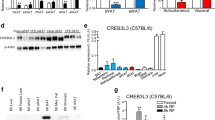
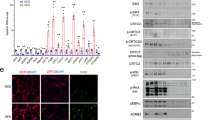
Abstract
The CBP protein (cAMP response element binding protein (CREB) binding protein)1 is a co-activator2 for several transcription factors with a wide range of important biological functions, such as sterol regulatory element binding proteins (SREBPs)3, CCAAT/enhancer-binding proteins (C/EBPs)4, nuclear receptors5,6 (including peroxisome proliferator–activated receptors, PPARs)7, and signal transducers and activators of transcription (STATs)8. In contrast to these individual transcription factors, the biological roles of CBP are poorly understood. CBP enhances transcriptional activities via histone acetylation and the recruitment of additional co-activators such as SRC (steroid coactivator)−1 (ref. 9). To identify its physiological functions using a loss-of-function mutant, we analyzed CBP-deficient mice10,11,12. As Crebbp null mice (Crebbp−/−) died during embryogenesis10,11,12, we used Crebbp+/− mice12. Unexpectedly, Crebbp+/− mice showed markedly reduced weight of white adipose tissue (WAT) but not of other tissues. Despite this lipodystrophy, Crebbp+/− mice showed increased insulin sensitivity and glucose tolerance and were completely protected from body weight gain induced by a high-fat (HF) diet. We observed increased leptin sensitivity and increased serum adiponectin levels in Crebbp+/− mice. These increased effects of insulin-sensitizing hormones secreted from WAT may explain, at least in part, the phenotypes of Crebbp+/− mice. This study demonstrates that CBP may function as a 'master-switch' between energy storage and expenditure.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Goodman, R.H. & Smolik, S. CBP/p300 in cell growth, transformation, and development. Genes Dev. 14, 1553–1577 (2000).
Westin, S., Rosenfeld, M.G. & Glass, C.K. Nuclear receptor coactivators. Adv. Pharmacol. 47, 89–112 (2000).
Brown, M.S., Ye, J., Rawson, R.B. & Goldstein, J.L. Regulated intramembrane proteolysis: a control mechanism conserved from bacteria to humans. Cell 100, 391–398 (2000).
Darlington, G.J., Ross, S.E. & MacDougald, O.A. The role of C/EBP genes in adipocyte differentiation. J. Biol. Chem. 273, 30057–30060 (1998).
Perlmann, T. & Evans, R.M. Nuclear receptors in Sicily: all in the famiglia. Cell 90, 391–397 (1997).
Mark, M. et al. A genetic dissection of the retinoid signalling pathway in the mouse. Proc. Nutr. Soc. 58, 609–613 (1999).
Kersten, S., Desvergne, B. & Wahli, W. Roles of PPARs in health and disease. Nature 405, 421–424 (2000).
Horvath, C.M. STAT proteins and transcriptional responses to extracellular signals. Trends. Biochem. Sci. 25, 496–502 (2000).
McKenna, N.J. & O'Malley, B.W. From ligand to response: generating diversity in nuclear receptor coregulator function. J. Steroid Biochem. Mol. Biol. 74, 255–259 (2000).
Tanaka, Y. et al. Abnormal skeletal patterning in embryos lacking a single Cbp allele: a partial similarity with Rubinstein-Taybi syndrome. Proc. Natl Acad. Sci. USA 94, 10215–10220 (1997).
Yao, T.P., et al. Gene dosage-dependent embryonic development and proliferation defects in mice lacking the transcriptional integrator p300. Cell 93, 361–372 (1998).
Oike, Y. et al. Truncated CBP protein leads to classical Rubinstein-Taybi syndrome phenotypes in mice: implications for a dominant-negative mechanism. Hum. Mol. Genet. 8, 387–396 (1999).
Kubota, N. et al. PPAR gamma mediates high-fat diet-induced adipocyte hypertrophy and insulin resistance. Mol. Cell 4, 597–609 (1999).
Rosen, E.D., Walkey, C.J., Puigserver, P. & Spiegelman, B.M. Transcriptional regulation of adipogenesis. Genes Dev. 14, 1293–1307 (2000).
Kim, J.B. et al. Dual DNA binding specificity of ADD1/SREBP1 controlled by a single amino acid in the basic helix-loop-helix domain. Mol. Cell. Biol. 15, 2582–2588 (1995).
Kliewer, S.A. et al. Differential expression and activation of a family of murine peroxisome proliferator-activated receptors. Proc. Natl Acad. Sci. USA 91, 7355–7359 (1994).
Houstek, J., Kopecky, J., Rychter, Z. & Soukup, T. Uncoupling protein in embryonic brown adipose tissue existence of nonthermogenic and thermogenic mitochondria. Biochim. Biophys. Acta 935, 19–25 (1988).
Ailaud, G. & Hauner, H. Development of white adipose tissue. in Handbook of Obesity (eds Bray, G.A., Bouchard, C. and James, W.P.T.) 359–378 (Marcel Dekker, New York, 1998).
Friedman, J.M. Obesity in the new millennium. Nature 404, 632–634 (2000).
Yamauchi, T. et al. Replenishment of the fat-derived hormone adiponectin reverses insulin resistance associated with both lipoatrophy and obesity. Nature Med. 7, 941–946 (2001).
Berg, A.H., Combs, T.P., Du, X., Brownlee, M. & Scherer, P.E. The adipocyte-secreted protein Acrp30 enhances hepatic insulin action. Nature Med. 7, 947–953 (2001).
Yamauchi, T. et al. The mechanisms by which both heterozygous PPARγ deficiency and PPARγ agonist improve insulin resistance. J. Biol. Chem. 276, 41245–41254 (2001).
Shulman, G.I. Cellular mechanisms of insulin resistance. J. Clin. Invest. 106, 171–176 (2000).
Hotamisligil, G.S. The role of TNFalpha and TNF receptors in obesity and insulin resistance. J. Intern. Med. 245, 621–625 (1999).
Murakami, K. et al. A novel insulin sensitizer acts as a coligand for peroxisome proliferator-activated receptor-alpha (PPAR-alpha) and PPAR-gamma: effect of PPAR-alpha activation on abnormal lipid metabolism in liver of Zucker fatty rats. Diabetes 47, 1841–1847 (1998).
Lowell, B.B. & Spiegelman, B.M. Towards a molecular understanding of adaptive thermogenesis. Nature 404, 652–660 (2000).
Miles, P.D., Barak, Y., He, W., Evans, R.M. & Olefsky, J.M. Improved insulin-sensitivity in mice heterozygous for PPAR-gamma deficiency. J. Clin. Invest. 105, 287–292 (2000).
Fruebis, J. et al. Proteolytic cleavage product of 30-kDa adipocyte complement-related protein increases fatty acid oxidation in muscle and causes weight loss in mice. Proc. Natl Acad. Sci. USA 98, 2005–2010 (2001).
Gavrilova, O. et al. Surgical implantation of adipose tissue reverses diabetes in lipoatrophic mice. J. Clin. Invest. 105, 271–278 (2000).
Nakazato, M. et al. A role for ghrelin in the central regulation of feeding. Nature 409, 194–198 (2001).
Acknowledgements
We are grateful to S. Uchida, K. Kirii, S. Sakata and T. Nagano for their excellent technical assistance. This work was supported by a grant from the Research Fellowships of the Japan Society for the Promotion of Science for Young Scientists (to T.Y.), a grant from the Human Science Foundation (to T.K.), a Grant-in-Aid for the Development of Innovative Technology from the Ministry of Education, Culture, Sports, Science and Technology (to T. K.), a Grant-in Aid for Creative Scientific Research from the Japan Society for the Promotion of Science (to T. K.), and by Health Science Research Grants (Research on Human Genome and Gene Therapy) from the Ministry of Health and Welfare (to T. K.).
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Rights and permissions
About this article
Cite this article
Yamauchi, T., Oike, Y., Kamon, J. et al. Increased insulin sensitivity despite lipodystrophy in Crebbp heterozygous mice. Nat Genet 30, 221–226 (2002). https://doi.org/10.1038/ng829
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng829
This article is cited by
-
Browning of the white adipose tissue regulation: new insights into nutritional and metabolic relevance in health and diseases
Nutrition & Metabolism (2022)
-
Selective inhibition of CBP/p300 HAT by A-485 results in suppression of lipogenesis and hepatic gluconeogenesis
Cell Death & Disease (2020)
-
Protein acetylation in metabolism — metabolites and cofactors
Nature Reviews Endocrinology (2016)
-
Kat3 coactivators in somatic stem cells and cancer stem cells: biological roles, evolution, and pharmacologic manipulation
Cell Biology and Toxicology (2016)
-
Role of transcription factor acetylation in the regulation of metabolic homeostasis
Protein & Cell (2015)