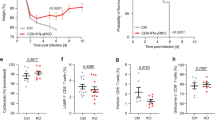
Abstract
Cytokines are soluble proteins that regulate immune responses. The present paradigm is that cytokine production in lymphoid tissues is tightly localized and signaling occurs between conjugate cells. Here we assess cytokine signaling during infection by measuring in vivo phosphorylation of intracellular signal transducer and activator of transcription (STAT) proteins. We show that interferon-γ (IFN-γ) and interleukin 4 (IL-4) signaled to the majority of lymphocytes throughout the reactive lymph node and that IL-4 conditioning of naive, bystander cells was sufficient to override opposing T helper type 1 (TH1) polarization. Our results demonstrate that despite localized production, cytokines can permeate a lymph node and modify the majority of cells therein. Cytokine conditioning of bystander cells could provide a mechanism by which chronic worm infections subvert the host response to subsequent infections or vaccination attempts.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
O'Garra, A. & Murphy, K. Role of cytokines in determining T-lymphocyte function. Curr. Opin. Immunol. 6, 458–466 (1994).
Anthony, R.M., Rutitzky, L.I., Urban, J.F. Jr., Stadecker, M.J. & Gause, W.C. Protective immune mechanisms in helminth infection. Nat. Rev. Immunol. 7, 975–987 (2007).
Zhu, J. & Paul, W.E. CD4 T cells: fates, functions, and faults. Blood 112, 1557–1569 (2008).
Shimoda, K. et al. Lack of IL-4-induced Th2 response and IgE class switching in mice with disrupted Stat6 gene. Nature 380, 630–633 (1996).
Kaplan, M.H., Schindler, U., Smiley, S.T. & Grusby, M.J. Stat6 is required for mediating responses to IL-4 and for development of Th2 cells. Immunity 4, 313–319 (1996).
Finkelman, F.D. et al. Cytokine regulation of host defense against parasitic gastrointestinal nematodes: lessons from studies with rodent models. Annu. Rev. Immunol. 15, 505–533 (1997).
Reinhardt, R.L., Liang, H.E. & Locksley, R.M. Cytokine-secreting follicular T cells shape the antibody repertoire. Nat. Immunol. 10, 385–393 (2009).
King, I.L. & Mohrs, M. IL-4-producing CD4+ T cells in reactive lymph nodes during helminth infection are T follicular helper cells. J. Exp. Med. 206, 1001–1007 (2009).
Zaretsky, A.G. et al. T follicular helper cells differentiate from Th2 cells in response to helminth antigens. J. Exp. Med. 206, 991–999 (2009).
Poo, W.J., Conrad, L. & Janeway, C.A. Jr. Receptor-directed focusing of lymphokine release by helper T cells. Nature 332, 378–380 (1988).
Kupfer, A., Mosmann, T.R. & Kupfer, H. Polarized expression of cytokines in cell conjugates of helper T cells and splenic B cells. Proc. Natl. Acad. Sci. USA 88, 775–779 (1991).
Paul, W.E. & Seder, R.A. Lymphocyte responses and cytokines. Cell 76, 241–251 (1994).
O'Shea, J.J. Jaks, STATs, cytokine signal transduction, and immunoregulation: are we there yet? Immunity 7, 1–11 (1997).
Takeda, K. et al. Essential role of Stat6 in IL-4 signalling. Nature 380, 627–630 (1996).
Nelms, K., Keegan, A.D., Zamorano, J., Ryan, J.J. & Paul, W.E. The IL-4 receptor: signaling mechanisms and biologic functions. Annu. Rev. Immunol. 17, 701–738 (1999).
Mohrs, K., Wakil, A.E., Killeen, N., Locksley, R.M. & Mohrs, M.A. Two-step process for cytokine production revealed by IL-4 dual-reporter mice. Immunity 23, 419–429 (2005).
Mohrs, M., Shinkai, K., Mohrs, K. & Locksley, R.M. Analysis of type 2 immunity in vivo with a bicistronic IL-4 reporter. Immunity 15, 303–311 (2001).
Perona-Wright, G., Mohrs, K., Mayer, K.D. & Mohrs, M. Differential regulation of IL-4Rα expression by antigen versus cytokine stimulation characterizes Th2 progression in vivo. J. Immunol. 184, 615–623 (2010).
Ohara, J. & Paul, W.E. Up-regulation of interleukin 4/B-cell stimulatory factor 1 receptor expression. Proc. Natl. Acad. Sci. USA 85, 8221–8225 (1988).
Schindler, C., Shuai, K., Prezioso, V.R. & Darnell, J.E. Jr. Interferon-dependent tyrosine phosphorylation of a latent cytoplasmic transcription factor. Science 257, 809–813 (1992).
Maldonado, R.A. et al. Control of T helper cell differentiation through cytokine receptor inclusion in the immunological synapse. J. Exp. Med. 206, 877–892 (2009).
Bach, E.A. et al. Ligand-induced autoregulation of IFN-γ receptor β chain expression in T helper cell subsets. Science 270, 1215–1218 (1995).
Zhou, F. Molecular mechanisms of IFN-γ to up-regulate MHC class I antigen processing and presentation. Int. Rev. Immunol. 28, 239–260 (2009).
Manetti, R. et al. Natural killer cell stimulatory factor (interleukin 12 [IL-12]) induces T helper type 1 (Th1)-specific immune responses and inhibits the development of IL-4-producing Th cells. J. Exp. Med. 177, 1199–1204 (1993).
Trinchieri, G. Interleukin-12: a proinflammatory cytokine with immunoregulatory functions that bridge innate resistance and antigen-specific adaptive immunity. Annu. Rev. Immunol. 13, 251–276 (1995).
Szabo, S.J., Dighe, A.S., Gubler, U. & Murphy, K.M. Regulation of the interleukin (IL)-12R β2 subunit expression in developing T helper 1 (Th1) and Th2 cells. J. Exp. Med. 185, 817–824 (1997).
Perona-Wright, G. et al. Systemic but not local infections elicit immunosuppressive IL-10 production by natural killer cells. Cell Host Microbe 6, 503–512 (2009).
Ekkens, M.J. et al. The role of OX40 ligand interactions in the development of the Th2 response to the gastrointestinal nematode parasite Heligmosomoides polygyrus. J. Immunol. 170, 384–393 (2003).
Hotson, A.N., Hardy, J.W., Hale, M.B., Contag, C.H. & Nolan, G.P. The T cell STAT signaling network is reprogrammed within hours of bacteremia via secondary signals. J. Immunol. 182, 7558–7568 (2009).
Andrews, R.P., Ericksen, M.B., Cunningham, C.M., Daines, M.O. & Hershey, G.K. Analysis of the life cycle of stat6. Continuous cycling of STAT6 is required for IL-4 signaling. J. Biol. Chem. 277, 36563–36569 (2002).
Galizzi, J.P., Zuber, C.E., Cabrillat, H., Djossou, O. & Banchereau, J. Internalization of human interleukin 4 and transient down-regulation of its receptor in the CD23-inducible Jijoye cells. J. Biol. Chem. 264, 6984–6989 (1989).
Abbas, A.K., Murphy, K.M. & Sher, A. Functional diversity of helper T lymphocytes. Nature 383, 787–793 (1996).
Robinson, R.T. et al. Yersinia pestis evades TLR4-dependent induction of IL-12(p40)2 by dendritic cells and subsequent cell migration. J. Immunol. 181, 5560–5567 (2008).
Maldonado, R.A., Irvine, D.J., Schreiber, R. & Glimcher, L.H. A role for the immunological synapse in lineage commitment of CD4 lymphocytes. Nature 431, 527–532 (2004).
Miller, M.J., Wei, S.H., Parker, I. & Cahalan, M.D. Two-photon imaging of lymphocyte motility and antigen response in intact lymph node. Science 296, 1869–1873 (2002).
Campbell, D.J. & Butcher, E.C. Rapid acquisition of tissue-specific homing phenotypes by CD4+ T cells activated in cutaneous or mucosal lymphoid tissues. J. Exp. Med. 195, 135–141 (2002).
Weninger, W., Crowley, M.A., Manjunath, N. & von Andrian, U.H. Migratory properties of naive, effector, and memory CD8+ T cells. J. Exp. Med. 194, 953–966 (2001).
Huse, M., Lillemeier, B.F., Kuhns, M.S., Chen, D.S. & Davis, M.M. T cells use two directionally distinct pathways for cytokine secretion. Nat. Immunol. 7, 247–255 (2006).
Haque, S.J. et al. Receptor-associated constitutive protein tyrosine phosphatase activity controls the kinase function of JAK1. Proc. Natl. Acad. Sci. USA 94, 8563–8568 (1997).
Haque, S.J., Harbor, P., Tabrizi, M., Yi, T. & Williams, B.R. Protein-tyrosine phosphatase Shp-1 is a negative regulator of IL-4- and IL-13-dependent signal transduction. J. Biol. Chem. 273, 33893–33896 (1998).
Hanson, E.M., Dickensheets, H., Qu, C.K., Donnelly, R.P. & Keegan, A.D. Regulation of the dephosphorylation of Stat6. Participation of Tyr-713 in the interleukin-4 receptor α, the tyrosine phosphatase SHP-1, and the proteasome. J. Biol. Chem. 278, 3903–3911 (2003).
Tanaka, Y. et al. T helper type 2 differentiation and intracellular trafficking of the interleukin 4 receptor-α subunit controlled by the Rac activator Dock2. Nat. Immunol. 8, 1067–1075 (2007).
Skrenta, H., Yang, Y., Pestka, S. & Fathman, C.G. Ligand-independent down-regulation of IFN-γ receptor 1 following TCR engagement. J. Immunol. 164, 3506–3511 (2000).
Toellner, K.M. et al. T helper 1 (Th1) and Th2 characteristics start to develop during T cell priming and are associated with an immediate ability to induce immunoglobulin class switching. J. Exp. Med. 187, 1193–1204 (1998).
Sutton, T.L. et al. Anti-inflammatory mechanisms of enteric Heligmosomoides polygyrus infection against trinitrobenzene sulfonic acid-induced colitis in a murine model. Infect. Immun. 76, 4772–4782 (2008).
Elliott, D.E. et al. Colonization with Heligmosomoides polygyrus suppresses mucosal IL-17 production. J. Immunol. 181, 2414–2419 (2008).
Bethony, J. et al. Soil-transmitted helminth infections: ascariasis, trichuriasis, and hookworm. Lancet 367, 1521–1532 (2006).
Mohrs, M. et al. Differences between IL-4- and IL-4 receptor α-deficient mice in chronic leishmaniasis reveal a protective role for IL-13 receptor signaling. J. Immunol. 162, 7302–7308 (1999).
Mayer, K.D. et al. The functional heterogeneity of type 1 effector T cells in response to infection is related to the potential for IFN-γ production. J. Immunol. 174, 7732–7739 (2005).
MacDonald, A.S., Straw, A.D., Bauman, B. & Pearce, E.J. CD8− dendritic cell activation status plays an integral role in influencing Th2 response development. J. Immunol. 167, 1982–1988 (2001).
Acknowledgements
We thank L.L. Johnson for T. gondii infection; J.-S. Lin and S.T. Smiley (both with the Trudeau Institute) for heat-killed Y. pestis; E.J. Pearce (Trudeau Institute) for S. mansoni eggs; R.T. Robinson and A.M. Cooper (both with the Trudeau Institute) for Ifng−/− mice; and E.J. Pearce and D. Gray for discussion and comments on the manuscript. Supported by the Trudeau Institute and the National Institutes of Health (AI072296 and AI076479).
Author information
Authors and Affiliations
Contributions
G.P.-W. and M.M. designed the research and prepared the manuscript; G.P.-W. and K.M. did the experiments; G.P.-W. analyzed and interpreted the results; and M.M. guided the study.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–9 (PDF 1184 kb)
Rights and permissions
About this article
Cite this article
Perona-Wright, G., Mohrs, K. & Mohrs, M. Sustained signaling by canonical helper T cell cytokines throughout the reactive lymph node. Nat Immunol 11, 520–526 (2010). https://doi.org/10.1038/ni.1866
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni.1866
This article is cited by
-
Tissue-based IL-10 signalling in helminth infection limits IFNγ expression and promotes the intestinal Th2 response
Mucosal Immunology (2022)
-
Long-distance modulation of bystander tumor cells by CD8+ T-cell-secreted IFN-γ
Nature Cancer (2020)
-
Bystander IFN-γ activity promotes widespread and sustained cytokine signaling altering the tumor microenvironment
Nature Cancer (2020)
-
Carpet-bombing tumors with IFN-γ
Nature Cancer (2020)
-
Virtual memory CD8 T cells expanded by helminth infection confer broad protection against bacterial infection
Mucosal Immunology (2019)