Abstract
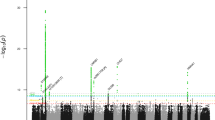
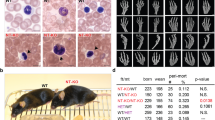
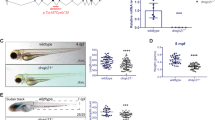
Pelger–Huët anomaly (PHA; OMIM *169400) is an autosomal dominant disorder characterized by abnormal nuclear shape and chromatin organization in blood granulocytes. Affected individuals show hypolobulated neutrophil nuclei with coarse chromatin. Presumed homozygous individuals have ovoid neutrophil nuclei, as well as varying degrees of developmental delay, epilepsy and skeletal abnormalities1,2,3. Homozygous offspring in an extinct rabbit lineage showed severe chondrodystrophy, developmental anomalies and increased pre- and postnatal mortality4,5. Here we show, by carrying out a genome-wide linkage scan, that PHA is linked to chromosome 1q41–43. We identified four splice-site, two frameshift and two nonsense mutations in LBR, encoding the lamin B receptor. The lamin B receptor (LBR), a member of the sterol reductase family6, is evolutionarily conserved and integral to the inner nuclear membrane; it targets heterochromatin and lamins to the nuclear membrane7,8. Lymphoblastoid cells from heterozygous individuals affected with PHA show reduced expression of the lamin B receptor, and cells homozygous with respect to PHA contain only trace amounts of it. We found that expression of the lamin B receptor affects neutrophil nuclear shape and chromatin distribution in a dose-dependent manner. Our findings have implications for understanding nuclear envelope–heterochromatin interactions, the pathogenesis of Pelger-like conditions in leukemia9, infection10 and toxic drug reactions11, and the evolution of neutrophil nuclear shape12.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Haverkamp Begemann, N. & van Lookeren Campagne, A. Homozygous form of Pelger–Huët's nuclear anomaly in man. Acta Haematol. Basel 7, 295–303 (1952).
Aznar, J. & Vaya, A. Homozygous form of the Pelger–Huët leukocyte anomaly in man. Acta Haematol. Basel 66, 59–62 (1981).
Siegert, E., Beier, L. & Gräbner, H. Ein Beitrag zur homozygoten Form der Pelger–Huëtschen–Kernanomalie. Kinderarztl. Prax. 51, 164–169 (1983).
Undritz, E. Das ausschliessliche Vorkommen reifer rundkerniger Leukozyten bei der reingezüchteten Pelger–Huëtschen Anomalie des Kaninchens und die Bedeutung der Pelger–Leukozyten in der vergleichenden Hämatologie. Folia Haemat. 67, 249–291 (1943).
Nachtsheim, H. The Pelger–Anomaly in man and rabbit. J. Hered. XLI, 131–137 (1950).
Holmer, L., Pezhman, A. & Worman, H.J. The human lamin B receptor/sterol reductase multigene family. Genomics 54, 469–476 (1998).
Ye, Q., Callebaut, I., Pezhman, A., Courvalin, J.C. & Worman, H.J. Domain-specific interactions of human HP1-type chromodomain proteins and inner nuclear membrane protein lamin B receptor. J. Biol. Chem. 272, 14983–14989 (1997).
Schuler, E., Lin, F. & Worman, H.J. Characterization of the human gene encoding lamin B receptor, an integral protein of the nuclear envelope inner membrane. J. Biol. Chem. 269, 11312–11317 (1994).
Sainty, D. et al. A new morphologic classification system for acute promyelocytic leukemia distinguishes cases with underlying PLZF/RARA gene rearrangements. Blood 96, 1287–1296 (2000).
Shenkenberg, T.D., Rice, L. & Waddell, C.C. Acquired Pelger–Huët nuclear anomaly with tuberculosis. Arch. Intern. Med. 142, 153–154 (1982).
Juneja, S.K. et al. Association of acquired Pelger–Huët anomaly with taxoid therapy. Br. J. Haematol. 93, 139–141 (1996).
Undritz, E. Das Pelger–Huëtsche Blutbild beim Tier und seine Bedeutung für die Entwicklungsgeschichte des Blutes. Schweiz. Med. Wochenschr. 69, 1177–1186 (1939).
Johansson, E.O. The Pelger Anomaly: Genetic, Cytologic and Epidemiologic Aspects of Some Data from Sweden. Thesis, Uppsala Univ. (1963).
Skendzel, L.P. & Hoffman, G.C. The Pelger anomaly of leukocytes: forty-one cases in seven families. Am. J. Clin. Pathol. 37, 294–301 (1962).
Karl, H. Die besondere Häufung von Merkmalsträgern der Pelger–Huët–Kernanomalie der Leukozyten in Gelenau im Erzgebirge. Thesis, Humboldt Univ., Berlin (1967).
Green, M.C., Shultz, L.D. & Nedzi, L.A. Abnormal nuclear morphology of leukocytes in the mouse mutant ichthyosis. Transplantation 20, 172–175 (1975).
Schirmer, E.C., Guan, T. & Gerace, L. Involvement of the lamin rod domain in heterotypic lamin interactions important for nuclear organization. J. Cell Biol. 153, 479–489 (2001).
Liu, J. et al. Essential roles for Caenorhabditis elegans lamin gene in nuclear organization, cell cycle progression, and spatial organization of nuclear pore complexes. Mol. Biol. Cell 11, 3937–3947 (2000).
Lenz–Böhme, B. et al. Insertional mutation of the Drosophila nuclear lamin Dm0 gene results in defective nuclear envelopes, clustering of nuclear pore complexes, and accumulation of annulate lamellae. J. Cell Biol. 137, 1001–1016 (1997).
Fatkin, D. et al. Missense mutations in the rod domain of the lamin A/C gene as causes of dilated cardiomyopathy and conduction-system disease. N. Engl. J. Med. 341, 1715–1724 (1999).
Bonne, G. et al. Mutations in the gene encoding lamin A/C cause autosomal dominant Emery–Dreifuss muscular dystrophy. Nature Genet. 21, 285–288 (1999).
Shackleton, S. et al. LMNA, encoding lamin A/C, is mutated in partial lipodystrophy. Nature Genet. 24, 153–156 (2000).
Ognibene, A. et al. Nuclear changes in a case of X-linked Emery–Dreifuss muscular dystrophy. Muscle Nerve 22, 864–869 (1999).
Vigouroux, C. et al. Nuclear envelope disorganization in fibroblasts from lipodystrophic patients with heterozygous R482Q/W mutations in the lamin A/C gene. J. Cell Sci. 114, 4459–4468 (2001).
Latimer, K.S., Rowland, G.N. & Mahaffey, M.B. Homozygous Pelger–Huët anomaly and chondrodysplasia in a stillborn kitten. Vet. Pathol. 25, 325–328 (1988).
Silve, S., Dupuy, P.H., Ferrara, P. & Loison, G. Human lamin B receptor exhibits sterol C14-reductase activity in Saccharomyces cerevisiae. Biochim. Biophys. Acta 1392, 233–244 (1998).
Fitzky, B.U. et al. Mutations in the Δ7-sterol reductase gene in patients with the Smith–Lemli–Opitz syndrome. Proc. Natl Acad. Sci. USA 95, 8181–8186 (1998).
Hoger, T.H., Zatloukal, K., Waizenegger, I. & Krohne, G. Characterization of a second highly conserved B-type lamin present in cells previously thought to contain only a single B-type lamin. Chromosoma 99, 379–390 (1990).
Olins, A.L. et al. Nuclear envelope and chromatin compositional differences comparing undifferentiated and retinoic acid- and phorbol ester-treated HL-60 cells. Exp. Cell Res. 268, 115–127 (2001).
Ye, Q. & Worman, H.J. Primary structure analysis and lamin B and DNA binding of human lamin B receptor, an integral protein of the nuclear envelope inner membrane. J. Biol. Chem. 269, 11306–11311 (1994).
Acknowledgements
We thank all families for their interest and participation; H. Stobbe, R. Witkowski, A. Harder, K. Saar and T.F. Wienker for help in initiating the study; S. Rothe, R. Eckhardt, H. Neitzel, H. Toennies for technical assistance and establishing cell lines; G. Nürnberg and F. Rüschendorf for bioinformatic support; P. Wittig and R. Wolf for preparing blood smears; and F.C. Luft, P. Nürnberg and P. Lichter for support. This work has been supported by a grant from the Deutsche Forschungsgemeinschaft and from the US National Institutes of Health. A.L.O. and D.E.O. were supported by grants from the German Cancer Research Center and from the Davis Family Foundation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Rights and permissions
About this article
Cite this article
Hoffmann, K., Dreger, C., Olins, A. et al. Mutations in the gene encoding the lamin B receptor produce an altered nuclear morphology in granulocytes (Pelger–Huët anomaly). Nat Genet 31, 410–414 (2002). https://doi.org/10.1038/ng925
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng925
This article is cited by
-
Nuclear morphology is shaped by loop-extrusion programs
Nature (2024)
-
A biallelic loss-of-function variant in TMEM147 causes profound intellectual disability and spasticity
neurogenetics (2023)
-
The transcription factor PREP1(PKNOX1) regulates nuclear stiffness, the expression of LINC complex proteins and mechanotransduction
Communications Biology (2022)
-
Deletion of LBR N-terminal domains recapitulates Pelger-Huet anomaly phenotypes in mouse without disrupting X chromosome inactivation
Communications Biology (2021)
-
Two novel cases further expand the phenotype of TOR1AIP1-associated nuclear envelopathies
Human Genetics (2020)