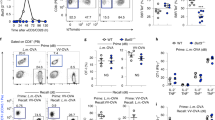
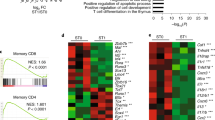
Abstract
Several gene-deficiency models promote the development of innate CD8+ T cells that have diverse T cell antigen receptors (TCRs) but have a memory phenotype and rapidly produce cytokines. We demonstrate here that similar cells developed in mice deficient in the transcription factor KLF2. However, this was not due to intrinsic deficiency in KLF2 but instead was due to interleukin 4 (IL-4) produced by an expanded population of T cells expressing the transcription factor PLZF. The development of innate CD8+ T cells in mice deficient in the tyrosine kinase Itk and coactivator CBP was also attributable to this IL-4-dependent mechanism. Finally, we show that the same mechanism drove the differentiation of innate CD8+ T cells in BALB/c mice. Our findings identify a previously unknown mechanism of regulation of CD8+ T cells via the production of IL-4 by PLZF+ T cells.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Williams, M.A. & Bevan, M.J. Effector and memory CTL differentiation. Annu. Rev. Immunol. 25, 171–192 (2007).
Bendelac, A., Savage, P.B. & Teyton, L. The biology of NKT cells. Annu. Rev. Immunol. 25, 297–336 (2007).
Treiner, E. & Lantz, O. CD1d- and MR1-restricted invariant T cells: of mice and men. Curr. Opin. Immunol. 18, 519–526 (2006).
Matsuda, J.L., Mallevaey, T., Scott-Browne, J. & Gapin, L. CD1d-restricted iNKT cells, the 'Swiss-Army knife' of the immune system. Curr. Opin. Immunol. 20, 358–368 (2008).
Kovalovsky, D. et al. The BTB-zinc finger transcriptional regulator PLZF controls the development of invariant natural killer T cell effector functions. Nat. Immunol. 9, 1055–1064 (2008).
Savage, A.K. et al. The transcription factor PLZF directs the effector program of the NKT cell lineage. Immunity 29, 391–403 (2008).
Kuo, C.T., Veselits, M.L. & Leiden, J.M. LKLF: A transcriptional regulator of single-positive T cell quiescence and survival. Science 277, 1986–1990 (1997).
Carlson, C.M. et al. Kruppel-like factor 2 regulates thymocyte and T-cell migration. Nature 442, 299–302 (2006).
Matloubian, M. et al. Lymphocyte egress from thymus and peripheral lymphoid organs is dependent on S1P receptor 1. Nature 427, 355–360 (2004).
Bai, A., Hu, H., Yeung, M. & Chen, J. Kruppel-like factor 2 controls T cell trafficking by activating L-selectin (CD62L) and sphingosine-1-phosphate receptor 1 transcription. J. Immunol. 178, 7632–7639 (2007).
Sebzda, E., Zou, Z., Lee, J.S., Wang, T. & Kahn, M.L. Transcription factor KLF2 regulates the migration of naive T cells by restricting chemokine receptor expression patterns. Nat. Immunol. 9, 292–300 (2008).
Weinreich, M.A. et al. KLF2 transcription-factor deficiency in T cells results in unrestrained cytokine production and upregulation of bystander chemokine receptors. Immunity 31, 122–130 (2009).
Qin, S. et al. The chemokine receptors CXCR3 and CCR5 mark subsets of T cells associated with certain inflammatory reactions. J. Clin. Invest. 101, 746–754 (1998).
Intlekofer, A.M. et al. Effector and memory CD8+ T cell fate coupled by T-bet and eomesodermin. Nat. Immunol. 6, 1236–1244 (2005).
Intlekofer, A.M. et al. Anomalous type 17 response to viral infection by CD8+ T cells lacking T-bet and eomesodermin. Science 321, 408–411 (2008).
Lewis, D.B. et al. Interleukin 4 expressed in situ selectively alters thymocyte development. J. Exp. Med. 173, 89–100 (1991).
Berg, R.E., Crossley, E., Murray, S. & Forman, J. Memory CD8+ T cells provide innate immune protection against Listeria monocytogenes in the absence of cognate antigen. J. Exp. Med. 198, 1583–1593 (2003).
Kreslavsky, T. et al. TCR-inducible PLZF transcription factor required for innate phenotype of a subset of γδ T cells with restricted TCR diversity. Proc. Natl. Acad. Sci. USA 106, 12453–12458 (2009).
Felices, M., Yin, C.C., Kosaka, Y., Kang, J. & Berg, L.J. Tec kinase Itk in γδT cells is pivotal for controlling IgE production in vivo. Proc. Natl. Acad. Sci. USA 106, 8308–8313 (2009).
Buaas, F.W. et al. Plzf is required in adult male germ cells for stem cell self-renewal. Nat. Genet. 36, 647–652 (2004).
Prince, A.L., Yin, C.C., Enos, M.E., Felices, M. & Berg, L.J. The Tec kinases Itk and Rlk regulate conventional versus innate T-cell development. Immunol. Rev. 228, 115–131 (2009).
Broussard, C. et al. Altered development of CD8+ T cell lineages in mice deficient for the Tec kinases Itk and Rlk. Immunity 25, 93–104 (2006).
Atherly, L.O. et al. The Tec family tyrosine kinases Itk and Rlk regulate the development of conventional CD8+ T cells. Immunity 25, 79–91 (2006).
Qi, Q. et al. Enhanced development of CD4+ γδ T cells in the absence of Itk results in elevated IgE production. Blood 114, 564–571 (2009).
Gadue, P. & Stein, P.L.N.K. T cell precursors exhibit differential cytokine regulation and require Itk for efficient maturation. J. Immunol. 169, 2397–2406 (2002).
Au-Yeung, B.B. & Fowell, D.J. A key role for Itk in both IFN γ and IL-4 production by NKT cells. J. Immunol. 179, 111–119 (2007).
Fukuyama, T. et al. Histone acetyltransferase CBP is vital to demarcate conventional and innate CD8+ T-cell development. Mol. Cell. Biol. 29, 3894–3904 (2009).
Rymarchyk, S.L. et al. Widespread natural variation in murine natural killer T-cell number and function. Immunology 125, 331–343 (2008).
Hammond, K.J. et al. CD1d-restricted NKT cells: an interstrain comparison. J. Immunol. 167, 1164–1173 (2001).
Azuara, V., Grigoriadou, K., Lembezat, M.P., Nagler-Anderson, C. & Pereira, P. Strain-specific TCR repertoire selection of IL-4-producing Thy-1 dull γδ thymocytes. Eur. J. Immunol. 31, 205–214 (2001).
Readinger, J.A., Mueller, K.L., Venegas, A.M., Horai, R. & Schwartzberg, P.L. Tec kinases regulate T-lymphocyte development and function: new insights into the roles of Itk and Rlk/Txk. Immunol. Rev. 228, 93–114 (2009).
Lauritsen, J.P. et al. Marked induction of the helix-loop-helix protein Id3 promotes the γδ T cell fate and renders their functional maturation Notch independent. Immunity 31, 565–575 (2009).
Ueda-Hayakawa, I., Mahlios, J. & Zhuang, Y. Id3 restricts the developmental potential of γδ lineage during thymopoiesis. J. Immunol. 182, 5306–5316 (2009).
Verykokakis, M. et al. Inhibitor of DNA binding 3 limits development of murine slam-associated adaptor protein-dependent 'innate' γδ T cells. PLoS One 5, e9303 (2010).
Alonzo, E.S. et al. Development of promyelocytic zinc finger and ThPOK-expressing innate γδ T cells is controlled by strength of TCR signaling and Id3. J. Immunol. 184, 1268–1279 (2010).
Allende, M.L. et al. S1P1 receptor expression regulates emergence of NKT cells in peripheral tissues. FASEB J. 22, 307–315 (2008).
Raberger, J. et al. The transcriptional regulator PLZF induces the development of CD44 high memory phenotype T cells. Proc. Natl. Acad. Sci. USA 105, 17919–17924 (2008).
Cerundolo, V., Silk, J.D., Masri, S.H. & Salio, M. Harnessing invariant NKT cells in vaccination strategies. Nat. Rev. Immunol. 9, 28–38 (2009).
Morris, S.C. et al. Endogenously produced IL-4 nonredundantly stimulates CD8+ T cell proliferation. J. Immunol. 182, 1429–1438 (2009).
Tan, J.T. et al. IL-7 is critical for homeostatic proliferation and survival of naive T cells. Proc. Natl. Acad. Sci. USA 98, 8732–8737 (2001).
Ueda, N. et al. CD1d-restricted NKT cell activation enhanced homeostatic proliferation of CD8+ T cells in a manner dependent on IL-4. Int. Immunol. 18, 1397–1404 (2006).
Lee, Y.J., Jung, K.C. & Park, S.H. MHC class II-dependent T-T interactions create a diverse, functional and immunoregulatory reaction circle. Immunol. Cell Biol. 87, 65–71 (2009).
Lee, Y.J. et al. Generation of PLZF+ CD4+ T cells via MHC class II-dependent thymocyte-thymocyte interaction is a physiological process in humans. J. Exp. Med. 207, 237–246 (2010).
Huck, K. et al. Girls homozygous for an IL-2-inducible T cell kinase mutation that leads to protein deficiency develop fatal EBV-associated lymphoproliferation. J. Clin. Invest. 119, 1350–1358 (2009).
Vesosky, B., Rottinghaus, E.K., Davis, C. & Turner, J. CD8+ T Cells in old mice contribute to the innate immune response to Mycobacterium tuberculosis via interleukin-12p70-dependent and antigen-independent production of γ interferon. Infect. Immun. 77, 3355–3363 (2009).
Dhanji, S., Chow, M.T. & Teh, H.S. Self-antigen maintains the innate antibacterial function of self-specific CD8+ T cells in vivo. J. Immunol. 177, 138–146 (2006).
Haque, A. et al. Role of T cells in innate and adaptive immunity against murine Burkholderia pseudomallei infection. J. Infect. Dis. 193, 370–379 (2006).
Berg, R.E., Crossley, E., Murray, S. & Forman, J. Relative contributions of NK and CD8+ T cells to IFN-γ mediated innate immune protection against Listeria monocytogenes. J. Immunol. 175, 1751–1757 (2005).
Acknowledgements
We thank F. Finkelman (University of Cincinnati) for B6 Il4ra−/− mice; D. Kioussis (National Institute for Medical Research, London) for Vav-Cre mice; J. Lingrel (University of Cincinnati) for Klf2fl/fl mice; R. Proia (National Institute of Diabetes and Digestive and Kidney Diseases, US National Institutes of Health) for S1pr1fl/fl mice; Y. Shimizu (University of Minnesota) for Itk−/− mice; R. Braun (Jackson Laboratory) for PLZFlu/lu mice; D. Masopust (University of Minnesota) for SMARTA TCR-transgenic mice; P. Brindle (St. Jude Children's Research Hospital) for Cbpfl/flLck-Cre bone marrow; X. Ding, J. Vevea, J. Reyes and C. Mora-Solano for technical assistance; and L. Berg and P. Schwartzberg for discussions. H. O'Donnell did initial experiments with Il4ra−/− mice. Supported by the US National Institutes of Health (R01 AI35296 to K.A.H., R37 AI38903 to S.C.J., and T32 AI007313 to M.A.W.) and the University of Minnesota (M.A.W.).
Author information
Authors and Affiliations
Contributions
M.A.W. designed and did experiments, analyzed data and wrote the manuscript; O.A.O. did analysis that inspired the direction of this research and provided input on interpretation; S.C.J. provided input to the research design and interpretation; and K.A.H. conceptualized the research, directed the study, analyzed data and edited the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–5 (PDF 1062 kb)
Rights and permissions
About this article
Cite this article
Weinreich, M., Odumade, O., Jameson, S. et al. T cells expressing the transcription factor PLZF regulate the development of memory-like CD8+ T cells. Nat Immunol 11, 709–716 (2010). https://doi.org/10.1038/ni.1898
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni.1898
This article is cited by
-
IL-4/IL-4 Ab complex enhances the accumulation of both antigen-specific and bystander CD8 T cells in mouse lungs infected with influenza A virus
Laboratory Animal Research (2023)
-
Single-cell analysis reveals differences among iNKT cells colonizing peripheral organs and identifies Klf2 as a key gene for iNKT emigration
Cell Discovery (2022)
-
Protein abundance of the cytokine receptor γc controls the thymic generation of innate-like T cells
Cellular and Molecular Life Sciences (2022)
-
SRSF1 plays a critical role in invariant natural killer T cell development and function
Cellular & Molecular Immunology (2021)
-
Signals for antigen-independent differentiation of memory CD8+ T cells
Cellular and Molecular Life Sciences (2021)