Abstract
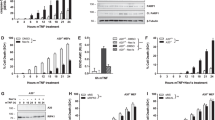
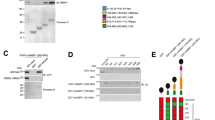
The adaptor and signaling proteins TRAF2, TRAF3, cIAP1 and cIAP2 may inhibit alternative nuclear factor-κB (NF-κB) signaling in resting cells by targeting NF-κB–inducing kinase (NIK) for ubiquitin-dependent degradation, thus preventing processing of the NF-κB2 precursor protein p100 to release p52. However, the respective functions of TRAF2 and TRAF3 in NIK degradation and activation of alternative NF-κB signaling have remained elusive. We now show that CD40 or BAFF receptor activation result in TRAF3 degradation in a cIAP1-cIAP2- and TRAF2-dependent way owing to enhanced cIAP1, cIAP2 TRAF3-directed ubiquitin ligase activity. Receptor-induced activation of cIAP1 and cIAP2 correlated with their K63-linked ubiquitination by TRAF2. Degradation of TRAF3 prevented association of NIK with the cIAP1-cIAP2-TRAF2 ubiquitin ligase complex, which resulted in NIK stabilization and NF-κB2-p100 processing. Constitutive activation of this pathway causes perinatal lethality and lymphoid defects.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Ghosh, S. & Karin, M. Missing pieces in the NF-kappaB puzzle. Cell 109 (suppl.), S81–S96 (2002).
Li, Q. & Verma, I.M. NF-kappaB regulation in the immune system. Nat. Rev. Immunol. 2, 725–734 (2002).
Ben-Neriah, Y. Regulatory functions of ubiquitination in the immune system. Nat. Immunol. 3, 20–26 (2002).
Senftleben, U. et al. Activation by IKKalpha of a second, evolutionary conserved, NF-kappa B signaling pathway. Science 293, 1495–1499 (2001).
Fong, A. & Sun, S.C. Genetic evidence for the essential role of beta-transducin repeat-containing protein in the inducible processing of NF-kappa B2/p100. J. Biol. Chem. 277, 22111–22114 (2002).
Xiao, G., Harhaj, E.W. & Sun, S.C. NF-kappaB-inducing kinase regulates the processing of NF-kappaB2 p100. Mol. Cell 7, 401–409 (2001).
Derudder, E. et al. RelB/p50 dimers are differentially regulated by tumor necrosis factor-alpha and lymphotoxin-beta receptor activation: critical roles for p100. J. Biol. Chem. 278, 23278–23284 (2003).
Yilmaz, Z.B., Weih, D.S., Sivakumar, V. & Weih, F. RelB is required for Peyer's patch development: differential regulation of p52-RelB by lymphotoxin and TNF. EMBO J. 22, 121–130 (2003).
Coope, H.J. et al. CD40 regulates the processing of NF-kappaB2 p100 to p52. EMBO J. 21, 5375–5385 (2002).
Claudio, E., Brown, K., Park, S., Wang, H. & Siebenlist, U. BAFF-induced NEMO-independent processing of NF-kappa B2 in maturing B cells. Nat. Immunol. 3, 958–965 (2002).
Bishop, G.A. The multifaceted roles of TRAFs in the regulation of B-cell function. Nat. Rev. Immunol. 4, 775–786 (2004).
Hacker, H. & Karin, M. Regulation and function of IKK and IKK-related kinases. Sci. STKE 2006, re13 (2006).
Chen, Z.J., Bhoj, V. & Seth, R.B. Ubiquitin, TAK1 and IKK: is there a connection? Cell Death Differ. 13, 687–692 (2006).
Chung, J.Y., Park, Y.C., Ye, H. & Wu, H. All TRAFs are not created equal: common and distinct molecular mechanisms of TRAF-mediated signal transduction. J. Cell Sci. 115, 679–688 (2002).
Shi, C.S. & Kehrl, J.H. Tumor necrosis factor (TNF)-induced germinal center kinase-related (GCKR) and stress-activated protein kinase (SAPK) activation depends upon the E2/E3 complex Ubc13-Uev1A/TNF receptor-associated factor 2 (TRAF2). J. Biol. Chem. 278, 15429–15434 (2003).
Deng, L. et al. Activation of the IkappaB kinase complex by TRAF6 requires a dimeric ubiquitin-conjugating enzyme complex and a unique polyubiquitin chain. Cell 103, 351–361 (2000).
Tada, K. et al. Critical roles of TRAF2 and TRAF5 in tumor necrosis factor-induced NF-kappa B activation and protection from cell death. J. Biol. Chem. 276, 36530–36534 (2001).
Ishida, T. et al. Identification of TRAF6, a novel tumor necrosis factor receptor-associated factor protein that mediates signaling from an amino-terminal domain of the CD40 cytoplasmic region. J. Biol. Chem. 271, 28745–28748 (1996).
Lomaga, M.A. et al. TRAF6 deficiency results in osteopetrosis and defective interleukin-1, CD40, and LPS signaling. Genes Dev. 13, 1015–1024 (1999).
Hacker, H. et al. Specificity in Toll-like receptor signalling through distinct effector functions of TRAF3 and TRAF6. Nature 439, 204–207 (2006).
Rothe, M., Sarma, V., Dixit, V.M. & Goeddel, D.V. TRAF2-mediated activation of NF-kappa B by TNF receptor 2 and CD40. Science 269, 1424–1427 (1995).
Cao, Z., Xiong, J., Takeuchi, M., Kurama, T. & Goeddel, D.V. TRAF6 is a signal transducer for interleukin-1. Nature 383, 443–446 (1996).
Matsuzawa, A. et al. Essential cytoplasmic translocation of a cytokine receptor-assembled signaling complex. Science 321, 663–668 (2008).
Grech, A.P. et al. TRAF2 differentially regulates the canonical and noncanonical pathways of NF-kappaB activation in mature B cells. Immunity 21, 629–642 (2004).
He, J.Q. et al. Rescue of TRAF3-null mice by p100 NF-kappa B deficiency. J. Exp. Med. 203, 2413–2418 (2006).
Gardam, S., Sierro, F., Basten, A., Mackay, F. & Brink, R. TRAF2 and TRAF3 signal adapters act cooperatively to control the maturation and survival signals delivered to B cells by the BAFF receptor. Immunity 28, 391–401 (2008).
Liao, G., Zhang, M., Harhaj, E.W. & Sun, S.C. Regulation of the NF-kappaB-inducing kinase by tumor necrosis factor receptor-associated factor 3-induced degradation. J. Biol. Chem. 279, 26243–26250 (2004).
Vince, J.E. et al. IAP antagonists target cIAP1 to induce TNFalpha-dependent apoptosis. Cell 131, 682–693 (2007).
Keats, J.J. et al. Promiscuous mutations activate the noncanonical NF-kappaB pathway in multiple myeloma. Cancer Cell 12, 131–144 (2007).
Annunziata, C.M. et al. Frequent engagement of the classical and alternative NF-kappaB pathways by diverse genetic abnormalities in multiple myeloma. Cancer Cell 12, 115–130 (2007).
Yeh, W.C. et al. Early lethality, functional NF-kappaB activation, and increased sensitivity to TNF-induced cell death in TRAF2-deficient mice. Immunity 7, 715–725 (1997).
Xu, Y., Cheng, G. & Baltimore, D. Targeted disruption of TRAF3 leads to postnatal lethality and defective T-dependent immune responses. Immunity 5, 407–415 (1996).
Vaux, D.L. & Silke, J. IAPs, RINGs and ubiquitylation. Nat. Rev. Mol. Cell Biol. 6, 287–297 (2005).
Varfolomeev, E. et al. IAP antagonists induce autoubiquitination of c-IAPs, NF-kappaB activation, and TNFalpha-dependent apoptosis. Cell 131, 669–681 (2007).
Hu, S. & Yang, X. Cellular inhibitor of apoptosis 1 and 2 are ubiquitin ligases for the apoptosis inducer Smac/DIABLO. J. Biol. Chem. 278, 10055–10060 (2003).
Li, L. et al. A small molecule Smac mimic potentiates TRAIL- and TNFalpha-mediated cell death. Science 305, 1471–1474 (2004).
Cohen, J.J. Glucocorticoid-induced apoptosis in the thymus. Semin. Immunol. 4, 363–369 (1992).
Dinarello, C.A. & Mier, J.W. Lymphokines. N. Engl. J. Med. 317, 940–945 (1987).
Xie, P., Stunz, L.L., Larison, K.D., Yang, B. & Bishop, G.A. Tumor necrosis factor receptor-associated factor 3 is a critical regulator of B cell homeostasis in secondary lymphoid organs. Immunity 27, 253–267 (2007).
Nutt, S.L., Urbanek, P., Rolink, A. & Busslinger, M. Essential functions of Pax5 (BSAP) in pro-B cell development: difference between fetal and adult B lymphopoiesis and reduced V-to-DJ recombination at the IgH locus. Genes Dev. 11, 476–491 (1997).
Igarashi, H. et al. Early lymphoid progenitors in mouse and man are highly sensitive to glucocorticoids. Int. Immunol. 17, 501–511 (2005).
He, J.Q., Saha, S.K., Kang, J.R., Zarnegar, B. & Cheng, G. Specificity of TRAF3 in its negative regulation of the noncanonical NF-kappa B pathway. J. Biol. Chem. 282, 3688–3694 (2007).
Rothe, M., Pan, M.G., Henzel, W.J., Ayres, T.M. & Goeddel, D.V. The TNFR2-TRAF signaling complex contains two novel proteins related to baculoviral inhibitor of apoptosis proteins. Cell 83, 1243–1252 (1995).
Gallagher, E. et al. Kinase MEKK1 is required for CD40-dependent activation of the kinases Jnk and p38, germinal center formation, B cell proliferation and antibody production. Nat. Immunol. 8, 57–63 (2007).
Brown, K.D., Hostager, B.S. & Bishop, G.A. Differential signaling and tumor necrosis factor receptor-associated factor (TRAF) degradation mediated by CD40 and the Epstein-Barr virus oncoprotein latent membrane protein 1 (LMP1). J. Exp. Med. 193, 943–954 (2001).
Enzler, T. et al. Alternative and classical NF-kappa B signaling retain autoreactive B cells in the splenic marginal zone and result in lupus-like disease. Immunity 25, 403–415 (2006).
Sivakumar, V., Hammond, K.J., Howells, N., Pfeffer, K. & Weih, F. Differential requirement for Rel/nuclear factor kappa B family members in natural killer T cell development. J. Exp. Med. 197, 1613–1621 (2003).
Budanov, A.V., Sablina, A.A., Feinstein, E., Koonin, E.V. & Chumakov, P.M. Regeneration of peroxiredoxins by p53-regulated sestrins, homologs of bacterial AhpD. Science 304, 596–600 (2004).
Gurova, K.V., Hill, J.E., Razorenova, O.V., Chumakov, P.M. & Gudkov, A.V. p53 pathway in renal cell carcinoma is repressed by a dominant mechanism. Cancer Res. 64, 1951–1958 (2004).
Acknowledgements
We thank T. Mak (University of Toronto), H. Kikutani (Osaka University) and Amgen Inc. for the Traf2+/−, Traf3+/− and Map3k14+/− mouse strains; P.M. Chumakov (Cleveland Clinic) for the pLSLPw lentiviral vector; Z. Ronai and J. Reed (Burnham Institute) for some of the expression vectors; and Santa Cruz Biotechnology for the donated antibodies. Supported by the US National Institutes of Health (M.K., D.A.A.V. and H.W.), the Kanzawa Medical Research Foundation (A.M., in part), the American Lung Association (P.-H.T., in part), a Cancer Center core grant and the American Lebanese Syrian Associated Charities (D.A.A.V. and H.W.) and the American Cancer Society (M.K.).
Author information
Authors and Affiliations
Contributions
S.V. and M.K. planned and designed all experiments and wrote the manuscript. S.V. performed most experiments. A.M. performed the ubiquitination experiments. W.Z. performed immunoprecipitation experiments. H.W. and D.A.A.V. provided Lys63-specific anti-ubiquitin. J.J.K. and P.L.B. provided the cIAP-wild-type and cIAP-deficient multiple myeloma cells. P.-H.T. made CD40-expressing HEK-293T cells.
Corresponding author
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–8 (PDF 1288 kb)
Rights and permissions
About this article
Cite this article
Vallabhapurapu, S., Matsuzawa, A., Zhang, W. et al. Nonredundant and complementary functions of TRAF2 and TRAF3 in a ubiquitination cascade that activates NIK-dependent alternative NF-κB signaling. Nat Immunol 9, 1364–1370 (2008). https://doi.org/10.1038/ni.1678
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni.1678
This article is cited by
-
cIAP1/TRAF2 interplay promotes tumor growth through the activation of STAT3
Oncogene (2023)
-
TRAF3/STAT6 axis regulates macrophage polarization and tumor progression
Cell Death & Differentiation (2023)
-
B-cell maturation antigen targeting strategies in multiple myeloma treatment, advantages and disadvantages
Journal of Translational Medicine (2022)
-
Extracellular fibrinogen-binding protein released by intracellular Staphylococcus aureus suppresses host immunity by targeting TRAF3
Nature Communications (2022)
-
A20 undermines alternative NF-κB activity and expression of anti-apoptotic genes in Helicobacter pylori infection
Cellular and Molecular Life Sciences (2022)