clinical outcome in the trauma patient. Br. J. Surg. 77, 204–207 (1990).
Docke, W.D. et al. Monocyte deactivation in septic patients: restoration by IFN-γ treatment. Nat. Med. 3, 678–681 (1997).
Wang, C. et al. The E3 ubiquitin ligase Nrdp1 'preferentially' promotes TLR-mediated production of type I interferon. Nat. Immunol. 10, 744–752 (2009).
An, H. et al. Phosphatase SHP-1 promotes TLR- and RIG-I-activated production of type I interferon by inhibiting the kinase IRAK1. Nat. Immunol. 9, 542–550 (2008).
Haziot, A. et al. Resistance to endotoxin shock and reduced dissemination of gram-negative bacteria in CD14-deficient mice. Immunity 4, 407–414 (1996).
Han, C. et al. Integrin CD11b negatively regulates TLR-triggered inflammatory responses by activating Syk and promoting degradation of MyD88 and TRIF via Cbl-b. Nat. Immunol. 11, 734–742 (2010).
Acknowledgements
We thank P. Ma, M. Jin and Y. Li for technical assistance; and N. Li, H. An, T. Chen, S. Xu and C. Han for discussions. Supported by the National Key Basic Research Program of China (2007CB512403), National 115 Key Project (2008ZX10002-008, 2009ZX09503-023) and the National Natural Science Foundation of China (30721091).
Author information
Authors and Affiliations
Contributions
X.C. and X.L. designed the experiments; X.L., Z.Z., D.L., L.X., F.M., P.Z. and H.Y. did the experiments; X.C. and X.L. analyzed data and wrote the paper; and X.C. was responsible for research supervision, coordination and strategy.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–16 (PDF 2994 kb)
Rights and permissions
About this article
Cite this article
Liu, X., Zhan, Z., Li, D. et al. Intracellular MHC class II molecules promote TLR-triggered innate immune responses by maintaining activation of the kinase Btk. Nat Immunol 12, 416–424 (2011). https://doi.org/10.1038/ni.2015
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni.2015
This article is cited by
-
Repopulating Kupffer cells originate directly from hematopoietic stem cells
Stem Cell Research & Therapy (2023)
-
Bruton tyrosine kinase inhibitors for multiple sclerosis
Nature Reviews Neurology (2023)
-
Soluble CD4 effectively prevents excessive TLR activation of resident macrophages in the onset of sepsis
Signal Transduction and Targeted Therapy (2023)
-
Histone demethylase KDM5B licenses macrophage-mediated inflammatory responses by repressing Nfkbia transcription
Cell Death & Differentiation (2023)
-
Opposing effects of apoE2 and apoE4 on microglial activation and lipid metabolism in response to demyelination
Molecular Neurodegeneration (2022)