Abstract
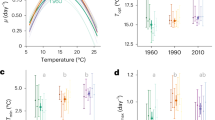
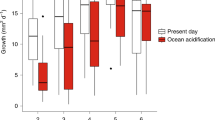
Ocean acidification, the drop in seawater pH associated with the ongoing enrichment of marine waters with carbon dioxide from fossil fuel burning, may seriously impair marine calcifying organisms. Our present understanding of the sensitivity of marine life to ocean acidification is based primarily on short-term experiments, in which organisms are exposed to increased concentrations of CO2. However, phytoplankton species with short generation times, in particular, may be able to respond to environmental alterations through adaptive evolution. Here, we examine the ability of the world’s single most important calcifying organism, the coccolithophore Emiliania huxleyi, to evolve in response to ocean acidification in two 500-generation selection experiments. Specifically, we exposed E. huxleyi populations founded by single or multiple clones to increased concentrations of CO2. Around 500 asexual generations later we assessed their fitness. Compared with populations kept at ambient CO2 partial pressure, those selected at increased partial pressure exhibited higher growth rates, in both the single- and multiclone experiment, when tested under ocean acidification conditions. Calcification was partly restored: rates were lower under increased CO2 conditions in all cultures, but were up to 50% higher in adapted compared with non-adapted cultures. We suggest that contemporary evolution could help to maintain the functionality of microbial processes at the base of marine food webs in the face of global change.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Change history
20 November 2012
In the version of this article originally published, the y axis scales of Fig. 3c–f were incorrect. This has been corrected in the PDF and HTML versions.
References
Orr, J. C. et al. Anthropogenic ocean acidification over the twenty-first century and its impact on calcifying organisms. Nature 437, 681–686 (2005).
Feely, R. A. et al. Impact of anthropogenic CO2 on the CaCO3 system in the oceans. Science 305, 362–366 (2004).
Caldeira, K. & Wickett, M. E. Oceanography: Anthropogenic carbon and ocean pH. Nature 425, 365–365 (2003).
Fabry, V. J., Seibel, B. A., Feely, R. A. & Orr, J. C. Impacts of ocean acidification on marine fauna and ecosystem processes. ICES J. Mar. Sci. 65, 414–432 (2008).
Frommel, A. Y. et al. Severe tissue damage in Atlantic cod larvae under increasing ocean acidification. Nature Clim. Change 2, 42–46 (2012).
Hoegh-Guldberg, O. et al. Coral reefs under rapid climate change and ocean acidification. Science 318, 1737–1742 (2007).
Fabricius, K. E. et al. Losers and winners in coral reefs acclimatized to elevated carbon dioxide concentrations. Nature Clim. Change 1, 165–169 (2011).
Hall-Spencer, J. M. et al. Volcanic carbon dioxide vents show ecosystem effects of ocean acidification. Nature 454, 96–99 (2008).
Westbroek, P., Young, J. R. & Linschooten, K. Coccolith production (biomineralization) in the marine alga Emiliania huxleyi. J. Eukaryot. Microbiol. 36, 368–373 (1989).
Armstrong, R. A., Lee, C., Hedges, J. I., Honjo, S. & Wakeham, S. G. A new, mechanistic model for organic carbon fluxes in the ocean based on the quantitative association of POC with ballast minerals. Deep-Sea Res. II 49, 219–236 (2001).
Riebesell, U. et al. Reduced calcification of marine plankton in response to increased atmospheric CO2 . Nature 407, 364–367 (2000).
Zondervan, I. The effects of light, macronutrients, trace metals and CO2 on the production of calcium carbonate and organic carbon in coccolithophores—a review. Deep-Sea Res. II 54, 521–537 (2007).
Riebesell, U. & Tortell, P. D. in Ocean Acidification (eds Gattuso, J-P. & Hansson, L.) 99–121 (Oxford Univ. Press, 2011).
Collins, S. Comment on Effects of long-term high CO2 exposure on two species of coccolithophores by Müller et al. (2010). Biogeosci. Disc. 7, 2673–2679 (2010).
Riebesell, U., Körtzinger, A. & Oschlies, A. Sensitivities of marine carbon fluxes to ocean change. Proc. Natl Acad. Sci. USA 106, 20602–20609 (2009).
Joint, I., Doney, S. C. & Karl, D. M. Will ocean acidification affect marine microbes? ISME J. 5, 1–7 (2011).
Collins, S. Many possible worlds: Expanding the ecological scenarios in experimental evolution. Evol. Biol. 38, 3–14 (2011).
Hoffmann, A. A. & Sgro, C. M. Climate change and evolutionary adaptation. Nature 470, 479–485 (2011).
Reusch, T. B. H. & Wood, T. E. Molecular ecology of global change. Mol. Ecol. 16, 3973–3992 (2007).
Collins, S. L. & Bell, G. Phenotypic consequences of 1,000 generations of selection at elevated CO2 in a green alga. Nature 431, 566–569 (2004).
Elena, S. F. & Lenski, R. E. Evolution experiments with microorganisms: The dynamics and genetic bases of adaptation. Nature Rev. Genet. 4, 457–469 (2003).
Lenski, R., Rose, M., Simpson, S. & Tadler, S. Long-term experimental evolution in Escherichia coli. I. Adaptation and divergence during 2,000 generations. Am. Nat. 138, 1315–1341 (1991).
Becks, L., Ellner, S. P., Jones, L. E. & Hairston, N. G. Jr Reduction of adaptive genetic diversity radically alters eco-evolutionary community dynamics. Ecol. Lett. 13, 989–997 (2010).
IPCC Climate Change 2007: Impacts, Adaptation and Vulnerability (eds Parry, M. L., Canziani, O. F., Palutikof, J. P., van der Linden, P. J. & Hanson, C. E.) (Cambridge Univ. Press, 2007).
Feely, R. A., Sabine, C. L., Hernandez-Ayon, J. M., Ianson, D. & Hales, B. Evidence for upwelling of corrosive ‘acidified’ water onto the continental shelf. Science 320, 1490–1492 (2008).
Bennett, A. F. & Lenski, R. E. Evolutionary adaptation to temperature. II thermal niches of experimental lines of Escherichia coli. Evolution 47, 1–12 (1993).
Lenski, R. E. et al. Evolution of competitive fitness in experimental populations of E. coli: What makes one genotype a better competitor than another? Anton. van Leeuw. 73, 35–47 (1998).
Gerrish, P. & Lenski, R. The fate of competing beneficial mutations in an asexual population. Genetica 102–103, 127–144 (1998).
Zondervan, I., Zeebe, R. E., Rost, B. & Riebesell, U. A time series study of silica production and flux in an eastern boundary region: Santa Barbara Basin, California. Glob. Biogeochem. Cycles 15, 507–516 (2001).
Mackinder, L. et al. Expression of biomineralization-related ion transport genes in Emiliania huxleyi. Environ. Microbiol. 13, 3250–3265 (2011).
Paasche, E. A review of the coccolithophorid Emiliania huxleyi (Prymnesiophyceae), with particular reference to growth, coccolith formation, and calcification-photosynthesis interactions. Phycologia 40, 503–529 (2002).
Zeyl, C., Vanderford, T. & Carter, M. An evolutionary advantage of haploidy in large yeast populations. Science 299, 555–558 (2003).
Desai, M. M., Fisher, D. S. & Murray, A. W. The speed of evolution and maintenance of variation in asexual populations. Curr. Biol. 17, 385–394 (2007).
Travisano, M., Vasi, F. & Lenski, R. E. Long-term experimental evolution in Escherichia coli. III. Variation among replicate populations in correlated responses to novel environments. Evolution 49, 189–200 (1995).
Iglesias-Rodriguez, M. D., Schofield, O. M., Batley, J., Medlin, L. K. & Hayes, P. K. Intraspecific genetic diversity in the marine coccolithophore Emiliania huxleyi (Prymnesiophyceae): The use of microsatellite analysis in marine phytoplankton population studies. J. Phycol. 42, 526–536 (2006).
Langer, G., Nehrke, G., Probert, I., Ly, J. & Ziveri, P. Strainspecific responses of Emiliania huxleyi to changing seawater carbonate chemistry. Biogeosciences 6, 2637–2646 (2009).
Kaltz, O. & Bell, G. The ecology and genetics of fitness in Chlamydomonas. XII: Repeated sexual episodes increase rates of adaptation to novel environments. Evolution 56, 1743–1753 (2002).
Beaufort, L. et al. Sensitivity of coccolithophores to carbonate chemistry and ocean acidification. Nature 476, 80–83 (2011).
Falkowski, P. G. & Oliver, M. J. Mix and match: How climate selects phytoplankton. Nature Rev. Microbiol. 5, 813–819 (2007).
Acknowledgements
We thank J. Meyer, A. Zavišić, K. Beining, A. Ludwig, S. Fessler and P. Fritsche for laboratory assistance; J. Czerny and C. Eizaguirre for advice on the experimental design, H. Schulenburg, J. Olsen and O. Roth for comments on earlier drafts; L. Bach, S. Febiri, T. Großkopf, L. Mackinder, D. Haase and K. Schulz for support during the experiments. T.B.H.R. and U.R. received financial support for this project from the German Federal Ministry of Education and Research (BMBF; project BIOACID).
Author information
Authors and Affiliations
Contributions
T.B.H.R. conceived the project, all authors designed the experiment and K.T.L. carried out the experiment. All authors analysed and interpreted the data and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Information (PDF 495 kb)
Supplementary Information
Supplementary Information (XLS 29 kb)
Rights and permissions
About this article
Cite this article
Lohbeck, K., Riebesell, U. & Reusch, T. Adaptive evolution of a key phytoplankton species to ocean acidification. Nature Geosci 5, 346–351 (2012). https://doi.org/10.1038/ngeo1441
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ngeo1441
This article is cited by
-
Systematic review of the uncertainty of coral reef futures under climate change
Nature Communications (2024)
-
The genetic basis of the leafy seadragon’s unique camouflage morphology and avenues for its efficient conservation derived from habitat modeling
Science China Life Sciences (2023)
-
Malformation in coccolithophores in low pH waters: evidences from the eastern Arabian Sea
Environmental Science and Pollution Research (2023)
-
Intraspecific variation in metal tolerance modulate competition between two marine diatoms
The ISME Journal (2022)
-
Selection on offspring size and contemporary evolution under ocean acidification
Nature Climate Change (2022)